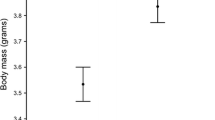
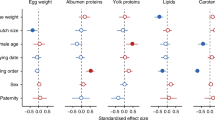
Abstract
In egg-laying animals, mothers can influence the development of their offspring via the suite of biochemicals they incorporate into the nourishing yolk (e.g. lipids, hormones). However, the long-lasting fitness consequences of this early nutritional environment have often proved elusive. Here, we show that the colorful carotenoid pigments that female zebra finches (Taeniopygia guttata) deposit into egg yolks influence embryonic and nestling survival, the sex ratio of fledged offspring, and the eventual ornamental coloration displayed by their offspring as adults. Mothers experimentally supplemented with dietary carotenoids prior to egg-laying incorporated more carotenoids into eggs, which, due to the antioxidant activity of carotenoids, rendered their embryos less susceptible to free-radical attack during development. These eggs were subsequently more likely to hatch, fledge offspring, produce more sons than daughters, and produce sons who exhibited more brightly colored carotenoid-based beak pigmentation. Provisioned mothers also acquired more colorful beaks, which directly predicted levels of carotenoids found in eggs, thus indicating that these pigments may function not only as physiological ‘damage-protectants’ in adults and offspring but also as morphological signals of maternal reproductive capabilities.


Similar content being viewed by others
References
Amundsen T (2000) Why are female birds ornamented? Trends Ecol Evol 15:149–155
Blount JD, Houston DC, Møller AP (2000) Why egg yolk is yellow. Trends Ecol Evol 15:47–49
Blount JD, Houston DC, Surai PF, Møller AP (2004) Egg-laying capacity is limited by carotenoid pigment availability in wild gulls Larus fuscus. Proc R Soc Lond B (Suppl.) 271:S79–S81
Blount JD, Metcalfe NB, Arnold KE, Surai PF, Devevey GL, Monaghan P (2003b) Neonatal nutrition, adult antioxidant defences and sexual attractiveness in the zebra finch. Proc R Soc Lond B 270:1691–1696
Blount JD, Metcalfe NB, Birkhead TR, Surai PF (2003a) Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 300:125–127
Blount JD, Surai PF, Nager RG, Houston DC, Møller AP, Trewby ML, Kennedy MW (2002) Carotenoids and egg quality in the lesser black-backed gull Larus fuscus: a supplemental feeding study of maternal effects. Proc R Soc Lond B 269:29–36
Bortolotti GR, Negro JJ, Surai PF, Prieto P (2003) Carotenoids in eggs and plasma of red-legged partridges: effects of diet and reproductive output. Physiol Biochem Zool 76:367–374
Bradbury RB, Blakey JK (1998) Diet, maternal condition, and offspring sex ratio in the zebra finch, Poephila guttata. Proc R Soc Lond B 265:895–899
Brief S, Chew BP (1985) Effects of vitamin-A and beta-carotene on reproductive performance in gilts. J Anim Sci 60:998–1004
Burley N, Coopersmith C (1987) Bill color preferences of zebra finches. Ethology 76:133–151
Chew BP, Archer RG (1983) Comparative role of vitamin-A and beta-carotene on reproduction and neonate survival in rats. Theriogenology 20:459–472
Clutton-Brock TH (1991) The evolution of parental care. Princeton University Press, Princeton, NJ
Collins SA, ten Cate C (1996) Does beak color affect female preference in zebra finches? Anim Behav 52:105–112
George SB, Lawrence JM, Lawrence AL, Smiley J, Plank L (2001) Carotenoids in the adult diet enhance egg and juvenile production in the sea urchin Lytechinus variegatus. Aquaculture 199:353–369
Hill GE (1998) An easy, inexpensive method to quantify plumage coloration. J Field Ornithol 69:353–363
Houtman AM (1992) Female zebra finches choose extra-pair copulations with genetically attractive males. Proc R Soc Lond B 249:3–6
Khachik F, Spangler CJ, Smith JC, Canfield LM, Steck A, Pfander H (1997) Identification, quantification, and relative concentrations of carotenoids and their metabolites in human milk and serum. Analyt Chem 69:1873–1881
Kilner R (1998) Primary and secondary sex ratio manipulation by zebra finches. Anim Behav 56:155–164
Koutsos EA, Clifford AJ, Calvert CC, Klasing KC (2003) Maternal carotenoid status modifies incorporation of dietary carotenoids into immune tissues of growing chickens (Gallus gallus domesticus). J Nutr 133:1132–1138
Krinsky NI (2001) Carotenoids as antioxidants. Nutrition 17:815–817
McGraw KJ, Ardia DR (2003) Carotenoids, immunocompetence, and the information content of sexual colors: an experimental test. Am Nat 162:704–712
McGraw KJ, Adkins-Regan E, Parker RS (2002) Anhydrolutein in the zebra finch: a new, metabolically derived carotenoid in birds. Comp Biochem Physiol B 132:813–820
McGraw KJ, Hill GE, Navara KJ, Parker RS (2004) Differential accumulation and pigmenting ability of dietary carotenoids in colorful finches. Physiol Biochem Zool 77:484–491
McGraw KJ, Hill GE, Stradi R, Parker RS (2001) The influence of carotenoid acquisition and utilization on the maintenance of species-typical plumage pigmentation in male American goldfinches (Carduelis tristis) and northern cardinals (Cardinalis cardinalis). Physiol Biochem Zool 74:843–852
Møller AP, Biard C, Blount JD, Houston DC, Ninni P, Saino N, Surai PF (2000) Carotenoid-dependent signals: indicators of foraging efficiency, immunocompetence, or detoxification ability? Avian Poult Biol Rev 11:137–159
Monaghan P, Metcalfe NB, Houston DC (1996) Male finches selectively pair with fecund females. Proc R Soc Lond B 263:1183–1186
Mousseau TA, Fox CW (1998) Maternal effects as adaptations. Oxford University Press, Oxford
Nowicki S, Peters S, Podos J (1998) Song learning, early nutrition, and sexual selection in songbirds. Am Zool 38:179–190
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analyt Biochem 139:292–298
Ohlsson T, Smith HG, Raberg L, Hasselquist D (2002) Pheasant sexual ornaments reflect nutritional conditions during early growth. Proc R Soc Lond B 269:21–27
Olson VA, Owens IPF (1998) Costly sexual signals: are carotenoids rare, risky or required? Trends Ecol Evol 13:510–514
Price DK, Burley NT (1993) Constraints on the evolution of attractive traits: genetic variance of zebra finch bill color. Heredity 71:405–412
Price DK (1996) Sexual selection, selection load and quantitative genetics of zebra finch bill color. Proc R Soc Lond B 263:217–221
Rutkowska J, Cichón M (2002) Maternal investment during egg laying and offspring sex: an experimental study of zebra finches. Anim Behav 64:817–822
Saino N, Bertacche V, Ferrari RP, Martinelli R, Møller AP, Stradi R (2002) Carotenoid concentration in barn swallow eggs is influenced by laying order, maternal infection, and paternal ornamentation. Proc R Soc Lond B 269:1729–1733
Saino N, Ferrari R, Romano M, Martinelli R, Møller AP (2003) Experimental manipulation of egg carotenoids affects immunity of barn swallow nestlings. Proc R Soc Lond B 270:2485–2489
Shahidi F, Metusalach, Brown JA (1998) Carotenoid pigments in seafoods and aquaculture. Crit Rev Food Sci Nutr 38:1–67
Sharoni Y, Danilenko M, Walfisch S, Amir H, Nahum A, Ben-Dor A, Hirsch K, Khanin M, Steiner M, Agemy L, Zango G, Levy J (2002) Role of gene regulation in the anticancer activity of carotenoids. Pure Appl Chem 74:1469–1477
Surai PF, Noble RC, Speake BK (1996) Tissue-specific differences in antioxidant distribution and susceptibility to lipid peroxidation during development of the chick embryo. Biochim Biophys Acta 1304:1–10
Torrissen OJ, Christiansen R (1995) Requirements for carotenoids in fish diets. J Appl Ichthyol 11:225–230
Trivers RL, Willard DE (1973) Natural selection of parental ability to vary the sex ratio of offspring. Science 179:90–92
Tsushima M, Kawakami T, Mine M, Matsuno T (1997) The role of carotenoids in the development of the sea urchin Pseudocentrotus depressus. Invert Repro Devel 32:149–153
Weng BC, Chew BP, Wong TS, Park JS, Kim HW, Lepine AJ (2000) Beta-carotene uptake and changes in ovarian steroids and uterine proteins during the estrous cycle in the canine. J Anim Sci 78:1284–1290
Zann RA (1996) The zebra finch. Oxford University Press, Oxford
Acknowledgements
This work was supported by grants from the National Science Foundation (IBN-9514088 and –0130986 to EAR) and the Environmental Protection Agency (STAR Fellowship # 91584201-0 to KJM). We thank Roche Vitamins Inc. for donating carotenoid and placebo beadlets, E. Mackillop for assistance with data collection, T. Van Deusen, P. Smith, and D. Sheils for animal care, Cornell's Behavior Lunch Bunch and Queen's University's EEB group for offering insightful criticism, and A. Badyaev, J. Dale, S. Doucet, M. Hauber, G. Hill, K. Pilz, and A. Stoehr for constructive comments on the manuscript. All procedures were approved by the Institutional Animal Care and Use Committee at Cornell University (Protocol #'s 99-89, 00-34).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McGraw, K.J., Adkins-Regan, E. & Parker, R.S. Maternally derived carotenoid pigments affect offspring survival, sex ratio, and sexual attractiveness in a colorful songbird. Naturwissenschaften 92, 375–380 (2005). https://doi.org/10.1007/s00114-005-0003-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-005-0003-z