Abstract
Floral nectar is the main source of carbohydrates for many insects including mosquitoes. Nonetheless, the physiological mechanisms underlying feeding on carbohydrates by the Afrotropical malaria mosquito Anopheles gambiae remain poorly understood. Here, we tested whether sugar sensitivity and sugar feeding preferences correlate with longevity in A. gambiae. We also tested whether feeding females on different sugar diets influences their biting behaviours. Electrophysiological recordings show that sugar neurones on the labella of females are most sensitive to sucrose, mixtures of glucose and fructose, and to melezitose; other sugars tested, including glucose and fructose presented alone, only weakly activate these taste neurones. Mosquitoes survive longest on sucrose, the most preferred sugar. Whereas feeding on a mixture of glucose and fructose is preferred over fructose or glucose alone, fructose supports higher longevity than either glucose or the mixture of the two hexoses. Females that had previously fed on glucose show a stronger biting response than those fed on sucrose, perhaps in an effort to compensate for their lower energetic state. These findings contribute to our understanding of the physiological basis of sugar feeding in A. gambiae and indicate how the sugar diet can affect laboratory-reared A. gambiae biting behaviours.








Similar content being viewed by others
References
Altner H, Prillinger L (1980) Ultrastructure of invertebrate chemo-, thermo-, and hygroreceptors and its functional significance. Int Rev Cytol 67:69–139
Andersson IH (1992) The effect of sugar meals and body size on fecundity and longevity of female Aedes communis (Diptera, Culicidae). Physiol Entomol 17:203–207
Burke CJ, Waddell S (2011) Remembering nutrient quality of sugar in Drosophila. Curr Biol 21:746–750
Crawley MJ (2007) The R book, 1st edn. John Wiley & Sons Ltd, England
Dahanukar A, Lei YT, Kwon JY, Carlson JR (2007) Two Gr genes underlie sugar reception in Drosophila. Neuron 56:503–516
Dethier VG, Evans DR, Rhoades MV (1956) Some factors controlling the ingestion of carbohydrates by the blowfly. Biol Bull 111:204–222
Dus M, Min S, Keene AC, Lee GY, Suh GS (2011) Taste-independent detection of the caloric content of sugar in Drosophila. Proc Natl Acad Sci USA 108:11644–11649
Dye C (1986) Vectorial capacity: must we measure all its components? Parasitol. Today 2:203–209
Foster WA (1995) Mosquito sugar feeding and reproductive energetics. Annu Rev Entomol 40:443–474
Freeman GE, Wisotsky Z, Dahanukar A (2014) Detection of sweet tastants by a conserved group of insect gustatory receptors. Proc Natl Acad Sci USA 111:1598–1603
Fujita M, Tanimura T (2011) Drosophila evaluates and learns the nutritional value of sugars. Curr Biol 21:751–755
Garrett-Jones C (1964) Prognosis for interruption of malaria transmission through assessment of mosquitos vectorial capacity. Nature 204:1173–1175
Gary RE, Foster WA (2001) Effects of available sugar on the reproductive fitness and vectorial capacity of the malaria vector Anopheles gambiae (Diptera: Culicidae). J Med Entomol 38:22–28
Gary RE, Foster WA (2004) Anopheles gambiae feeding and survival on honeydew and extra-floral nectar of peridomestic plants. Med Vet Entomol 18:102–107
Hassett CC (1948) The utilization of sugars and other substances by Drosophila. Biol Bull 95:114–123
Hassett CC, Dethier VG, Gans J (1950) A comparison of nutritive values and taste thresholds of carbohydrates for the blowfly. Biol Bull 99:446–453
Heimpel GE, Jervis MA (2005) Does nectar improve biological control by parasitoids. In: Wäckers FL, van Rijn PC, Bruin J (eds) Plant-provided food for carnivorous insects: a protective mutualism and its applications, 1st edn. Cambridge University Press, New York, pp 267–304
Ignell R, Okawa S, Englund JE, Hill SR (2010) Assessment of diet choice by the yellow fever mosquito Aedes aegypti. Physiol Entomol 35:274–286
Kaufmann C, Briegel H (2004) Flight performance of the malaria vectors Anopheles gambiae and Anopheles atroparvus. J Vector Ecol 29:140–153
Kent LB, Robertson HM (2009) Evolution of the sugar receptors in insects. BMC Evol Biol 9:41
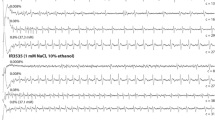
Kessler S, Vlimant M, Guerin PM (2013) The sugar meal of the African malaria mosquito Anopheles gambiae and how deterrent compounds interfere with it: a behavioural and neurophysiological study. J Exp Biol 216:1292–1306
Kröber T, Kessler S, Frei J, Bourquin M, Guerin PM (2010) An in vitro assay for testing mosquito repellents employing a warm body and carbon dioxide as a behavioral activator. J Am Mosq Control Assoc 26:381–386
Manda H, Gouagna LC, Nyandat E, Kabir EW, Jackson RR, Foster WA, Githure JI, Beier JC, Hassanali A (2007a) Discriminative feeding behaviour of Anopheles gambiae s.s. on endemic plants in western Kenya. Med Vet Entomol 21:103–111
Manda H, Gouagna LC, Foster WA, Jackson RR, Beier JC, Githure JI, Hassanali A (2007b) Effect of discriminative plant-sugar feeding on the survival and fecundity of Anopheles gambiae. Malar J 6:113
McIver SB (1982) Sensilla of mosquitoes (Diptera: Culicidae). J Med Entomol 19:489–535
Miyamoto T, Slone J, Song X, Amrein H (2012) A fructose receptor functions as a nutrient sensor in the Drosophila brain. Cell 151:1113–1125
MR4: Malaria Research and Reference Reagent Resource Center (2011) Method in Anopheles research, 2nd edn. pp 267–304. http://www.mr4.org/Portals/3/MR4_Publications/Anopheles%20Protocol%20Manual%20Second%20Ed%20v2011/2011%20Complete%20Manual%20PDF%20TOC.pdf
Nayar JK, Sauerman DM (1971) Physiological effects of carbohydrates on survival, metabolism, and flight potential of female Aedes taeniorhynchus. J Insect Physiol 17:2221–2233
Nayar JK, Sauerman DM (1975) Effects of nutrition on survival and fecundity in Florida mosquitoes. Part 1. Utilization of sugar for survival. J Med Entomol 12:92–98
Nayar JK, Van Handel E (1971) The fuel to sustained mosquito flight. J Insect Physiol 17:471–481
Nyasembe VO, Teal PEA, Mukabana WR, Tumlinson JH, Torto B (2012) Behavioural response of the malaria vector Anopheles gambiae to host plant volatiles and synthetic blends. Parasit Vectors 5:234
Omand E, Dethier VG (1969) An electrophysiological analysis of the action of carbohydrates on the sugar receptor of the blowfly. Proc Nati Acad Sci USA 62:136–143
Pappas LG, Larsen JR (1976) Gustatory hairs on the mosquito, Culiseta inornata. J Exp Zool 196:351–360
Pappas LG, Larsen JR (1978) Gustatory mechanisms and sugar-feeding in the mosquito, Culiseta inornata. Physiol Entomol 3:115–119
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2014) Linear and nonlinear mixed effects models. R package version 3.1-117. http://CRAN.R-project.org/package=nlme
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.r-project.org/
Rasband WS (2011) ImageJ, U.S. National Institutes of Health, Bethesda, Maryland, USA. http://imagej.nih.gov/ij/
Schmidt JM, Friend WG (1991) Ingestion and diet destination in the mosquito Culiseta inornata—effects of carbohydrate configuration. J Insect Physiol 37:817–828
Skaug H, Fournier D, Nielsen A, Magnusson A, Bolker B (2014) Generalized linear mixed models using AD model builder. R package version 0.8.0. http://glmmadmb.r-forge.r-project.org/glmmADMB.html
Stafford JW, Lynd KM, Jung AY, Gordon MD (2012) Integration of taste and calorie sensing in Drosophila. J Neurosci 32:14767–14774
Stone CM, Hamilton IM, Foster WA (2011) A survival and reproduction trade-off is resolved in accordance with resource availability by virgin female mosquitoes. Anim Behav 81:765–774
Stone CM, Jackson BT, Foster WA (2012) Effects of plant-community composition on the vectorial capacity and fitness of the malaria mosquito Anopheles gambiae. Am J Trop Med Hyg 87:727–736
Therneau T (2014) Survival. R package version 2.37–7. http://cran.r-project.org/web/packages/survival/
Vrzal EM, Allan SA, Hahn DA (2010) Amino acids in nectar enhance longevity of female Culex quinquefasciatus mosquitoes. J Insect Physiol 56:1659–1664
Whitehead AT, Larsen JR (1976) Electrophysiological responses of galeal contact chemoreceptors of Apis mellifera to selected sugars and electrolytes. J Insect Physiol 22:1609–1616
Wright GA (2011) Appetitive learning: memories need calories. Curr Biol 21:R301–R302
Acknowledgments
Radu Slobodeanu, Institute of Mathematics, University of Neuchâtel, is cordially acknowledged for his help with statistics. Our gratitude is also extended to Thomas Kröber, Animal Physiology Laboratory, University of Neuchâtel, for improvements to the biting assay in collaboration with Virgile Rumo, HE-ARC, Neuchâtel and Daniel Varidel, Technical Service, University of Neuchâtel. All members of the Animal Physiology Laboratory are acknowledged, particularly Martine Bourquin, for her help in mosquito rearing and Charles Chappuis, Alexandre Gurba as well as two anonymous reviewers for their worthwhile suggestions for improvements to the manuscript. The authors wish to thank Geraldine Wright, Institute of Neuroscience, Newcastle University, and Andrew McMullin, Institute of Biology, University of Neuchâtel for help with a revision of the manuscript. The Guinea pigs used for the mosquito rearing were maintained in an animal house under permit, in compliance with Swiss Federal Regulations. This research was funded by the Swiss National Science Foundation [Grant no. 138207].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
359_2015_978_MOESM1_ESM.pdf
Supplementary material 1 Mean ± s.e.m. feeding preferences by male A. gambiae offered sugar solutions in paired choice tests: 584 mM fructose versus 584 mM glucose; 584 mM glucose or 584 mM fructose versus water; 292 mM glucose with 292 mM fructose versus 584 mM glucose or 584 mM fructose; 292 mM sucrose versus 292 mM melezitose, 584 mM glucose or 584 mM fructose; 292 mM sucrose versus 292 mM glucose with 292 mM fructose or the same solution of 292 mM sucrose. Each choice test consisted of 4–7 replicates with 19 ± 3 males in each. Percentages of mosquitoes tested that fed on both solutions are in grey (PDF 16 kb)
359_2015_978_MOESM2_ESM.pdf
Supplementary material 2 Survival curves in days for male A. gambiae (between 82 and 99 mosquitoes tested per treatment) fed on 584 mM fructose, 584 mM glucose, 292 mM glucose with 292 mM fructose, 292 mM sucrose or water (PDF 19 kb)
Rights and permissions
About this article
Cite this article
Kessler, S., Vlimant, M. & Guerin, P.M. Sugar-sensitive neurone responses and sugar feeding preferences influence lifespan and biting behaviours of the Afrotropical malaria mosquito, Anopheles gambiae . J Comp Physiol A 201, 317–329 (2015). https://doi.org/10.1007/s00359-015-0978-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-015-0978-7