Abstract
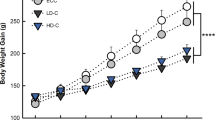
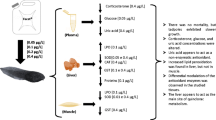
Birds control body homeostasis through the secretion of corticosterone. This hormone is the end-product of the hypothalamic–pituitary–adrenal (HPA) axis response to stressors. High levels of corticosterone may be associated with low individual fitness and may affect balance between pro-oxidants and antioxidants. Given these points, chronic stress modulated by hormones could undermine individual fitness by increasing oxidative tissue damage. In this study, we administered corticosteroids by diet (20 mg/kg of diet) to captive adult kestrels (Falco tinnunculus) over a 14-day period to evaluate the effects of a simulated chronic stress modulated by corticosteroids. We found that dietary administration of corticosterone caused a 32% increase of reactive oxygen metabolites, but did not impair total serum antioxidant capacity, serum carotenoids or body mass. Oxidative stress had a 64% increase in treated birds compared to 30% in controls. The two groups did not differ in the total serum antioxidant capacity, which showed a significant decrease over the study period. In contrast, circulating carotenoids and body mass increased in both groups. These results suggest that stress hormones, such as corticosterone, may also act as modulators of oxidative stress in birds.

Similar content being viewed by others
References
Beckman KB, Ames BN (1998) The free radical theory of aging matures. Physiol Rev 78:547–581
Behl C, Lezoualc’h F, Trapp T, Widmann M, Skutella T, Holsboer F (1997) Glucocorticoids enhance oxidative stress-induced cell death in hippocampal neurons in vitro. Endocrinology 138:101–106
Blount JD, Metcalfe NB, Birkhead TR, Surai PF (2003) Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 300:125–127
Blount JD (2004) Carotenoids and life-history evolution in animals. Arch Biochem Biophys 430:10–15
Caro P, Gómez J, Sanz A, Portero-Otín M, Pamplona R, Barja G (2007) Effect of graded corticosterone treatment on aging-related markers of oxidative stress in rat liver mitochondria. Biogerontology 8:1–11
Casagrande S, Csermely D, Pini E, Bertacche V, Tagliavini J (2006) Skin carotenoid concentration correlates with male hunting skill and territory quality in the kestrel (Falco tinnunculus). J Avian Biol 37:190–196
Charmandari E, Tsigos C, Chrousos G (2005) Endocrinology of the stress response. Annu Rev Physiol 67:259–284
Costantini D, Casagrande S, De Filippis S, Brambilla G, Fanfani A, Tagliavini J, Dell’Omo G (2006) Correlates of oxidative stress in wild kestrel nestlings (Falco tinnunculus). J Comp Physiol B 176:329–337
Costantini D, Dell’Omo G (2006a) Effects of T-cell-mediated immune response on avian oxidative stress. Comp Biochem Physiol Part A 145:137–142
Costantini D, Dell’Omo G (2006b) Environmental and genetic components of oxidative stress in wild kestrel nestlings (Falco tinnunculus). J Comp Physiol B 176:575–579
Costantini D, Fanfani A, Dell’Omo G (2007a) Carotenoid availability does not limit the capability to cope with oxidative stress in nestling kestrels (Falco tinnunculus). J Exp Biol 210:1238–1244
Costantini D, Coluzza C, Fanfani A, Dell’Omo G (2007b) Effects of carotenoid supplementation on colour expression, oxidative stress and body mass in rehabilitated captive adult kestrels (Falco tinnunculus). J Comp Physiol B 177:723–731
Costantini D, Møller AP (2008) Carotenoids are minor antioxidants for birds. Funct Ecol 22:367–370
Dietz MW, Daan S, Masman D (1992) Energy requirements for molt in the kestrel Falco tinnunculus. Physiol Zool 65:1217–1235
Dröge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
Eraud C, Devevey G, Gaillard M, Prost J, Sorci G, Faivre B (2007) Environmental stress affects the expression of a carotenoid-based sexual trait in male zebra finches. J Exp Biol 210:3571–3578
Faivre B, Grégoire A, Préault M, Cézilly F, Sorci G (2003) Immune activation rapidly mirrored in a secondary sexual trait. Science 300:103
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408:239–247
Gray JM, Yarian D, Ramenofsky M (1990) Corticosterone, foraging behaviour, and metabolism in dark-eyed juncos, Junco hyemalis. Gen Comp Endocrinol 79:375–384
Halliwell BH, Gutteridge JMC (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford
Hõrak P, Zilmer M, Saks L, Ots I, Karu U, Zilmer K (2006) Antioxidant protection, carotenoids, and the costs of immune challenge in greenfinches. J Exp Biol 209:4329–4338
Krinsky NI, Yeum K-J (2003) Carotenoid-radical interactions. Biochem Biophys Res Comm 305:754–760
Lawrence MS, Sapolsky RM (1994) Glucocorticoids accelerate ATP loss following metabolic insults in cultured hippocampal neurons. Brain Res 646:303–306
Lin H, Decuypere E, Buyse J (2004a) Oxidative stress induced by corticosterone administration in broiler chickens (Gallus gallus domesticus) 1. Chronic exposure. Comp Biochem Physiol Part B 139:737–744
Lin H, Decuypere E, Buyse J (2004b) Oxidative stress induced by corticosterone administration in broiler chickens (Gallus gallus domesticus) 2. Short-term effect. Comp Biochem Physiol Part B 139:745–751
Love OP, Bird DM, Shutt LJ (2003a) Corticosterone levels during post-natal development in captive American kestrels (Falco sparverius). Gen Comp Endocr 130:135–141
Love OP, Bird DM, Shutt LJ (2003b) Plasma corticosterone in American kestrel siblings: effect of age, hatching order, and hatching asynchrony. Horm Behav 43:480–488
Love OP, Shutt LJ, Silfies JS, Bird DM (2003c) Repeated restraint and sampling results in reduced corticosterone levels in developing and adult captive American kestrels. Physiol Biochem Zool 76:753–761
Malheiros RD, Moraes VMB, Collin A, Decuypere E, Buyse J (2003) Free diet selection by broilers as influenced by dietary macronutrient ratio and corticosterone supplementation. 1. Diet selection, organ weights, and plasma metabolites. Poult Sci 82:123–131
Masman D (1986) The annual cycle of the kestrel, Falco tinnunculus: a study in behavioural energetics. Ph.D. thesis, University of Groningen.
Maurice D, Lightsey SF, Toler JE, Canty S (2007) Effect of chronic oxidative/corticosterone-induced stress on ascorbic acid metabolism and total antioxidant capacity in chickens (Gallus gallus domesticus). J Anim Physiol Anim Nutr 91:355–360
McGraw KJ, Ardia DR (2003) Carotenoids, immunocompetence, and the information content of sexual colors: an experimental test. Am Nat 162:704–712
McGraw KJ, Adkins-Regan E, Parker RS (2005) Maternally derived carotenoid pigments affect offspring survival, sex ratio, and sexual attractiveness in a colorful songbird. Naturwissenschaften 92:375–380
McIntosh LJ, Cortopassi KM, Sapolsky RM (1998a) Glucocorticoids may alter antioxidant enzyme capacity in the brain: kainic acid studies. Brain Res 791:215–222
McIntosh LJ, Hong KE, Sapolsky RM (1998b) Glucocorticoids may alter antioxidant enzyme capacity in the brain: baseline studies. Brain Res 791:209–214
Møller AP, Biard C, Blount JD, Houston DC, Ninni P, Saino N, Surai PF (2000) Carotenoid-dependent signals: indicators of foraging efficiency, immunocompetence or detoxification ability? Avian Poult Biol Rev 11:137–159
Nagae M, Fuda H, Ura K, Kawamura H, Adachi S, Hara A, Yamauchi K (1994) The effect of cortisol administration on blood plasma immunoglobulin M (IgM) concentrations in masu salmon (Oncorhynchus masou). Fish Physiol Biochem 13:41–48
Olson VA, Owens IPF (1998) Costly sexual signals: are carotenoids rare, risky or required? Trends Ecol Evol 13:510–514
Orzechowski O, Ostaszewski P, Wilczak J, Jank M, Balasinska B, Wareski P, Fuller J Jr (2002) Rats with a glucocorticoid-induced catabolic state show symptoms of oxidative stress and spleen atrophy: the effects of age and recovery. J Vet Med A 49:256–263
Pride RE (2005) High fecal glucocorticoid levels predict mortality in ring-tailed lemurs (Lemur catta). Biol Lett 1:60–63
Romero LM (2004) Physiological stress in ecology: lessons from biomedical research. Trends Ecol Evol 19:249–255
Romero LM, Wikelski M (2001) Corticosterone levels predict survival probabilities of Galapagos marine iguanas during El Nino events. Proc Nat Acad Sci USA 98:7366–7370
Roussel D, Dumas JF, Simard G, Malthiery Y, Ritz P (2004) Kinetics and control of oxidative phosphorylation in rat liver mitochondria after dexamethasone treatment. Biochem J 382:491–499
Saino N, Ferrari R, Romano M, Martinelli R, Møller AP (2003) Experimental manipulation of egg quality affects immunity of barn swallow nestlings. Proc R Soc Lond B 270:2485–2489
Sapolsky RM, Romero LM, Munck AU (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocrinol Rev 21:55–89
Sies H (1991) Oxidative stress II. Oxidants and antioxidants. Academic Press, London
Silverin B (1986) Corticosterone-binding proteins and behavioural effects of high plasma levels of corticosterone during the breeding period. Gen Comp Endocrinol 64:67–74
Sockman KW, Schwabl H (2001) Plasma corticosterone in nestling American kestrels: effects of age, handling stress, yolk androgens, and body condition. Gen Comp Endocrinol 122:205–212
Surai P (2002) Natural antioxidants in avian nutrition and reproduction. Nottingham University Press, Nottingham
Tummeleht L, Mägi M, Kilgas P, Mänd R, Hõrak P (2006) Antioxidant protection and plasma carotenoids of incubating great tits (Parus major L.) in relation to health state and breeding conditions. Comp Biochem Physiol Part C 144:166–172
Village A (1990) The kestrel. T & AD Poyser, London
Wingfield JC, Silverin B (1986) Effects of corticosterone on territorial behaviour of free-living male song sparrows, Melospiza melodia. Horm Behav 20:405–417
Wingfield JC, Maney DL, Breuner CW, Jacobs JD, Lynn S, Ramenofsky M, Richardson RD (1998) Ecological bases of hormone-behavior interactions: the “emergency life history stage”. Am Zool 38:191–206
Yu PB (1994) Cellular defenses against damage from reactive oxygen species. Physiol Rev 74:139–162
Zhu Z, Jiang W, Thompson HJ (1998) Effect of corticosterone administration on mammary gland development and p27 expression and their relationship to the effects of energy restriction on mammary carcinogenesis. Carcinogenesis 19:2101–2106
Acknowledgments
We are grateful to the rehabilitation centres of the Lega Italiana Protezione Uccelli (LIPU) sections of Latina and Rome, the Circeo National Park, the Forestry Service of Pescara, Bosco San Silvestro (Caserta), the Vico Natural Reserve (Caprarola, VT) and WWF Astroni for providing the birds used in this study. We thank Nadia Macciocchi, Fernando and Serena Costantini for help with the animal care; the scientific association Ornis italica for supporting part of this research; the three anonymous reviewers for valuable comments on the manuscript; Martina Muller for revising the English. David Costantini is grateful to Gianfranco Brambilla and Edoardo Vignolo for technical and logistical support at the Istituto Superiore di Sanità, Rome. David Costantini was supported by a PhD fellowship issued from the University La Sapienza.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Costantini, D., Fanfani, A. & Dell’Omo, G. Effects of corticosteroids on oxidative damage and circulating carotenoids in captive adult kestrels (Falco tinnunculus). J Comp Physiol B 178, 829–835 (2008). https://doi.org/10.1007/s00360-008-0270-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-008-0270-z