Abstract
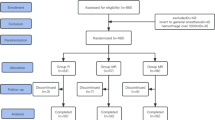
Recently, most studies reported magnesium as a N-methyl-d-aspartate receptor antagonist and its analgesic and perioperative anaesthetic effects have been discussed with central desensitization pathway. We investigated the effects of caudal ropivacaine plus magnesium and compared with ropivacaine alone on postoperative analgesia requirements. After hospital ethic committee’s consent, 60 patients (ASA I-II, 2–10 years old) who had lower abdominal or penoscrotal surgery were enrolled in the study. After general anaesthesia induction, caudal blockage was applied. Patients were randomly assigned in two groups. Ropivacaine 0.25% was administered to Group R (n = 37), ropivacaine 0.25% plus 50 mg magnesium to Group RM (n = 23) in 0.5 ml kg−1 volume. Postoperative analgesia level was recorded at 15 min and 1, 2, 3, 4, 6 h by using Paediatric Objective Pain Scale (POPS) and The Children’s Hospital of Eastern Ontoria Pain Scale (CHEOPS). Postoperative motor blocks were evaluated with Modified Bromage Motor Block Scale. According to demographic characteristics, there were no significant differences between the two groups (P > 0.05). POPS, CHEOPS, Bromage Motor Scales, analgesia duration and adverse effects were similar in Group R and Group RM. It has been shown that addition of magnesium as an adjuvant agent to local anaesthetics for caudal analgesia has no effect on postoperative pain and analgesic need.
Similar content being viewed by others
References
Dalens B, Hasnoui (1989) A caudal anesthesia in pediatric surgery. Success rate and adverse effects in 750 consecutive patients. Anesth Analg 68:83–89
Giaufré E,Dalens B, Gombert A (1996) Epidemiology and morbidity of regional anesthesia in children a one-year prospective survey of the French Language Society of Pediatric Anesthesiologists. Anesth Analg 83:904–912
Gentili M, Huu PC, Enel D, Hollande J, Bonnet F (1998) Sedation depends on the level of sensory block induced by spinal anaesthesia. Br J Anesth 81:970–971
Hannallah RS (2001) Outpatient anesthesia. In: Coté C, Todres ID, Ryan JF, Goudsouzian NG (eds) A practise of anesthesia for infants, children, 3rd edn. W.B. Saunders, Philadelphia, p 61
Constant I, Gall O, Gouyet L, Chauvin M, Murat I (1998) Addition of clonidine or fentanyl to local anaesthetics prolongs the duration of surgical analgesia after single shot caudal block in children. Br J Anesth 80:294–298
Cook B, Grubb DJ, Aldridge LA, Doyle E (1995) Comparison of the effects of adrenaline, clonidine and ketamine on the duration of caudal analgesia produced by bupivacaine in children. Br J Anaesth 75:698–701
Naguib M, Sharif AMY, Seraj M, el Gammal M, Dawlatly AA (1991) Ketamine for caudal analgesia in children comparison with caudal bupivacaine. Br J Anaesth 67:559–564
Semple D, Findlow D, Aldrige LM, Doyle E (1996) The optimal dose of ketamine for caudal epidural blockade in children. Anaesthesia 51:1170–1172
James MFM 1992 Clinical use of magnesium infusions in anaesthesia. Anesth Analg 74:129–136
Woolf CJ, Thompson SW (1991) The induction and maintenance of central sensitization is dependent on N-methyl-d-aspartic acid receptor activation implications for the treatment of post-injury pain and hypersensitivity states. Pain 44:293–299
Koinig H, Wallner T, Marhofer P, Andel H, Hörauf K, Mayer N (1998) Magnesium sulfate reduces intra and postoperative analgesic requirements. Anesth Analg 87:206–210
Ko SH, Lim HR, Kim DC, Han YJ, Choe H, Song HS (2001) Magnesium sulfate does not reduce postoperative analgesic requirements. Anesthesiology 95:640–646
Kroin JS, McCarthy RJ, Von Roenn N, Schwab B, Tuman KJ, Ivankovich AD (2000) Magnesium sulfate potentiates morphine antinociception at the spinal level. Anesth Analg 90:13–17
Xiao W, Bennett G (1994) Magnesium suppresses neuropathic pain response in rats via spinal site of action. Brain Res 666:168–172
Chanimov M, Cohen ML, Grinspun Y, Herbert M, Reif R, Kaufman I, Bahar M (1997) Neurotoxicity after spinal anaesthesia induced by serial intrathecal injections of magnesium sulphate an experimental study in a rat model. Anaesthesia 52:223–228
Simpson JI, Eide TR, Schiff GA, Clagnaz JF, Hossain I, Tverskoy A, Koski G (1994) Intrathecal magnesium sulfate protects the spinal cord from ischemic injury during thoracic aortic cross clamping. Anesthesiology 81:1493–1499
Liu HT, Hollmann MW, Liu WH, Hoenemann CW, Durieux ME (2001) Modulation of NMDA receptor function by ketamine and magnesium: Part I. Anesth Analg 92:1173–1181
O’Flaherty JE, Lın CX (2003) Does ketamine or magnesium affect posttonsillectomy pain in children? Paediatr Anaesth 13:413–421
Lejuste MJLR (1985) Inadvertent intrathecal administration of magnesium sulfate. S Afr Med J 68:367–368
Buvanendran A, McCarthy JR, Kroin SJ, Leong W, Perry P, Tuman KJ (2002) Intrathecal magnesium prolongs fentanyl analgesia: a prospective, randomized, controlled trial. Anesth Analg 95:661–666
Perry P, Buvanendran A, Kroin J et al (2001) Intrathecal magnesium sulfate prolongs fentanyl labor analgesia in the lateral compared to the sitting position: effect of baricity. Anesth Analg 92:S301
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Birbicer, H., Doruk, N., Cinel, I. et al. Could adding magnesium as adjuvant to ropivacaine in caudal anaesthesia improve postoperative pain control?. Pediatr Surg Int 23, 195–198 (2007). https://doi.org/10.1007/s00383-006-1779-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-006-1779-4