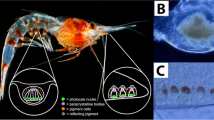
Abstract
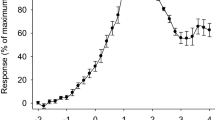
Chemical cues from fish, or kairomones, often impact the behavior of zooplankton. These behavioral changes are thought to improve predator avoidance. For example, marine and estuarine crustacean zooplankton become more sensitive to light after kairomone exposure, which likely deepens their vertical distribution into darker waters during the day and thereby reduces their visibility to fish predators. Here, we show that kairomones from an estuarine fish induce similar behavioral responses in adult brine shrimp (Artemia franciscana) from an endorheic, hypersaline lake, Great Salt Lake, Utah, USA. Given downwelling light stimuli, kairomone-exposed A. franciscana induce a descent response upon dimmer light flashes than they do in the absence of kairomones. Using extracellular electroretinogram (ERG) recordings, we also find that kairomones induce physiological changes in the retina that may lead to increased visual sensitivity, suggesting that kairomone-induced changes to photobehavior are mediated at the photoreceptor level. However, kairomones did not induce structural changes within the eye. Although A. franciscana inhabit endorheic environments that are too saline for most fish, kairomones from an estuarine fish amplify photobehavior in these branchiopod crustaceans. The mechanism for this behavioral change has both similarities to and differences from that described in marine malacostracan crustaceans.




Similar content being viewed by others
References
Aréchiga H, Rodriguez-Sosa L (1998) Circadian clock function in isolated eyestalk tissue of crayfish. Proc R Soc B 265:1819–1823. doi: https://doi.org/10.1098/rspb.1998.0507
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. doi: https://doi.org/10.18637/jss.v067.i01
Battelle B-A (2002) Circadian efferent input to Limulus eyes: anatomy, circuitry, and impact. Microsc Res Tech 58:345–355. doi: https://doi.org/10.1002/jemt.10142
Belovsky GE, Stephens D, Perschon C, Birdsey P, Paul D, Naftz D, Baskin R, Larson C, Mellison C, Luft J, Mosley R, Mahon H, Van Leeuwen J, Allen DV (2011) The Great Salt Lake ecosystem (Utah, USA): long term data and a structural equation approach. Ecosphere 2:33. doi: https://doi.org/10.1890/ES10-00091.1
Bierbower SM, Nadolski J, Cooper RL (2013) Sensory systems and environmental change on behavior during social interactions. Int J Zool 2013:1–16. doi: https://doi.org/10.1155/2013/573802
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White J-SS (2008) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135. doi: https://doi.org/10.1016/j.tree.2008.10.008
Bradley DJ, Forward RB (1984) Phototaxis of adult brine shrimp, Artemia salina. Can J Zool 62:2357–2359. doi: https://doi.org/10.1139/z84-347
Brönmark C, Hansson L-A (2012) Chemical ecology in aquatic systems-an introduction. In: Brönmark C, Hansson L-A (eds) Chemical ecology in aquatic systems. Oxford University Press, New York, pp 1–19
Bryceson KP, McIntyre P (1983) Image quality and acceptance angle in a reflecting superposition. J Comp Physiol A 151:367–380. doi: https://doi.org/10.1007/BF00623912
Buskey EJ, Mann CG, Swift E (1987) Photophobic responses of calanoid copepods: possible adaptive value. J Plankton Res 9:857–870. doi: https://doi.org/10.1093/plankt/9.5.857
Chamberlain SC, Barlow RB (1987) Control of structural rhythms in the lateral eye of Limulus: interactions of natural lighting and circadian efferent activity. J Neurosci 7:2135–2144. doi: https://doi.org/10.1002/jemt.10142
Charpentier CL, Cohen JH (2015) Chemical cues from fish heighten visual sensitivity in larval crabs through changes in photoreceptor structure and function. J Exp Biol 218:3381–3390. doi: https://doi.org/10.1242/jeb.125229
Charpentier CL, Cohen JH (2016) Acidification and γ-aminobutyric acid independently alter behaviour. R Soc Open Sci 3:160311. doi: https://doi.org/10.1098/rsos.160311
Cohen JH, Forward RB (2003) Ctenophore kairomones and modified aminosugar disaccharides alter the shadow response in a larval crab. J Plankton Res 25:203–214. doi: https://doi.org/10.1093/plankt/25.2.203
Cohen JH, Forward RB (2005) Photobehavior as an inducible defense in the marine copepod Calanopia americana. Limnol Oceanogr 50:1269–1277. doi: https://doi.org/10.4319/lo.2005.50.4.1269
Cohen JH, Forward RB (2009) Zooplankton diel vertical migration—a review of proximate control. Oceanogr Mar Biol Ann Rev 47:77–110. doi: https://doi.org/10.1201/9781420094220.ch2
Cohen JH, Cronin TW, Lessios N, Forward RB (2010) Visual physiology underlying orientation and diel behavior in the sand beach amphipod Talorchestia longicornis. J Exp Biol 213:3843–3851. doi: https://doi.org/10.1242/jeb.048686
Cronin TW (1986) Optical design and evolutionary adaptation in crustacean compound eyes. J Crust Biol 6:1–23. doi: https://doi.org/10.1163/193724086X00686
Cronin TW, Forward RB (1988) The visual pigments of crabs I. Spectral characteristics. J Comp Physiol A 162:463–478. doi: https://doi.org/10.1007/BF00612512
Eguchi E, Horikoshi T (1984) Comparison of stimulus-response (V – log I) functions in five types of lepidoteran compound eyes (46 species). J Comp Physiol 154:3–12
Forward RB, Hettler WF (1992) Effects of feeding and predator exposure on photoresponses during diel vertical migration of brine shrimp larvae. Limnol Oceanogr 37:1261–1270. doi: https://doi.org/10.4319/lo.1992.37.6.1261
Forward RB, Rittschof D (1993) Activation of photoresponses of brine shrimp nauplii involved in diel vertical migration by chemical cues from fish. J Plankton Res 15:693–701. doi: https://doi.org/10.1093/plankt/15.6.693
Forward RB, Rittschof D (1999) Brine shrimp larval photoresponses involved in diel vertical migration: activation by fish mucus and modified amino sugars. Limnol Oceanogr 44:1904–1916. doi: https://doi.org/10.4319/lo.1999.44.8.1904
Forward RB, Rittschof D (2000) Alteration of photoresponses involved in diel vertical migration of a crab larva by fish mucus and degradation products of mucopolysaccharides. J Exp Mar Biol Ecol 245:277–292. doi: https://doi.org/10.1016/S0022-0981(99)00169-0
Forward RB, Cronin TW, Stearns DE (1984) Control of diel vertical migration: photoresponses of a larval crustacean. Limnol Oceanogr 29:146–154. doi: https://doi.org/10.4319/lo.1984.29.1.0146
Fox J, Weisberg S (2011) An R companion to applied regression. Sage, Thousand Oaks
Frank TM (1999) Comparative study of temporal resolution in the visual systems of mesopelagic crustaceans. Biol Bull 196:137–144. doi: https://doi.org/10.2307/1542559
Frank TM (2003) Effects of light adaptation on the temporal resolution of deep-sea crustaceans. Integr Comp Biol 43:559–570. doi: https://doi.org/10.1093/icb/43.4.559
Giddings EMP, Stephens DW (1999) Selected aquatic biological investigations in the Great Salt Lake basins, 1875–1998, National Water-Quality Assessment Program. Water-Resources Investigations Report, Salt Lake City
Glantz R (2014) Visual systems of crustaceans. In: Derby C, Thiel M (eds) Nervous systems & control of behavior. The natural history of the crustacean, 3rd edn. Oxford University Press, Oxford, pp 206–234
Greenwood PG (2009) Acquisition and use of nematocysts by cnidarian predators. Toxicon 54:1065–1070. doi: https://doi.org/10.1016/j.toxicon.2009.02.029
Greiner B, Ribi WA, Warrant EJ (2004) Retinal and optical adaptations for nocturnal vision in the halictid bee Megalopta genalis. Cell Tiss Res 316:377–390. doi: https://doi.org/10.1007/s00441-004-0883-9
Greiner B, Ribi WA, Warrant EJ (2005) A neural network to improve dim-light vision? Dendritic fields of first-order interneurons in the nocturnal bee Megalopta genalis. Cell Tiss Res 322:313–320. doi: https://doi.org/10.1007/s00441-005-0034-y
Griffith RW (1974) Environment and salinity tolerance in the genus Fundulus. Copeia 1974:319–331. doi: https://doi.org/10.2307/1442526
Hariyama T, Meyer-Rochow VB, Kawauchi T, Takaku Y, Tsukahara Y (2001) Diurnal changes in retinula cell sensitivities and receptive fields (two-dimensional angular sensitivity functions) in the apposition eyes of Ligia exotica (Crustacea, Isopoda). J Exp Biol 248:239–248
Harzsch S, Dawir RR (1993) On the morphology of the central nervous system in larval stages of Carcinus maenas L (Decapoda, Brachyura). Helgol Meeresunters 47:61–79. doi: https://doi.org/10.1007/BF02366185
Harzsch S, Glötzner J (2002) An immunohistochemical study of structure and development of the nervous system in the brine shrimp Artemia salina Linnaeus, 1758 (Branchiopoda, Anostraca) with remarks on the evolution of the arthropod brain. Arthropod Struct Dev 30:251–270. doi: https://doi.org/10.1016/S1467-8039(02)00012-9
Hays GC (2003) A review of the adaptive significance and ecosystem consequences of zooplankton diel vertical migrations. Hydrobiologia 503:163–170. doi: https://doi.org/10.1023/B:HYDR.0000008476.23617.b0
Hiller-Adams P, Case JF (1988) Eye size of pelagic crustaceans as a function of habitat depth and possession of photophores. Vision Res 28:667–680. doi: https://doi.org/10.1016/0042-6989(88)90047-8
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. doi: https://doi.org/10.1002/bimj.200810425
Land MF (1981) Optics and vision in invertebrates. In: Autrum H (ed) Handbook of sensory physiology, vol. VII/6B. Springer, Berlin, pp 471–592
Lenth RV (2016) Least-squares means: the R package lsmeans. J Stat Softw 69:1–33. doi: https://doi.org/10.18637/jss.v069.i01
Lenz PH (1980) Ecology of an alkali adapted variety of Artemia from Mono Lake, California, U.S.A. In: Browne RA, Sorgeloss P, Trotman CNA (eds) The brine shrimp Artemia, vol 3. Universal Press, Wetter, pp 79–96
Lenz PH, Browne RA (1991) Ecology of Artemia. In: Browne RA, Sorgeloss P, Trotman CNA (eds) Artemia biology. CRC Press, Boca Raton
Loving BL, Waddell KM, Miller CW (2000) Water and salt balance of Great Salt Lake, Utah, and simulation of water and salt movement through the causeway, 1987–98. USGS Water-Resources Investigations Report, Reston pp 00–4221
McKelvey LM (1997) Predator chemical cues mediate zooplankton diel vertical migration. Dissertation, Duke University
Mckelvey LM, Forward RB (1995) Activation of brine shrimp nauplii photoresponses involved in diel vertical migration by chemical cues from visual and non-visual planktivores. J Plankton Res 17:2191–2206. doi: https://doi.org/10.1093/plankt/17.12.2191
Meyer-Rochow VB (2001) The crustacean eye: dark/light adaptation, polarization sensitivity, flicker fusion frequency, and photoreceptor damage. Zool Sci 1197:1175–1197. doi: https://doi.org/10.2108/zsj.18.1175
Meyer-Rochow VB, Tiang KM (1984) The eye of Jasus edwardsii (Crustacea, Decapod): electrophysiology, histology, and behaviour. Zoologica 45:1–61
Nilsson D, Odselius R (1981) A new mechanism for light-dark adaptation in the Artemia compound eye (Anostraca, Crustacea). J Comp Physiol A 143:389–399. doi: https://doi.org/10.1007/BF00611178
Persoone G, Sorgeloos P (1980) General aspects of the ecology and biogeography of Artemia. In: Persoone G, Sorgeloos P, Roels O, Jaspers E (eds) The brine shrimp Artemia. Universal Press, Wetter, pp 3–24
Plachetzki DC, Fong CR, Oakley TH (2012) Cnidocyte discharge is regulated by light and opsin-mediated phototransduction. BMC Biol 10:17. doi: https://doi.org/10.1186/1741-7007-10-17
Price D (1985) Ground water in Utah’s densely populated Wasatch Front area; the challenge and the choices. U.S. Geological Survey Water Supply Paper No. 2232
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Ringelberg J (2010) Diel vertical migration of Zooplankton in lakes and oceans. Springer, Dordrecht
Rittschof D, Cohen JH (2004) Crustacean peptide and peptide-like pheromones and kairomones. Peptides 25:1503–1516. doi: https://doi.org/10.1016/j.peptides.2003.10.024
Sandeman D, Sandeman R, Derby C, Schmidt M (1992) Morphology of the brain of crayfish, crabs, and spiny lobsters: a common nomenclature for homologous structures. Biol Bull 183:304–326. doi: https://doi.org/10.2307/1542217
Sandeman DC, Kenning M, Harzsch S (2014) Adaptive trends in malacostracan brain form and function related to behavior. In: Derby C, Thiel M (eds) Nervous systems & control of behavior. The natural history of the crustacean, 3rd edn. Oxford University Press, Oxford, pp 11–48
Schaum CE, Batty R, Last KS (2013) Smelling danger—alarm cue responses in the polychaete Nereis (Hediste) diversicolor (Müller, 1776) to potential fish predation. PloS ONE 8:e77431. doi: https://doi.org/10.1371/journal.pone.0077431
Schmidt M (2007) The olfactory pathway of decapod crustaceans—an invertebrate model for life-long neurogenesis. Chem Senses 32:365–384. doi: https://doi.org/10.1093/chemse/bjm008
Snyder AW (1979) Physics of vision in compound eyes. In: Autrum H (ed) Handbook of sensory physiology, VII/6A. Springer, Berlin, pp 225–313
Stephens DW (1990) Changes in lake levels, salinity and the biological community of Great Salt Lake (Utah, USA), 1847–1987. Hydrobiologia 197:139–146. doi: https://doi.org/10.1007/BF00026946
Stowe S (1980) Effects of illumination changes on rhabdom synthesis in a crab. J Comp Physiol A 142:19–25. doi: https://doi.org/10.1007/BF00605472
Strausfeld NJ (1998) Crustacean-insect relationships: the use of brain characters to derive phylogeny amongst segmented invertebrates. Brain Behav Evol 52:186–206. doi: https://doi.org/10.1159/000006563
Warrant EJ (1999) Seeing better at night: life style: eye design and the optimum strategy of spatial and temporal summation. Vis Res 39:1611–1630. doi: https://doi.org/10.1016/S0042-6989(98)00262-4
Warrant E, Oskarsson M, Malm H (2014) The remarkable visual abilities of nocturnal insects: neural principles and bioinspired night-vision algorithms. Proc IEE 102:1411–1426. doi: https://doi.org/10.1109/JPROC.2014.2332533
Watson GM, Hessinger DA (1989a) Cnidocytes and adjacent supporting cells form receptor-effector complexes in anemone tentacles. Tissue Cell 21:17–24
Watson GM, Hessinger DA (1989b) Cnidocyte mechanoreceptors are tuned to the movements of swimming prey by chemoreceptors. Science 243:1589–1591. doi: https://doi.org/10.1126/science.2564698
Weiss LC, Kruppert S, Laforsch C, Tollrian R (2012) Chaoborus and gasterosteus anti-predator responses in Daphnia pulex are mediated by independent cholinergic and gabaergic neuronal signals. PloS ONE 7:e36879. doi: https://doi.org/10.1371/journal.pone.0036879
Weiss LC, Leimann J, Tollrian R (2015) Predator-induced defences in Daphnia longicephala: location of kairomone receptors and timeline of sensitive phases to trait formation. J Exp Biol 218:2918–2926. doi: https://doi.org/10.1242/jeb.124552
Wildt M, Harzsch S (2002) A new look at an old visual system: structure and development of the compound eyes and optic ganglia of the brine shrimp Artemia salina Linnaeus, 1758 (Branchiopoda, anostraca). J Neurobiol 52:117–132. doi: https://doi.org/10.1002/neu.10074
Acknowledgements
We thank Haley Glos for her help with animal maintenance, as well as Dr. Daniel Rittschof, Dr. Mark Warner, Dr. Timothy Targett, and two anonymous reviewers for their valuable input on the manuscript. This work was supported in part by awards from the University of Delaware Research Foundation (Award no. 12A00755) to J.H.C. and Marian R. Okie, Joanne Currier Daiber and the Delaware Environmental Institute Fellowships to C.L.C.
Author information
Authors and Affiliations
Contributions
C.L.C. and J.H.C. developed the concept and approach; C.L.C. conducted experiments and data analysis; C.L.C. and J.H.C. prepared and revised the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Specifically, procedures including animals were conducted in accordance with the University of Delaware’s Institutional Animal Care and Use Committee (animal use Protocol no. 1236–2015).
Rights and permissions
About this article
Cite this article
Charpentier, C.L., Cohen, J.H. Kairomones from an estuarine fish increase visual sensitivity in brine shrimp (Artemia franciscana) from Great Salt Lake, Utah, USA. J Comp Physiol A 204, 197–208 (2018). https://doi.org/10.1007/s00359-017-1230-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-017-1230-4