Abstract
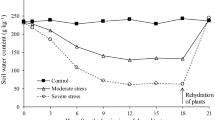
We assessed the effects of co-inoculation with Azospirillum brasilense or the application of its exudates via seeds or leaf spray on root morphological traits and nodulation, as well as on shoot development and grain yield of soybean inoculated with Bradyrhizobium. Two experiments were performed in sterile substrate under greenhouse, and two were performed at field conditions in sandy soils in a crop season with episodes of drought. The treatments in the greenhouse experiments comprised the non-inoculated control, sole inoculation with Bradyrhizobium, co-inoculation of Bradyrhizobium and A. brasilense, and inoculation of Bradyrhizobium with the application of Azospirillum exudates via seeds or leaf spray. Field treatments included non-inoculated controls without and with N-fertilizer and inoculation of Bradyrhizobium and co-inoculation with A. brasilense. Plants were assessed for root weight, total and specific lengths, volume, diameter, tissue density, branches number, root-hair length and incidence, nodule number and dry weight, shoot dry weight and N content, and grain yield. Co-inoculation of Bradyrhizobium and A. brasilense and seed application of A. brasilense exudates increased the number of root branches and nodules compared with the sole inoculation of Bradyrhizobium. However, leaf spray application of exudates was less effective. Co-inoculation also increased specific root length, root length density in soil, root-hair incidence and length, and total N content in shoot, altogether resulting in increases in grain yield. Co-inoculation of soybean with Bradyrhizobium and A. brasilense improved several root morphological traits, increasing the plant capacity to overcome moderate drought stress episodes in sandy soils, allowing to reach higher yields.




Similar content being viewed by others
References
Barbieri P, Galli E (1993) Effect on wheat root development of inoculation with an Azospirillum brasilense mutant with altered indole-3-acetic acid production. Res Microbiol 144:69–75. https://doi.org/10.1016/0923-2508(93)90216-O
Broughton WJ, Dilworth MJ (1971) Control of leghaemoglobin synthesis in snake beans. Biochem J 125:1075–1081. https://doi.org/10.1042/bj1251075
Cassán F, Vanderleyden J, Spaepen S (2014) Physiological and agronomical aspects of phytohormone production by model plant growth promoting rhizobacteria (PGPR) belonging to the genus Azospirillum. J Plant Growth Regul 33:440–459. https://doi.org/10.1007/s00344-013-9362-4
Cerezini P, Kuwano BH, Santos MB, Terassi F, Hungria M, Nogueira MA (2016) Strategies to promote early nodulation in soybean under drought. Field Crop Res 196:160–167. https://doi.org/10.1016/j.fcr.2016.06.017
Chibeba AM, Guimarães MF, Brito OR, Nogueira MA, Araujo RA, Hungria M (2015) Co-inoculation of soybean with Bradyrhizobium and Azospirillum promotes early nodulation. Am J Plant Sci 6:1641–1649. https://doi.org/10.4236/ajps.2015.610164
Comas LH, Mueller KE, Taylor LL, Midford PE, Callahan HS, Beerling DJ (2012) Evolutionary patterns and biogeochemical significance of angiosperm root traits. Int J Plant Sci 173:584–595. https://doi.org/10.1086/665823
Den Herder G, Van Isterdael G, Beeckman T, De Smet I (2010) The roots of a new green revolution. Trends Plant Sci 15:600–607. https://doi.org/10.1016/j.tplants.2010.08.009
Dobbelaere S, Croonenborghs A, Thys A, Vande Broek A, Vanderleyden J (1999) Phytostimulatory effect of Azospirillum brasilense wild type and mutant strains altered in IAA production on wheat. Plant Soil 212:153–162. https://doi.org/10.1023/A:1004658000815
Duca D, Lorv J, Patten CL, Rose D, Glick BR (2014) Indole-3-acetic acid in plant–microbe interactions. Antonie Van Leeuwenhoek 106:85–125. https://doi.org/10.1007/s10482-013-0095-y
Eissenstat DM (1991) On the relationship between specific root length and the rate of root proliferation: a field study using citrus rootstocks. New Phytol 118:63–68. https://doi.org/10.1111/j.1469-8137.1991.tb00565.x
Eissenstat DM (1992) Costs and benefits of constructing roots of small diameter. J Plant Nutr 15:763–782. https://doi.org/10.1080/01904169209364361
El-Khawas H, Adachi K (1999) Identification and quantification of auxins in culture media of Azospirillum and Klebsiella and their effect on rice roots. Biol Fertil Soils 28:377–381. https://doi.org/10.1007/s003740050507
Erktan A, McCormack ML, Roumet C (2018) Frontiers in root ecology: recent advances and future challenges. Plant Soil 424:1–9. https://doi.org/10.1007/s11104-018-3618-5
Fukami J, Abrantes JLF, del Cerro P, Nogueira MA, Ollero FJ, Megías M, Hungria M (2018a) Revealing different strategies of quorum sensing in Azospirillum brasilense strains Ab-V5 and Ab-V6. Arch Microbiol 200:47–56. https://doi.org/10.1007/s00203-017-1422-x
Fukami J, Cerezini P, Hungria M (2018b) Azospirillum: benefits that go far beyond biological nitrogen fixation. AMB Express 8:73. https://doi.org/10.1186/s13568-018-0608-1
Fukami J, Nogueira MA, Araujo RS, Hungria M (2016) Accessing inoculation methods of maize and wheat with Azospirillum brasilense. AMB Express 6:3. https://doi.org/10.1186/s13568-015-0171-y
Fukami J, Ollero FJ, Megías M, Hungria M (2017) Phytohormones and induction of plant-stress tolerance and defense genes by seed and foliar inoculation with Azospirillum brasilense cells and metabolites promote maize growth. AMB Express 7:153. https://doi.org/10.1186/s13568-017-0453-7
Giehl RFH, Gruber BD, von Wirén N (2014) It’s time to make changes: modulation of root system architecture by nutrient signals. J Exp Bot 65:769–778. https://doi.org/10.1093/jxb/ert421
Gilroy S, Jones DL (2000) Through form to function: root hair development and nutrient uptake. Trends Plant Sci 5:56–60. https://doi.org/10.1016/S1360-1385(99)01551-4
Glickmann E, Dessaux Y (1995) A critical examination of the specificity of the Salkowski reagent for indolic compounds produced by phytopathogenic bacteria. Appl Environ Microbiol 61:793–796
Hadas R, Okon Y (1987) Effect of Azospirillum brasilense inoculation on root development and respiration on tomato seedlings. Biol Fertil Soils 5:241–247. https://doi.org/10.1007/BF00256908
Haling RE, Brown LK, Bengough AG, Young IM, Hallett PD, White PJ, George TS (2013) Root hairs improve root penetration, root-soil contact, and phosphorus acquisition in soils of different strength. J Exp Bot 64:3711–3721. https://doi.org/10.1093/jxb/ert200
Haling RE, Yang Z, Shadwell N, Culvenor RA, Stefanski A, Ryan MH, Sandral GA, Kidd DR, Lambers H, Simpson RJ (2016) Root morphological traits that determine phosphorus-acquisition efficiency and critical external phosphorus requirement in pasture species. Funct Plant Biol 43:815–826. https://doi.org/10.1071/FP16037
Hodge A (2004) The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytol 162:9–24. https://doi.org/10.1111/j.1469-8137.2004.01015.x
Hungria M, Campo RJ, Souza EM, Pedrosa FO (2010) Inoculation with selected strains of Azospirillum brasilense and A. lipoferum improves yields of maize and wheat in Brazil. Plant Soil 331:413–425. https://doi.org/10.1007/s11104-009-0262-0
Hungria M, Franchini JC, Campo RJ, Crispino CC, Moraes JZ, Sibaldelli RNR, Mendes IC, Arihara J (2006) Nitrogen nutrition of soybean in Brazil: contributions of biological N2 fixation and of N fertilizer to grain yield. Can J Plant Sci 86:927–939. https://doi.org/10.4141/P05-098
Hungria M, Nogueira MA, Araujo RS (2013) Co-inoculation of soybeans and common beans with rhizobia and azospirilla: strategies to improve sustainability. Biol Fertil Soils 49:791–801. https://doi.org/10.1007/s00374-012-0771-5
Hungria M, Nogueira MA, Araujo RS (2015) Soybean seed co-inoculation with Bradyrhizobium spp. and Azospirillum brasilense: a new biotechnological tool to improve yield and sustainability. Am J Plant Sci 6:811–817. https://doi.org/10.4236/ajps.2015.66087
Hungria M, Nogueira MA, Araujo RS (2016a) Inoculation of Brachiaria spp. with the plant growth-promoting bacterium Azospirillum brasilense: an environment-friendly component in the reclamation of degraded pastures in the tropics. Agric Ecosyst Environ 221:125–131. https://doi.org/10.1016/j.agee.2016.01.024
Hungria M, O’Hara GW, Zilli JE, Araujo RS, Deaker R, Howieson JG (2016b) Isolation and growth or rhizobia. In: Howieson JG, Dilworth JG (eds) Working with rhizobia. ACIAR, Canberra, pp 39–60
Jungk A (2001) Root hairs and the acquisition of plant nutrients from soil. J Plant Nutr Soil Sci 164:121–129. https://doi.org/10.1002/1522-2624(200104)164:2<121::AID-JPLN121>3.0.CO;2-6
Klamer F, Vogel F, Li X, Bremer H, Neumann G, Neuhäuser B, Hochholdinger F, Ludewig U (2019) Estimating the importance of maize root hairs in low phosphorus conditions and under drought. Ann Bot mcz011. https://doi.org/10.1093/aob/mcz011
Lynch J (2007) Roots of the second green revolution. Aust J Bot 55:493–512. https://doi.org/10.1071/BT06118
Martins MR, Jantalia CP, Reis VM, Döwich I, Polidoro JC, Alves BJR, Boddey RM, Urquiaga S (2018) Impact of plant growth-promoting bacteria on grain yield, protein content, and urea-15N recovery by maize in a Cerrado Oxisol. Plant Soil 422:239–250. https://doi.org/10.1007/s11104-017-3193-1
Molina-Favero C, Creus CM, Simontacchi M, Puntarulo S, Lamattina L (2008) Aerobic nitric oxide production by Azospirillum brasilense Sp245 and its influence on root architecture in tomato. Mol Plant-Microbe Interact 21:1001–1009. https://doi.org/10.1094/MPMI-21-7-1001
Molla AH, Shamsuddin ZH, Halimi MS, Morziah M, Puteh AB (2001a) Potential for enhancement of root growth and nodulation of soybean co-inoculated with Azospirillum and Bradyrhizobium in laboratory systems. Soil Biol Biochem 33:457–463. https://doi.org/10.1016/S0038-0717(00)00186-3
Molla AH, Shamsuddin ZH, Saud HM (2001b) Mechanism of root growth and promotion of nodulation in vegetable soybean by Azospirillum brasilense. Commun Soil Sci Plant Anal 32:2177–2187. https://doi.org/10.1081/CSS-120000276
Moretti LG, Crusciol CAC, Kuramae EE, Bossolani JW, Moreira A, Costa NR, Alves CJ, Pascoaloto IM, Rondina ABL, Hungria M (2020) Effects of growth-promoting bacteria on soybean root activity, plant development and yield. Agron J 112:418–428. https://doi.org/10.1002/agj2.20010
Okon Y, Kapulnik Y (1986) Development and function of Azospirillum-inoculated roots. Plant Soil 90:3–16. https://doi.org/10.1007/BF02277383
Pitts RJ, Cernac A, Estelle M (1998) Auxin and ethylene promote root hair elongation in Arabidopsis. Plant J 16:553–560. https://doi.org/10.1046/j.1365-313x.1998.00321.x
Puente ML, Zawoznik M, de Sabando ML, Perez G, Gualpa JL, Carletti SM, Cassán FD (2019) Improvement of soybean grain nutritional quality under foliar inoculation with Azospirillum brasilense strain Az39. Symbiosis 77:41–47. https://doi.org/10.1007/s13199-018-0568-x
Puente ML, Gualpa JL, Lopez GA, Molina RM, Carletti SM, Cassán FD (2017) The benefits of foliar inoculation with Azospirillum brasilense in soybean are explained by an auxin signaling model. Symbiosis 76:41–49. https://doi.org/10.1007/s13199-017-0536-x
Rego CHQ, Cardoso FB, Cândido ACS, Teodoro PE, Alves CZ (2018) Co-inoculation with Bradyrhizobium and Azospirillum increases yield and quality of soybean seeds. Agron J 110:1–8. https://doi.org/10.2134/agronj2018.04.0278
Ribaudo CM, Krumpholz EM, Cassán FD, Bottini R, Cantore ML, Cura JA (2006) Azospirillum sp. promotes root hair development in tomato plants through a mechanism that involves ethylene. J Plant Growth Regul 25:175–185. https://doi.org/10.1007/s00344-005-0128-5
Ritchie SW, Hanway JJ, Thompson HE, Benson GO (1994) How a soybean plant develops. Special Report 53. Iowa State University of Science and Technology: Cooperative Extension Service, Ames
Rondina ABL, Tonon BC, Lescano LEAM, Hungria M, Nogueira MA, Zangaro W (2019) Plants of distinct successional stages have different strategies for nutrient acquisition in an Atlantic Rain Forest ecosystem. Int J Plant Sci 180:186–199. https://doi.org/10.1086/701353
Silva ER, Zoz J, Oliveira CES, Zuffo AM, Steiner F, Zoz T, Vendruscolo EP (2019) Can co-inoculation of Bradyrhizobium and Azospirillum alleviate adverse effects of drought stress on soybean (Glycine max L. Merrill)? Arch Microbiol 201:325–335. https://doi.org/10.1007/s00203-018-01617-5
Star L, Matan O, Dardanelli M, Kapulnik Y, Burdman S, Okon Y (2012) The Vicia sativa spp. nigra – Rhizobium leguminosarum bv. viciae symbiotic interaction is improved by Azospirillum brasilense. Plant Soil 356:165–174. https://doi.org/10.1007/s11104-010-0713-7
Tennant D (1975) A test of modified line intersect method of estimating root length. J Ecol 63:995–1001. https://doi.org/10.2307/2258617
Tien TM, Gaskins MH, Hubbel DH (1979) Plant growth substances produced by Azospirillum brasilense and their effect on the growth of pearl millet (Pennisetum americanum L.). Appl Environ Microbiol 37:1016–1024
Timmers ACJ (2000) Infection of root hairs by rhizobia: infection thread development with emphasis on the microtubular cytoskeleton. In: Ridge RW, Emons AMC (eds) Root hairs. Springer, Tokyo, pp 223–239
Vacheron J, Desbrosses G, Bouffaud ML, Touraine B, Moënne-Loccoz Y, Muller D, Legendre L, Wisniewski-Dyé F, Prigent-Combaret C (2013) Plant grow-promoting rhizobacteria and root system functioning. Front Plant Sci 4:356. https://doi.org/10.3389/fpls.2013.00356
Wheatley RM, Poole PS (2018) Mechanisms of bacterial attachment to roots. FEMS Microbiol Rev 42:448–461. https://doi.org/10.1093/femsre/fuy014
Willis RB, Montgomery ME, Allen PR (1996) Improved method for manual, colorimetric determination of total Kjeldahl nitrogen using salicylate. J Agric Food Chem 44:1804–1807. https://doi.org/10.1021/jf950522b
Yates RJ, Howieson JG, Hungria M, Bala A, O’Hara GW, Terpolilli J (2016) Authentication of rhizobia and assessment of the legume symbiosis in controlled plant growth systems. In: Howieson JG, Dilworth MJ (eds) Working with rhizobia. ACIAR, Canberra, pp 73–108
Zangaro W, Alves RA, Souza PB, Rostirola LV, Lescano LEAM, Rondina ABL, Nogueira MA (2014) Succession and environmental variation influence soil exploration potential by fine roots and mycorrhizal fungi in an Atlantic ecosystem in southern Brazil. J Trop Ecol 30:237–248. https://doi.org/10.1017/S0266467414000078
Zangaro W, Nishidate FR, Camargo FRS, Romagnoli GG, Vandresen J (2005) Relationships among arbuscular mycorrhizas, root morphology and seedling growth of tropical native woody species in southern Brazil. J Trop Ecol 21:529–540. https://doi.org/10.1017/S0266467405002555
Zangaro W, Rostirola LV, Souza PB, Alves RA, Lescano LEAM, Rondina ABL, Nogueira MA, Carrenho R (2013) Root colonization and spore abundance of arbuscular mycorrhizal fungi in distinct successional stages from an Atlantic rainforest biome in southern Brazil. Mycorrhiza 23:221–233. https://doi.org/10.1007/s00572-012-0464-9
Acknowledgments
A. B. L. Rondina acknowledges a post-doc fellowship from CNPq. A. W. S. Sanzovo and G. S. Guimarães are CAPES scholars. M. A. Nogueira and M. Hungria are CNPq research fellows. This manuscript was reviewed by internal researchers of the Embrapa Soybean Center, Dr. Renan A. Ribeiro and Dr. Jakeline R. M. Delamuta, prior to submission.
Funding
This study was partially funded by Embrapa (02.13.08.003.00.00) and INCT – Plant Growth-Promoting Microorganisms for Agricultural Sustainability and Environmental Responsibility (CNPq-465133/2014-2, Fundação Araucária-STI, CAPES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rondina, A.B.L., dos Santos Sanzovo, A.W., Guimarães, G.S. et al. Changes in root morphological traits in soybean co-inoculated with Bradyrhizobium spp. and Azospirillum brasilense or treated with A. brasilense exudates. Biol Fertil Soils 56, 537–549 (2020). https://doi.org/10.1007/s00374-020-01453-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-020-01453-0