Abstract
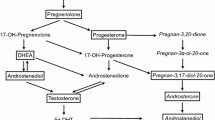
Traditionally, sexual differentiation of the brain was thought to be driven by gonadal hormones, particularly testosterone (T). However, recent studies in songbirds suggest that other steroids may also be important. For example, dehydroepiandrosterone (DHEA) can be synthesized by the gonads, adrenal glands, and/or brain and locally metabolized into T and 17β-estradiol (E2). Here, we examined DHEA and E2 levels in the brain, peripheral tissues, and plasma of wild European starlings (Sturnus vulgaris). In Study 1, samples were collected from males and females at P0 (day of hatch), P6, and P8. In Study 2, samples were collected at P4. At P0, DHEA levels in the diencephalon were higher in males than females. DHEA levels were generally high in the gonads and adrenals, and they were higher in testes than ovaries at P8. Further, E2 levels were non-detectable in most brain samples, suggesting that DHEA was not metabolized to E2 or that locally produced E2 was rapidly inactivated. At P4, DHEA levels in telencephalic regions were lower in males than females. Taken together, these data suggest that sex differences in peripheral DHEA secretion and neural DHEA metabolism at specific ages during development might play a role in sexual differentiation of the songbird brain.





Similar content being viewed by others
References
Ackerley, SK (2010) Histogenesis of the nervous system: the brain. http://www.uoguelph.ca/zoology/devobio/210labs/ecto3.html
Adkins-Regan E, Ascenzi M (1990) Sexual differentiation of behavior in the zebra finch: effect of early gonadectomy or androgen treatment. Horm Behav 24(1):114–127. doi:0018-506X(90)90031-R
Adkins-Regan E, Abdelnabi M, Mobarak M, Ottinger MA (1990) Sex steroid levels in developing and adult male and female zebra finches (Poephila guttata). Gen Comp Endocrinol 78(1):93–109. doi:0016-6480(90)90051-M
Arnold AP (1975) The effects of castration on song development in zebra finches (Poephila guttata). J Exp Zool 191:261–278
Arnold AP (1997) Sexual differentiation of the zebra finch song system: positive evidence, negative evidence, null hypotheses, and a paradigm shift. J Neurobiol 33(5):572–584. doi:10.1002/(SICI)1097-4695(19971105)33:5<572::AID-NEU6>3.0.CO;2-1
Ball GF, MacDougall-Shackleton SA (2001) Sex differences in songbirds 25 years later: what have we learned and where do we go? Microsc Res Tech 54(6):327–334
Bergeron R, de Montigny C, Debonnel G (1996) Potentiation of neuronal NMDA response induced by dehydroepiandrosterone and its suppression by progesterone: effects mediated via sigma receptors. J Neurosci 16(3):1193–1202
Bock W, Farrand J (1980) The number of species and genera of recent birds: a contribution to comparative systematics. Am Mus Novitates 2703:1–29
Butte JC, Kakihana R, Farnham ML, Noble EP (1973) The relationship between brain and plasma corticosterone stress response in developing rats. Endocrinology 92(6):1775–1779
Casto JM, Ball GF (1996) Early administration of 17beta-estradiol partially masculinizes song control regions and alpha2-adrenergic receptor distribution in European starlings (Sturnus vulgaris). Horm Behav 30(4):387–406. doi:10.1006/hbeh.1996.0044
Chard T (1995) An introduction to radioimmunoassays and related techniques, vol 6. Laboratory Techniques in Biochemistry and Molecular Biology, 5th edn. Elsevier Science B.V, Amsterdam
Charlier TD, Po KW, Newman AE, Shah AH, Saldanha CJ, Soma KK (2010) 17beta-Estradiol levels in male zebra finch brain: combining Palkovits punch and an ultrasensitive radioimmunoassay. Gen Comp Endocrinol 167(1):18–26. doi:10.1016/j.ygcen.2010.02.002
Chen F, Knecht K, Birzin E, Fisher J, Wilkinson H, Mojena M, Moreno CT, Schmidt A, Harada S, Freedman LP, Reszka AA (2005) Direct agonist/antagonist functions of dehydroepiandrosterone. Endocrinology 146(11):4568–4576. doi:10.1210/en.2005-0368
Chin EH, Love OP, Clark AM, Williams TD (2005) Brood size and environmental conditions sex-specifically affect nestling immune response in the European starling (Sturnus vulgaris). J Avian Biol 36:549–554
Chin EH, Shah AH, Schmidt KL, Sheldon LD, Love OP, Soma KK (2008) Sex differences in DHEA and estradiol during development in a wild songbird: jugular versus brachial plasma. Horm Behav 54(1):194–202
Compagnone NA, Mellon SH (1998) Dehydroepiandrosterone: a potential signalling molecule for neocortical organization during development. Proc Natl Acad Sci USA 95(8):4678–4683
Corpechot C, Robel P, Axelson M, Sjovall J, Baulieu EE (1981) Characterization and measurement of dehydroepiandrosterone sulfate in rat brain. Proc Natl Acad Sci USA 78(8):4704–4707
Goldsmith AR, Ivings WE, Pearce-Kelly AS, Parry DM, Plowman G, Nicholls TJ, Follett BK (1989) Photoperiodic control of the development of the LHRH neurosecretory system of European starlings (Sturnus vulgaris) during puberty and the onset of photorefractoriness. J Endocrinol 122(1):255–268
Gomez-Sanchez EP, Ahmad N, Romero DG, Gomez-Sanchez CE (2005) Is aldosterone synthesized within the rat brain? Am J Physiol Endocrinol Metab 288(2):E342–E346. doi:10.1152/ajpendo.00355.2004
Goodson JL, Evans A, Soma KK (2005) Neural responses to aggressive challenge correlate with behavior in nonbreeding sparrows. Neuroreport 16(6):1719–1723
Granger DA, Schwartz EB, Booth A, Curran M, Zakaria D (1999) Assessing dehydroepiandrosterone in saliva: a simple radioimmunoassay for use in studies of children, adolescents and adults. Psychoneuroendocrinology 24(5):567–579
Gurney ME, Konishi M (1980) Hormone-induced sexual differentiation of brain and behavior in zebra finches. Science 208(4450):1380–1383. doi:10.1126/science.208.4450.1380
Holloway CC, Clayton DE (2001) Estrogen synthesis in the male brain triggers development of the avian song control pathway in vitro. Nat Neurosci 4(2):170–175
Hutchison JB, Wingfield JC, Hutchison RE (1984) Sex differences in plasma concentrations of steroids during the sensitive period for brain differentiation in the zebra finch. J Endocrinol 103(3):363–369
Jacobs EC, Arnold AP, Campagnoni AT (1999) Developmental regulation of the distribution of aromatase- and estrogen-receptor-mRNA-expressing cells in the zebra finch brain. Dev Neurosci 21:453–472
Jost A (1970) Hormonal factors in the sex differentiation of the mammalian foetus. Philos Trans R Soc Lond B Biol Sci 259(828):119–130
Kallonen SE, Tammimaki A, Piepponen P, Raattamaa H, Ketola RA, Kostiainen R (2009) Discovery of neurosteroid glucuronides in mouse brain. Anal Chim Acta 651(1):69–74. doi:10.1016/j.aca.2009.07.059
Katagiri M, Tatsuta K, Imaoka S, Funae Y, Honma K, Matsuo N, Yokoi H, Isimura K, Ishibashi F, Kagawa N (1998) Evidence that immature rat liver is capable of participating in steroidogenesis by expressing 17alpha hydroxylase/17, 20-lyase P450c17. J Steroid Biochem Mol Biol 64(1/2):121–128
Kirn JR, DeVoogd TJ (1989) Genesis and death of vocal control neurons during sexual differentiation in the zebra finch. J Neurosci 9(9):3176–3187
London SE, Boulter J, Schlinger BA (2003) Cloning of the zebra finch androgen synthetic enzyme CYP17: a study of its neural expression throughout posthatch development. J Comp Neurol 467:496–508
London SE, Monks DA, Wade J, Schlinger BA (2006) Widespread capacity for steroid synthesis in the avian brain and song system. Endocrinology 147(12):5975–5987
Love OP, Chin EH, Wynne-Edwards KE, Williams TD (2005) Stress hormones: a link between maternal condition and sex-biased reproductive investment. Am Nat 166(6):751–766
Lu S-F, Mo Q, Hu S, Garippa C, Simon NG (2003) Dehydroepiandrosterone upregulates neural androgen receptor level and transcriptional activity. J Neurobiol 57(2):163–171
Migues PV, Johnston ANB, Rose SPR (2002) Dehydroepiandosterone and its sulphate enhance memory retention in day-old chicks. Neuroscience 109(2):243–251
Morris JA, Jordan CL, Breedlove SM (2004) Sexual differentiation of the vertebrate nervous system. Nat Neurosci 7(10):1034–1039
Newman AE, Soma KK (2009) Corticosterone and dehydroepiandrosterone in songbird plasma and brain: effects of season and acute stress. Eur J Neurosci 29(9):1905–1914. doi:10.1111/j.1460-9568.2009.06748.x
Newman AEM, Chin EH, Schmidt KL, Bond L, Wynne-Edwards KE, Soma KK (2008a) Analysis of steroids in songbird plasma and brain by coupling solid phase extraction to radioimmunoassay. Gen Comp Endocrinol 155(3):503–510
Newman AEM, Pradhan DS, Soma KK (2008b) Dehydroepiandrosterone and corticosterone are regulated by season and acute stress in a wild songbird: jugular versus brachial plasma. Endocrinology 149(5):2537–2545
Park-Chung M, Malayev A, Purdy RH, Gibbs TT, Farb DH (1999) Sulfated and unsulfated steroids modulate gamma-aminobutyric acidA receptor function through distinct sites. Brain Res 830(1):72–87. doi:S0006-8993(99)01381-5
Phoenix CH, Goy RW, Gerall AA, Young WC (1959) Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology 65:369–382
Pradhan DS, Lau LY, Schmidt KL, Soma KK (2010a) 3beta-HSD in songbird brain: subcellular localization and rapid regulation by estradiol. J Neurochem 115(3):667–675. doi:10.1111/j.1471-4159.2010.06954.x
Pradhan DS, Newman AE, Wacker DW, Wingfield JC, Schlinger BA, Soma KK (2010b) Aggressive interactions rapidly increase androgen synthesis in the brain during the non-breeding season. Horm Behav 57(4–5):381–389. doi:10.1016/j.yhbeh.2010.01.008
Schlinger BA, Arnold AP (1992) Plasma sex steroids and tissue aromatization in hatchling zebra finches: implications for the sexual differentiation of singing behavior. Endocrinology 130(1):289–299
Schlinger BA, Arnold AP (1993) Estrogen synthesis in vivo in the adult zebra finch—additional evidence that circulating estrogens can originate in brain. Endocrinology 133(6):2610–2616
Schlinger BA, Pradhan DS, Soma KK (2008) 3 beta-HSD activates DHEA in the songbird brain. Neurochem Int 52(4–5):611–620
Schmidt KL, Soma KK (2008) Cortisol and corticosterone in the songbird immune and nervous systems: local versus systemic levels during development. Am J Physiol Regul Integr Comp Physiol 295(1):R103–R110. doi:10.1152/ajpregu.00002.2008
Schmidt KL, Chin EH, Shah AH, Soma KK (2009) Cortisol and corticosterone in immune organs and brain of European starlings: developmental changes, effects of restraint stress, comparison with zebra finches. Am J Physiol Regul Integr Comp Physiol 297(1):R42–R51. doi:10.1152/ajpregu.90964.2008
Shirtcliff EA, Granger DA, Schwartz EB, Curran MJ, Booth A, Overman WH (2000) Assessing estradiol in biobehavioral studies using saliva and blood spots: simple radioimmunoassay protocols, reliability, and comparative validity. Horm Behav 38(2):137–147
Silverin B, Sharp P (1996) The development of the hypothalamic–pituitary–gonadal axis in juvenile great tits. Gen Comp Endocrinol 103:150–166
Soma KK (2006) Testosterone and aggression: Berthold, birds and beyond. J Neuroendocrinol 18(7):543–551
Soma KK, Wingfield JC (2001) Dehydroepiandrosterone in songbird plasma: seasonal regulation and relationship to territorial aggression. Gen Comp Endocrinol 123:144–155
Soma KK, Alday NA, Hau M, Schlinger BA (2004) Dehydroepiandrosterone metabolism by 3 beta-hydroxysteroid dehydrogenase/Delta 5-Delta 4 isomerase in adult zebra finch brain: sex difference and rapid effect of stress. Endocrinology 145(4):1668–1677
Tagawa N, Katagiri M, Kobayashi Y (2005) Developmental changes of serum steroids produced by cytochrome P450c17 in rat. Steroids 71(2):165–170
Taves MD, Schmidt KL, Ruhr IM, Kapusta K, Prior NH, Soma KK (2010) Steroid concentrations in plasma, whole blood and brain: effects of saline perfusion to remove blood contamination from brain. PLoS One 5(12):e15727. doi:10.1371/journal.pone.0015727
Toran-Allerand CD, Tinnikov A, Singh RJ, Nethrapalli IS (2005) 17alpha-estradiol: a brain active estrogen? Endocrinology 146(9):3843–3850
Wade J, Arnold AP (1996) Functional testicular tissue does not masculinize development of the zebra finch song system. Proc Natl Acad Sci USA 93(11):5264–5268
Williams TD, Dawson A, Nicholls TJ, Goldsmith AR (1987) Reproductive endocrinology of free-living nestling and juvenile starlings, Sturnus vulgaris; an altricial species. J Zool 212(4):619–628
Zann RE (1996) The Zebra Finch: Synthesis of Field and Laboratory Studies. Oxford University Press, New York
Acknowledgments
We thank D. Pradhan, L. Sheldon, and O. Love for help in the field, Dr. T.D. Williams for access to the Langley field site, the UBC Farm, and the Davis family for allowing us to conduct this research at the Davistead farms. We also wish to thank Dr. J. Weinberg for sharing equipment. This research was funded by grants from the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Michael Smith Foundation for Health Research to K.K.S., and NSERC Canada Graduate Scholarships to E.H.C and K.L.S. All experiments were conducted under a UBC Animal Care permit (A04-277) and followed the guidelines of the Canadian Council on Animal Care.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. H. Shah and E. H. Chin are co-first authors.
Rights and permissions
About this article
Cite this article
Shah, A.H., Chin, E.H., Schmidt, K.L. et al. DHEA and estradiol levels in brain, gonads, adrenal glands, and plasma of developing male and female European starlings. J Comp Physiol A 197, 949–958 (2011). https://doi.org/10.1007/s00359-011-0655-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-011-0655-4