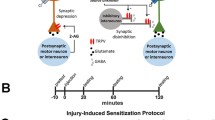
Abstract
Endocannabinoids are traditionally thought to have an analgesic effect. However, it has been shown that while endocannabinoids can depress nociceptive signaling, they can also enhance non-nociceptive signaling. Therefore, endocannabinoids have the potential to contribute to non-nociceptive sensitization after an injury. Using Hirudo verbana (the medicinal leech), a model of injury-induced sensitization was developed in which a reproducible piercing injury was delivered to the posterior sucker of Hirudo. Injury-induced changes in the non-nociceptive threshold of Hirudo were determined through testing with Von Frey filaments and changes in the response to nociceptive stimuli were tested by measuring the latency to withdraw to a nociceptive thermal stimulus (Hargreaves apparatus). To test the potential role of endocannabinoids in mediating injury-induced sensitization, animals were injected with tetrahydrolipstatin (THL), which inhibits synthesis of the endocannabinoid transmitter 2-arachidonoylglycerol (2-AG). Following injury, a significant decrease in the non-nociceptive response threshold (consistent with non-nociceptive sensitization) and a significant decrease in the response latency to nociceptive stimulation (consistent with nociceptive sensitization) were observed. In animals injected with THL, a decrease in non-nociceptive sensitization in injured animals was observed, but no effect on nociceptive sensitization was observed.






Similar content being viewed by others
Availability of data and material
Data are available upon request.
Abbreviations
- ANOVA:
-
Analysis of variance
- 2-AG:
-
2-Arachidonoylglycerol
- CB:
-
Cannabinoid
- CNS:
-
Central nervous system
- DMSO:
-
Dimethyl sulfoxide
- LPS:
-
Lipopolysaccharide
- N cell:
-
Nociceptive cell
- P cell:
-
Pressure cell
- SUDO:
-
Simplified up and down
- THL:
-
Tetrahydrolipstatin
- T cell:
-
Touch cell
- TRPV:
-
Transient receptor potential vanilloid
References
Anderson David J, Adolphs R (2014) A framework for studying emotions across species. Cell 157(1):187–200. https://doi.org/10.1016/j.cell.2014.03.003
Arcourt A, Lechner SG (2015) Peripheral and spinal circuits involved in mechanical allodynia. Pain 156(2):220–221. https://doi.org/10.1097/01.j.pain.0000460818.62406.38
Baba H, Ji RR, Kohno T, Moore KA, Ataka T, Wakai A, Okamoto M, Woolf CJ (2003) Removal of GABAergic inhibition facilitates polysynaptic A fiber-mediated excitatory transmission to the superficial spinal dorsal horn. Mol Cell Neurosci 24(3):818–830
Babcock DT, Landry C, Galko MJ (2009) Cytokine signaling mediates UV-induced nociceptive sensitization in Drosophila larvae. Curr Biol: CB 19(10):799–806. https://doi.org/10.1016/j.cub.2009.03.062
Blackshaw SE, Nicholls JG, Parnas I (1982) Physiological responses, receptive fields and terminal arborizations of nociceptive cells in the leech. J Physiol 326:251–260
Burrell BD (2017) Comparative biology of pain: What invertebrates can tell us about how nociception works. J Neurophysiol 117(4):1461–1473. https://doi.org/10.1152/jn.00600.2016
Carey LM, Slivicki RA, Leishman E, Cornett B, Mackie K, Bradshaw H, Hohmann AG (2016) A pro-nociceptive phenotype unmasked in mice lacking fatty-acid amide hydrolase. Mol Pain. https://doi.org/10.1177/1744806916649192
Crook RJ, Hanlon RT, Walters ET (2013) Squid have nociceptors that display widespread long-term sensitization and spontaneous activity after bodily injury. J Neurosci 33(24):10021–10026. https://doi.org/10.1523/jneurosci.0646-13.2013
De Vita MJ, Moskal D, Maisto SA, Ansell EB (2018) Association of cannabinoid administration with experimental pain in healthy adults: a systematic review and meta-analysis. JAMA Psychiat 75(11):1118–1127. https://doi.org/10.1001/jamapsychiatry.2018.2503
Elphick MR (2012) The evolution and comparative neurobiology of endocannabinoid signalling. Philos Trans R Soc Lond B Biol Sci 367(1607):3201–3215. https://doi.org/10.1098/rstb.2011.0394
Habib AM, Okorokov AL, Hill MN, Bras JT, Lee M-C, Li S, Gossage SJ, van Drimmelen M, Morena M, Houlden H (2019) Microdeletion in a FAAH pseudogene identified in a patient with high anandamide concentrations and pain insensitivity. Br J Anaesth 123(2):e249–e253
Higgins A, Yuan S, Wang Y, Burrell B (2013) Differential modulation of nociceptive versus non-nociceptive synapses by endocannabinoids. Molec Pain 9(1):26
Hill KP, Palastro MD, Johnson B, Ditre JW (2017) Cannabis and pain: a clinical review. Cannabis Cannabinoid Res 2(1):96–104
Im SH, Galko MJ (2012) Pokes, sunburn, and hot sauce: Drosophila as an emerging model for the biology of nociception. Dev Dyn 241(1):16–26. https://doi.org/10.1002/dvdy.22737
Kato A, Punnakkal P, Pernia-Andrade AJ, von Schoultz C, Sharopov S, Nyilas R, Katona I, Zeilhofer HU (2012) Endocannabinoid-dependent plasticity at spinal nociceptor synapses. J Physiol 590(Pt 19):4717–4733. https://doi.org/10.1113/jphysiol.2012.234229
Katona I, Freund TF (2012) Multiple functions of endocannabinoid signaling in the brain. Ann Rev Neurosci 35:529–558. https://doi.org/10.1146/annurev-neuro-062111-150420
Kim YH, Back SK, Davies AJ, Jeong H, Jo HJ, Chung G, Na HS, Bae YC, Kim SJ, Kim JS, Jung SJ, Oh SB (2012) TRPV1 in GABAergic interneurons mediates neuropathic mechanical allodynia and disinhibition of the nociceptive circuitry in the spinal cord. Neuron 74(4):640–647. https://doi.org/10.1016/j.neuron.2012.02.039
Kraft B (2012) Is there any clinically relevant cannabinoid-induced analgesia? Pharmacology 89(5–6):237–246. https://doi.org/10.1159/000337376
Kristan WB, McGirr SJ, Simpson GV (1982) Behavioural and mechanosensory neurone responses to skin stimulation in leeches. J of Exp Biol 96(1):143–160
Kristan WB Jr, Calabrese RL, Friesen WO (2005) Neuronal control of leech behavior. Prog Neurobiol 76(5):279–327
Kuner R, Kuner T (2021) Cellular circuits in the brain and their modulation in acute and chronic pain. Physiol Rev 101(1):213–258
Liu CN, Wall PD, Ben-Dor E, Michaelis M, Amir R, Devor M (2000) Tactile allodynia in the absence of C-fiber activation: altered firing properties of DRG neurons following spinal nerve injury. Pain 85(3):503–521 (pii: S0304395900002517)
Lu Y, Dong H, Gao Y, Gong Y, Ren Y, Gu N, Zhou S, Xia N, Sun YY, Ji RR, Xiong L (2013) A feed-forward spinal cord glycinergic neural circuit gates mechanical allodynia. J Clin Invest 123(9):4050–4062. https://doi.org/10.1172/JCI70026
McMackin MZ, Lewin MR, Tabuena DR, Arreola FE, Moffatt C, Fuse M (2016) Use of von Frey filaments to assess nociceptive sensitization in the hornworm, Manduca sexta. J Neurosci Methods 257:139–146. https://doi.org/10.1016/j.jneumeth.2015.09.015
Mücke M, Phillips T, Radbruch L, Petzke F, Häuser W (2018) Cannabis-based medicines for chronic neuropathic pain in adults. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD012182.pub2
Muller KJ, Nicholls JG, Stent GS (1981) Neurobiology of the leech. Cold Spring Harbor Laboratory Press, Cold Spring Habbor
Nicholls JG, Baylor DA (1968) Specific modalities and receptive fields of sensory neurons in CNS of the leech. J Neurophysiol 31(5):740–756
Pastor J, Soria B, Belmonte C (1996) Properties of the nociceptive neurons of the leech segmental ganglion. J Neurophysiol 75(6):2268–2279
Paulsen RT, Burrell BD (2019) Comparative studies of endocannabinoid modulation of pain. Philos Trans R Soc Lond B Biol Sci 374(1785):20190279. https://doi.org/10.1098/rstb.2019.0279
Pernia-Andrade AJ, Kato A, Witschi R, Nyilas R, Katona I, Freund TF, Watanabe M, Filitz J, Koppert W, Schuttler J, Ji G, Neugebauer V, Marsicano G, Lutz B, Vanegas H, Zeilhofer HU (2009) Spinal endocannabinoids and CB1 receptors mediate C-fiber-induced heterosynaptic pain sensitization. Science 325(5941):760–764. https://doi.org/10.1126/science.1171870
Perry CJ, Baciadonna L (2017) Studying emotion in invertebrates: what has been done, what can be measured and what they can provide. J Exp Biol 220(21):3856–3868
Petitjean H, Pawlowski SA, Fraine SL, Sharif B, Hamad D, Fatima T, Berg J, Brown CM, Jan LY, Ribeiro-da-Silva A, Braz JM, Basbaum AI, Sharif-Naeini R (2015) Dorsal horn parvalbumin neurons are gate-keepers of touch-evoked pain after nerve injury. Cell Rep 13(6):1246–1257. https://doi.org/10.1016/j.celrep.2015.09.080
Sandkuhler J (2009) Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 89(2):707–758. https://doi.org/10.1152/physrev.00025.2008
Sargent PB, Yau KW, Nicholls JG (1977) Extrasynaptic receptors on cell bodies of neurons in central nervous system of the leech. J Neurophysiol 40(2):446–452
Shaw BK, Kristan WB Jr (1995) The whole-body shortening reflex of the medicinal leech: motor pattern, sensory basis, and interneuronal pathways. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 177(6):667–681
Smith ES, Lewin GR (2009) Nociceptors: a phylogenetic view. J Comp Physiol A Neuroeth Sen Neural Beh Physiol 195(12):1089–1106. https://doi.org/10.1007/s00359-009-0482-z
Stockings E, Campbell G, Hall WD, Nielsen S, Zagic D, Rahman R, Murnion B, Farrell M, Weier M, Degenhardt L (2018) Cannabis and cannabinoids for the treatment of people with chronic noncancer pain conditions: a systematic review and meta-analysis of controlled and observational studies. Pain 159(10):1932–1954
Summers T, Hanten B, Peterson W, Burrell B (2017) Endocannabinoids have opposing effects on behavioral responses to nociceptive and non-nociceptive stimuli. Sci Rep 7(1):5793. https://doi.org/10.1038/s41598-017-06114-1
Torsney C, MacDermott AB (2006) Disinhibition opens the gate to pathological pain signaling in superficial neurokinin 1 receptor-expressing neurons in rat spinal cord. J Neurosci 26(6):1833–1843. https://doi.org/10.1523/JNEUROSCI.4584-05.2006
Wagenaar DA (2015) A classic model animal in the 21st century: recent lessons from the leech nervous system. J Exp Biol 218:3353–3359. https://doi.org/10.1242/jeb.113860
Walters ET (1994) Injury-related behavior and neuronal plasticity: an evolutionary perspective on sensitization, hyperalgesia, and analgesia. Int Rev Neurobiol 36:325–427
Walters ET, Williams ACdC (2019) Evolution of mechanisms and behaviour important for pain. Phil Trans R so B 374(1785):1–8. https://doi.org/10.1098/rstb.2019.0275
Wang Y, Burrell BD (2016) Differences in chloride gradients allow for three distinct types of synaptic modulation by endocannabinoids. J Neurophysiol 116(2):619–628. https://doi.org/10.1152/jn.00235.2016
Wang Y, Burrell BD (2018) Endocannabinoid-mediated potentiation of nonnociceptive synapses contributes to behavioral sensitization. J Neurophysiol 119(2):641–651. https://doi.org/10.1152/jn.00092.2017
Wang Y, Summers T, Peterson W, Miiller E, Burrell BD (2015) Differential effects of GABA in modulating nociceptive vs. non-nociceptive synapses. Neuroscience 298:397–409. https://doi.org/10.1016/j.neuroscience.2015.04.040
Woolf CJ, Ma Q (2007) Nociceptors–noxious stimulus detectors. Neuron 55(3):353–364. https://doi.org/10.1016/j.neuron.2007.07.016
Yeomans DC, Proudfit HK (1994) Characterization of the foot withdrawal response to noxious radiant heat in the rat. Pain 59(1):85–94. https://doi.org/10.1016/0304-3959(94)90051-5
Yuan S, Burrell BD (2010) Endocannabinoid-dependent LTD in a nociceptive synapse requires activation of a presynaptic TRPV-like receptor. J Neurophysiol 104(5):2766–2777. https://doi.org/10.1152/jn.00491.2010
Yuan S, Burrell BD (2013) Nonnociceptive afferent activity depresses nocifensive behavior and nociceptive synapses via an endocannabinoid-dependent mechanism. J Neurophysiol 110(11):2607–2616. https://doi.org/10.1152/jn.00170.2013
Funding
This work was supported by the University of South Dakota’s Summer Program for Undergraduate Research in Addiction (SPURA), which is funded by the National Institute on Drug Abuse (NIDA) grant R25-DA033674. It was also supported by a National Institute of Neural Disorders and Stroke (NINDS), NIH R01-NS092716 grant.
Author information
Authors and Affiliations
Contributions
MMJ and BDB designed the experiments. MMJ carried out the experiments. MMJ and BDB carried out data analysis. MMJ wrote the first draft of the manuscript in fulfillment of her honors thesis, with revisions made by BDB. BDB obtained funding.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Ethics approval
No vertebrate animals were used in these experiments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
359_2021_1540_MOESM1_ESM.tif
Supplementary file1 Supplemental Figure 1: Effects of multiple concentrations of THL on response threshold and response latency. (b) 25 and 50 µM THL had no effect on response latency when compared to DMSO-injected animals. 100 µM THL also had no statistically significant effect, although there did appear to be a decrease in response threshold on days 4 and 5. (b) For the initial 4 hour post injury measurement of response latency, 100 µM THL significant reduced response latency compared to the other THL concentrations and the DMSO injected group (*). Data is represented as the average ± the standard error (TIF 6753 kb)
Rights and permissions
About this article
Cite this article
Jorgensen, M.M., Burrell, B.D. Approaches to studying injury-induced sensitization and the potential role of an endocannabinoid transmitter. J Comp Physiol A 208, 313–323 (2022). https://doi.org/10.1007/s00359-021-01540-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-021-01540-z