Abstract
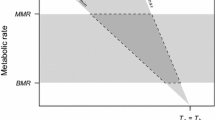
For decades, there was an intense debate in relation to the mechanism behind the entry into metabolic depression (EMD) of mammals and birds. The fulcrum of the argument was whether the depression of metabolic rate (\(\dot{W}\)) was caused by the drop in body temperature, the so-called “Q10 effect”, or whether it was caused by a metabolic downregulation. One present-day model of this process is a qualitative (textual) description: the initial step of EDM would be a downregulation in \(\dot{W}\) from the value maintaining euthermia at a given ambient temperature to the basal metabolic rate of the animal and, then, Q10 effect would take over and drop \(\dot{W}\) to its lower levels. Despite widely accepted, this qualitative description still misses a theoretical analysis. Here, we transpose the descriptive model to a formal quantitative one and analyze it under necessary thermodynamic conditions of a system. We, then, compare the results of the formal model to empirical data of EMD by mammals and birds. The comparisons indicate that the metabolic evolution in the course of the entry phase does not follow the descriptive model. Instead, as proposed by alternate models, EMD is a downregulated process as a whole until a new equilibrium Tb is attained.








Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
Not applicable.
Abbreviations
- EMD:
-
Entry into metabolic depression
- T A :
-
Ambient temperature
- W :
-
Total metabolic demand
- \({\dot{W}}_{\mathrm{E}}\) :
-
Euthermic metabolic rate
- \({\dot{W}}_{\mathrm{B}}\) :
-
Basal metabolic rate
- \({\dot{W}}_{\mathrm{D}}\) :
-
Depressed metabolic rate
- T b :
-
Body temperature
- T E :
-
Euthermic body temperature
- T D :
-
Depressed body temperature
- T x :
-
Body temperature due to basal metabolic rate at a given TA
- T y :
-
Body temperature at basal metabolic rate during EMD
References
Bartels W, Law BS, Geiser F (1998) Daily torpor and energetics in a tropical mammal, the northern blossom-bat Macroglossus minimus (Megachiroptera). J Comp Physiol B Biochem Syst Environ Physiol 168:233–239. https://doi.org/10.1007/s003600050141
Bozinovic F, Rosenmann M (1988) Daily torpor in Calomys musculinus, a South American rodent. J Mammal 69:150–152. https://doi.org/10.2307/1381762
Bozinovic F, Ruiz G, Rosenmann M (2004) Energetics and torpor of a South American living fossil, the microbiotheriid Dromiciops gliroides. J Comp Physiol B Biochem Syst Environ Physiol 174:293–297. https://doi.org/10.1007/s00360-004-0414-8
Brown JCL, Gerson AR, Staples JF (2007) Mitochondrial metabolism during daily torpor in the dwarf Siberian hamster: role of active regulated changes and passive thermal effects. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.00310.2007
Callen HB (1985) Thermodynamics and an introduction to thermostatistics, 2nd edn. Wiley, New York
Chaui-Berlinck JG, Alves Monteiro LH, Navas CA, Bicudo JEPW (2002) Temperature effects on energy metabolism: a dynamic system analysis. Proc R Soc Lond Ser B Biol Sci 269:15–19. https://doi.org/10.1098/rspb.2001.1845
Cheng CL (2008) Is human hibernation possible? Annu Rev Med 59:177–186. https://doi.org/10.1146/annurev.med.59.061506.110403
de Groot SR, Mazur P (1984) Non-equilibrium thermodynamics, Dover unab. ed. Dover Publications, Mineola
Geiser F (2004) Metabolic rate and body temperature reduction during hibernation and daily torpor. Annu Rev Physiol 66:239–274. https://doi.org/10.1146/annurev.physiol.66.032102.115105
Geiser F, Song X, Körtner G (1996) The effect of He-O2 exposure on metabolic rate, thermoregulation and thermal conductance during normothermia and daily torpor. J Comp Physiol B Biochem Syst Environ Physiol 166:190–196. https://doi.org/10.1007/BF00263982
Geiser F, Kortner G, Schmidt I (1998) Leptin increases energy expenditure of a marsupial by inhibition of daily torpor. Am J Physiol Integr Comp Physiol 275:1627–1632
Geiser F, Currie SE, O’Shea KA, Hiebert SM (2014) Torpor and hypothermia: reversed hysteresis of metabolic rate and body temperature. Am J Physiol Integr Comp Physiol 307:R1324–R1329. https://doi.org/10.1152/ajpregu.00214.2014
Glansdorff P, Prigogine I (1971) Structure, Stabilité et Fluctuations. Paris, Masson & Cie Éditeurs
Hainsworth FR, Wolf LL (1970) Regulation of oxygen consumption and body temperature during torpor in a hummingbird, Eulampis jugularis. Science (80-) 168:368–369. https://doi.org/10.1126/science.168.3929.368
Heldmaier G, Ruf T (1992) Body temperature and metabolic rate during natural hypothermia in endotherms. J Comp Physiol B 162:696–706. https://doi.org/10.1007/BF00301619
Hudson JW, Scott IM (1979) Daily torpor in the laboratory mouse, Mus musculus var. Albino. Physiol Zool 52:205–218. https://doi.org/10.1086/physzool.52.2.30152564
Karpovich SA, Tøien Ø, Buck CL, Barnes BM (2009) Energetics of arousal episodes in hibernating arctic ground squirrels. J Comp Physiol B Biochem Syst Environ Physiol 179:691–700. https://doi.org/10.1007/s00360-009-0350-8
Lovegrove BG, Raman J, Perrin MR (2001) Heterothermy in elephant shrews, Elephantulus spp. (Macroscelidea): daily torpor or hibernation? J Comp Physiol B 171:1–10
Ortmann S, Heldmaier G (2000) Regulation of body temperature and energy requirements of hibernating Alpine marmots (Marmota marmota). Am J Physiol Integr Comp Physiol 278:R698–R704. https://doi.org/10.1152/ajpregu.2000.278.3.R698
Plank M (1926) Treatise in thermodynamics, 7th edn. Dover Publications, New York
Ruf T, Gasch K, Stalder G, Gerritsmann H, Giroud S (2021) An hourglass mechanism controls torpor bout length in hibernating garden dormice. J Exp Biol. https://doi.org/10.1242/jeb.243456
Snapp BD, Heller HC (1981) Suppression of metabolism during hibernation in ground squirrels (Citellus lateralis). Physiol Zool 54:297–307. https://doi.org/10.1086/physzool.54.3.30159944
Snyder GK, Nestler JR (1990) Relationships between body temperature, thermal conductance, Q10 and energy metabolism during daily torpor and hibernation in rodents. J Comp Physiol B 159:667–675. https://doi.org/10.1007/BF00691712
Westman W, Geiser F (2004) The effect of metabolic fuel availability on thermoregulation and torpor in a marsupial hibernator. J Comp Physiol B Biochem Syst Environ Physiol 174:49–57. https://doi.org/10.1007/s00360-003-0388-y
Willis CKR, Turbill C, Geiser F (2005) Torpor and thermal energetics in a tiny Australian vespertilionid, the little forest bat (Vespadelus vulturnus). J Comp Physiol B 175:479–486. https://doi.org/10.1007/s00360-005-0008-0
Wilz M, Heldmaier G (2000) Comparison of hibernation, estivation and daily torpor in the edible dormouse, Glis glis. J Comp Physiol B Biochem Syst Environ Physiol 170:511–521. https://doi.org/10.1007/s003600000129
Withers PC (1977) Respiration, metabolism, and heat exchange of euthermic and torpid poorwills and hummingbirds. Physiol Zool 50:43–52
Funding
This study had no funding.
Author information
Authors and Affiliations
Contributions
PGNS: conceptual ideas, modeling, and writing; JGCB: conceptual ideas, modeling, and writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by G. Heldmaier.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1
Thermodynamical compatibility of Ty
The pair {\({\dot{W}}_{\mathrm{B}}\), Tx} is inherently compatible from a thermodynamic standpoint. This will become clear shortly. Because of this, the pair {\({\dot{W}}_{\mathrm{B}}\), Ty} cannot represent another solution (equilibrium point) of the system unless the thermal conductance had changed, and such a change would preclude Q10 computations correctly (see “Relaxing assumptions #1 and #6: another intermediate body temperature landmark—Ty”). However, we can relax the condition of equilibrium and consider that the system undergoes a series of quasi-static transitions between its states, without changes in thermal conductance. This means that despite ever-changing, the temperature of the system is a reliable measurement.
Under these assumptions, using Eq. (3c), we rewrite Eq. (2) as
Therefore, when \({\dot{W}=\dot{W}}_{\mathrm{B}}\) and Tb = Ty, using Eq. (4b) we obtain
Equation (11) can then be used to compute the expected rate of change in body temperature when the pair {\({\dot{W}}_{\mathrm{B}}\), Ty} occurs. Such an expected ratio is thermodynamically correct due to the quasi-static assumption presented above. Because of this and the constancy of the thermal conductance also assumed, Q10 computations would make sense even with an ever-changing body temperature.
Therefore, the ratio between the expected \(\frac{\mathrm{d}{T}_{\mathrm{b}}}{\mathrm{d}t}{|}_{{T}_{y}}\) (Eq. (A1)) and the one observed experimentally gives an index of the thermodynamic adequacy to compute Q10 values. In other words, this is a direct comparison between the model and the actual behavior of the animal when it crosses the Ty temperature. Values close to 1 indicate such adequacy. On the other hand, values not close to 1 indicate that the system is not following a dynamic compatible with the assumption of the pair {\({\dot{W}}_{\mathrm{B}}\), Ty}.
If we use Ty = Tx in Eq. (A1), it can be seen that \(\frac{\mathrm{d}{T}_{\mathrm{b}}}{\mathrm{d}t}{|}_{{T}_{x}}=0\) and the system is in internal equilibrium. This gives the correctness of using the pair {\({\dot{W}}_{\mathrm{B}}\), Tx} which would be exactly the end of the first phase of EMD.
The mixed model for analytical solution
Consider that the energy input to the system is negligible in face of energy loss as heat, leading to a cooling process. For a Newtonian cooling, Eq. (2) becomes
Considering Eq. (12), Eq. (7) becomes
Strictly speaking, in a Newtonian cooling process, there is no energy input to the system and then, obviously, the total metabolic demand during the cooling would be zero (a useless result for the analysis). To make a comparison between the numerical solution of Eq. (9) and an analytical one, we mixed Eq. (14) with metabolic rate dictated by temperature effects (as in Eq. (9)) but without functioning as an energy input to the system. The cooling process will lead to \({T}_{\mathrm{b}}={T}_{\mathrm{A}}\), and we write the variable \(\tau \) as
Then, from Eqs. (12) and (13), we can re-express Eq. (9) as the so-called exponential integral:
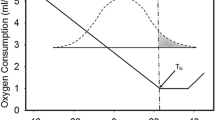
where the superscript “N” is to indicate that this total metabolic demand was computed as if “a Newtonian cooling process”. If we plot the analytical solution of Eq. (15) and the numerical integration of Eq. (9), we can see that the two equations have very similar behaviors. Figure 2 illustrates this.
Appendix B
The plots in Fig. 8 suggest that thermal conductance X changes during EMD, and that such changes are a linear function of metabolic rate \(\dot{W}\) within a range of the process. Therefore, let us assume this linear relationship:
where X0 is a constant of minimal thermal conductance and K is the rate of change in X due to \(\dot{W}\). We shall show that Eq. (16) leads to contradictions, implying that to assume thermal conductance computations during EMD as true values is incorrect.
For the sake of notation, we use ΔT instead of (Tb − TA). The subscript “E”, as stated before, refers to euthermic conditions. From Eqs. (2) and (16), one obtains
Thus, if \(\dot{W}\) = 0, ΔT = 0 (as expected), and, as \(\dot{W}\) increases:
Consequently, given the limiting of euthermic conditions:
In addition,
The euthermic steady-state condition demands
which poses a problem in Eq. (16), since, then, \({X}_{0}=0\). One could envisage two possible ways to circumvent such a problem, although none of them can be true solutions as it will be demonstrated.
\({X}_{0}=0\)
Assuming \({X}_{0}=0\), Eq. (16) becomes
Thus, the quasi-static condition of \(\frac{\mathrm{d}{T}_{\mathrm{b}}}{\mathrm{d}t}=0\) during EMD now implies
or:
However, since ΔT is changing during EMD, Eq. (25) implies that K is not a constant as previously assumed (Eq. (16) or (23)). In other words, the linear relationship between X and \(\dot{W}\) observed in Fig. 8 evidences the absence of quasi-static conditions of the system, as the R values in Table 2 also show, or, given a non-constant K, a closed-control loop of body temperature regulation.
\({X}_{\mathrm{total}}={X}_{0}+K\cdot \dot{W}\)
This assumption is quite similar to Eq. (16), but we shall now write the euthermic thermal conductance as
Considering that XE is usually computed as the slope of the (linear) relationship of metabolic rate and TA below the critical lower temperature of the thermoneutral zone (see Fig. 1), Eq. (16) must be a constant irrespectively to TA below such a critical temperature. However, euthermic metabolic rate \({\dot{W}}_{\mathrm{E}}\) increases as TA falls, and, thus, as in the previous section, K cannot be a constant as supposed (Eq. (16) or (23)).
Rights and permissions
About this article
Cite this article
Nogueira de Sá, P.G., Chaui-Berlinck, J.G. A thermodynamic-based approach to model the entry into metabolic depression by mammals and birds. J Comp Physiol B 192, 593–610 (2022). https://doi.org/10.1007/s00360-022-01442-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-022-01442-9