Abstract
Introduction
Conventional magnetic resonance imaging (MRI), which is mainly used to detect complications, is ineffective in determining the neurological status of patients with meningitis. Hemodynamic change in the brain may be more indicative of the neurological status but few imaging studies have verified this. Arterial spin-labeling (ASL) perfusion, a noninvasive MR method requiring no contrast agent injection, can be used to measure cerebral blood flow (CBF).
Case reports
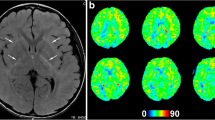
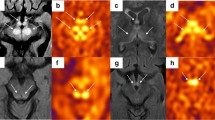
We describe three pediatric patients with meningitis, who all showed regions of increased CBF on perfusion imaging. One patient, presenting with headache and conscious disturbance, had CBF changes in the frontal, temporal, and occipital regions. The other two patients, presenting with hallucinations, memory deficits, and seizures, had CBF changes in the frontal and temporal regions.
Conclusion
ASL perfusion imaging may be helpful in assessing patients with meningitis, demonstrating CBF changes more strongly correlating with the neurological status, and detecting active brain abnormalities.



Similar content being viewed by others
References
Mohan S, Jain KK, Arabi M, Shah GV (2012) Imaging of meningitis and ventriculitis. Neuroimaging Clin N Am 22:557–583
Tunkel AR, Scheld WM (1993) Pathogenesis and pathophysiology of bacterial meningitis. Clin Microbiol Rev 6:118–136
Forderreuther S, Tatsch K, Einhaupl KM, Pfister HW (1992) Abnormalities of cerebral blood flow in the acute phase of bacterial meningitis in adults. J Neurol 239:431–436
Tureen JH, Dworkin RJ, Kennedy SL, Sachdeva M, Sande MA (1990) Loss of cerebrovascular autoregulation in experimental meningitis in rabbits. J Clin Invest 85:577–581
Jarnum H, Steffensen EG, Knutsson L, Frund ET, Simonsen CW, Lundbye-Christensen S, Shankaranarayanan A, Alsop DC, Jensen FT, Larsson EM (2010) Perfusion MRI of brain tumours: a comparative study of pseudo-continuous arterial spin labelling and dynamic susceptibility contrast imaging. Neuroradiology 52:307–317
Wong AM, Yan FX, Liu HL (2014) Comparison of three-dimensional pseudo-continuous arterial spin labeling perfusion imaging with gradient-echo and spin-echo dynamic susceptibility contrast MRI. J Magn Reson Imaging 39:427–433
Lennox BR, Park SB, Medley I, Morris PG, Jones PB (2000) The functional anatomy of auditory hallucinations in schizophrenia. Psychiatry Res 100:13–20
Chaudhary UJ, Duncan JS (2014) Applications of blood-oxygen-level-dependent functional magnetic resonance imaging and diffusion tensor imaging in epilepsy. Neuroimaging Clin N Am 24:671–694
McAndrews MP (2014) Memory assessment in the clinical context using functional magnetic resonance imaging: a critical look at the state of the field. Neuroimaging Clin N Am 24:585–597
Wintermark M, Sesay M, Barbier E, Borbely K, Dillon WP, Eastwood JD, Glenn TC, Grandin CB, Pedraza S, Soustiel JF, Nariai T, Zaharchuk G, Caille JM, Dousset V, Yonas H (2005) Comparative overview of brain perfusion imaging techniques. Stroke J Cereb Circ 36:e83–e99
Moller K, Hogh P, Larsen FS, Strauss GI, Skinhoj P, Sperling BK, Knudsen GM (2000) Regional cerebral blood flow during hyperventilation in patients with acute bacterial meningitis. Clin Physiol 20:399–410
Nishikawa M, Matsubara T, Yoshitomi T, Ichiyama T, Hayashi T, Furukawa S (2000) Abnormalities of brain perfusion in echovirus type 30 meningitis. J Neurol Sci 179:122–126
Merkelbach S, Muller M, Huber G, Schimrigk K (1998) Alteration of cerebral blood flow in patients with bacterial and viral meningoencephalitis. AJNR Am J Neuroradiol 19:433–438
Pedersen M, Brandt CT, Knudsen GM, Ostergaard C, Skinhoj P, Frimodt-Moller N, Moller K (2007) Cerebral blood flow autoregulation in early experimental S. pneumoniae meningitis. J Appl Physiol 102:72–78
Moller K, Larsen FS, Qvist J, Wandall JH, Knudsen GM, Gjorup IE, Skinhoj P (2000) Dependency of cerebral blood flow on mean arterial pressure in patients with acute bacterial meningitis. Crit Care Med 28:1027–1032
Baldwin AC, Kielian T (2004) Persistent immune activation associated with a mouse model of Staphylococcus aureus-induced experimental brain abscess. J Neuroimmunol 151:24–32
van der Flier M, Hoppenreijs S, van Rensburg AJ, Ruyken M, Kolk AH, Springer P, Hoepelman AI, Geelen SP, Kimpen JL, Schoeman JF (2004) Vascular endothelial growth factor and blood-brain barrier disruption in tuberculous meningitis. Pediatr Infect Dis J 23:608–613
Haris M, Gupta RK, Singh A, Husain N, Husain M, Pandey CM, Srivastava C, Behari S, Rathore RK (2008) Differentiation of infective from neoplastic brain lesions by dynamic contrast-enhanced MRI. Neuroradiology 50:531–540
Pfister HW, Koedel U, Haberl RL, Dirnagl U, Feiden W, Ruckdeschel G, Einhaupl KM (1990) Microvascular changes during the early phase of experimental bacterial meningitis. J Cereb Blood Flow Metab 10:914–922
Deibler AR, Pollock JM, Kraft RA, Tan H, Burdette JH, Maldjian JA (2008) Arterial spin-labeling in routine clinical practice, part 3: hyperperfusion patterns. AJNR Am J Neuroradiol 29:1428–1435
Lee SK, Lee SY, Yun CH, Lee HY, Lee JS, Lee DS (2006) Ictal SPECT in neocortical epilepsies: clinical usefulness and factors affecting the pattern of hyperperfusion. Neuroradiology 48:678–684
Catafau AM (2001) Brain SPECT in clinical practice. Part I: perfusion. J Nucl Med Off Publ Soc Nucl Med 42:259–271
Wong EC (2014) An introduction to ASL labeling techniques. J Magn Reson Imaging 40:1–10
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wong, A.MC., Yeh, CH., Liu, HL. et al. Arterial spin-labeling perfusion imaging of childhood meningitis: a case series. Childs Nerv Syst 32, 563–567 (2016). https://doi.org/10.1007/s00381-015-2858-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-015-2858-4