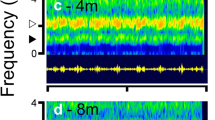
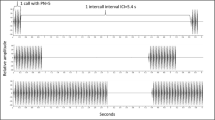
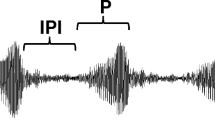
Abstract
During the reproductive season, male Hyla versicolor produce advertisement calls to attract females. Females exhibit phonotaxis and approach the individual callers, resulting in amplexus. For frogs that call from dense choruses, the extent to which and the range from which a male’s advertisement call within a chorus can be heard by a receptive female leading to phonotaxis is unclear. We investigated females’ responses to natural choruses in the field and found that they were attracted and showed directed orientation to breeding choruses at distances up to 100 m. To assess the role of acoustic cues in the directed orientation, we conducted acoustic playback experiments in the laboratory using conspecific call and noise as stimuli, as well as chorus sounds (that contained calls from a focal male) recorded at various distances, all played at naturalistic intensities. Using two response metrics (females’ normalized response times and their phonotaxis trajectories) we found that, unlike the field experiments, females oriented and were attracted to chorus sounds from 1 to 32 m only, but not from >32 m, or to band-limited noise. Possible reasons for the observed difference in phonotaxis behavior in the two experimental conditions were discussed.






Similar content being viewed by others
References
Aubin J, Jouventin P (2002) Localisation of an acoustic signal in a noisy environment: the display call of the king penguin Aptenodytes patagonicus. J Exp Biol 205:3793–3798
Batschelet E (1981) Circular statistics in biology. Academic Press, London
Beckers O, Schul J (2004) Phonotaxis in Hyla versicolor (Anura, Hylidae): the effect of absolute call amplitude. J Comp Physiol A 190:869–876
Bee MA (2007a) Sound source segregation in grey treefrogs: spatial release from masking by the sound of a chorus. Anim Behav 74:549–558
Bee MA (2007b) Selective phonotaxis by male wood frogs (Rana sylvatica) to the sound of a chorus. Behav Ecol Sociobiol 61:955–966
Bee MA, Swanson EM (2007) Auditory masking of anuran advertisement calls by road traffic noise. Anim Behav 74:1765–1776
Bush SL, Gerhardt HC, Schul J (2002) Pattern recognition and call preferences in treefrogs (Anura: Hylidae): a quantitative analysis using a no-choice paradigm. Anim Behav 63:7–14
Candolin U (2003) The use of multiple cues in mate choice. Biol Rev 78:575–595
Dabelsteen T, Larsen ON, Pedersen SB (1993) Habitat-induced degradation of sound signals: quantifying the effects of communication sounds and bird location on blur ratio, excess attenuation, and signal-to-noise ration in blackbird song. J Acoust Soc Am 93:2206–2220
Diekamp B, Gerhardt HC (1995) Selective phonotaxis to advertisement calls in the gray treefrog Hyla versicolor: behavioral experiments and neurophysiological correlates. J Comp Physiol A 117:173–190
Eggert C (2002) Use of fluorescent pigments and implantable transmitters to track a fossorial toad (Pelobates fuscus). Herpetol J 12:69–74
Feng AS, Shofner WP (1981) Peripheral basis of sound localization in anurans, acoustic properties of the frog’s ear. Hear Res 5:201–216
Feng AS, Schul J (2007) Sound processing in real-world environments. In: Narins PM, Feng AS, Fay RR, Popper AN (eds) Hearing and sound communication in amphibians. Springer, New York, pp 323–350
Feng AS, Gerhardt HC, Capranica RR (1976) Sound localization behavior of the green treefrog (Hyla cinerea) and the barking treefrog (Hyla gratiosa). J Comp Physiol A 107:241–252
Forrest TG (1994) From sender to receiver: propagation and environmental effects on acoustic signals. Am Zool 34:644–654
Gerhardt HC (1975) Sound pressure levels and radiation patterns of the vocalizations of some North American frogs and toads. J Comp Physiol A 102:1–12
Gerhardt HC (1981) Mating call recognition in the green treefrog (Hyla cinerea): importance of two frequency bands as a function of sound pressure level. J Comp Physiol A 144:9–16
Gerhardt HC (2005) Acoustic spectral preferences in two cryptic species of grey treefrogs: implications for mate choice and sensory mechanisms. Anim Behav 70:39–48
Gerhardt HC, Doherty JA (1988) Acoustic communication in the gray treefrog, Hyla versicolor: evolutionary and neurobiological implications. J Comp Physiol A162:261–278
Gerhardt HC, Klump GM (1988a) Masking of acoustic signals by the chorus background noise in the green tree frog: a limitation on mate choice. Anim Behav 36:1247–1249
Gerhardt HC, Klump GM (1988b) Phonotactic responses and selectivity of barking treefrogs (Hyla gratiosa) to chorus sounds. J Comp Physiol A 163:795–802
Gerhardt HC, Schul J (1999) A quantitative analysis of behavioral selectivity for pulse rise-time in the gray treefrog, Hyla versicolor. J Comp Physiol A 185:33–40
Gerhardt HC, Tanner SD, Corrigan CM, Walton HC (2000) Female preference functions based on call duration in the gray tree frog (Hyla versicolor). Behav Ecol 11:663–669
Holland J, Dabelsteen T, Pedersen SB, Larsen ON (1998) Degradation of wren Troglodytes troglodytes song: implications for information transfer and ranging. J Acoust Soc Am 103:2154–2166
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54:187–211
Johnson JR, Semlitsch RD (2003) Defining core habitat of local populations of the gray treefrog (Hyla versicolor) based on choice of oviposition site. Oecologia 137:205–210
Jones DL, Ratnam R (2009) Blind location and separation of callers in a natural chorus using a microphone array. J Acoust Soc Am 126:895–910
Jørgensen MB, Gerhardt HC (1991) Directional hearing in the gray tree frog Hyla versicolor: eardrum vibrations and phonotaxis. J Comp Physiol A 169:177–183
Jouventin P, Aubin T, Lengagne T (1999) Finding a parent in a king penguin colony: the acoustic system of individual recognition. Anim Behav 57:1175–1183
Lang F (2000) Acoustic communication distances of a Gomphocerine grasshopper. Bioacoustics 10:233–258
Lohr B, Wright TF, Dooling RJ (2003) Detection and discrimination of natural calls in masking noise by birds: estimating the active space of a signal. Anim Behav 65:763–777
Luther DA, Wiley RH (2009) Production and perception of communicatory signals in a noisy environment. Biol Lett 5:183–187
Murphy CG (2008) Assessment of distance to potential mates by female barking treefrogs (Hyla gratiosa). J Comp Psychol 122:264–273
Narins PM, Zelick RD (1988) The effects of noise on auditory processing and behavior in amphibians. In: Fritzsch B, Ryan MJ, Wilczynski W, Hetherington TE, Walkowiak W (eds) The evolution of the amphibian auditory system. Wiley, New York, pp 511–536
Narins PM, Feng AS, Fay RR, Popper AN (2007) Hearing and sound communication in amphibians. Springer, New York
Penna M, Solis R (1998) Frog call intensities and sound propagation in the South American temperate forest region. Behav Ecol Sociobiol 42:371–381
Ptacek MB, Gerhardt HC, Sage RD (1994) Speciation by polyploidy in treefrogs: multiple origins of the tetraploid, Hyla versicolor. Evolution 48:898–908
Richards DG, Wiley RH (1980) Reverberations and amplitude fluctuations in the propagation of sounds in a forest: implications for animal communication. Am Nat 115:381–399
Schwartz JJ, Gerhardt HC (1995) Directionality of the auditory system and call pattern recognition during acoustic interference in the gray tree frog, Hyla versicolor. Aud Neurosci 1:195–206
Schwartz JJ, Huth K, Hutchin T (2004) How long do females really listen? Assessment time for female mate choice in the grey treefrog, Hyla versicolor. Anim Behav 68:533–540
Sinsch U (1990a) Migration and orientation in anuran amphibians. Ethol Ecol Evol 2:65–79
Sinsch U (1990b) The orientation behaviour of three toad species (genus Bufo) displaced from the breeding site. In: Hanke W (ed) Biology and physiology of amphibians. Gustav Fischer Verlag, Stuttgart, pp 75–83
Slabbekoorn H, Bouton N (2008) Soundscape orientation: a new field in need of sound investigation. Anim Behav 76:e5–e6
Swanson EM, Tekman S, Bee MA (2007) Do female frogs exploit inadvertent social information to locate breeding aggregations? Can J Zool 85:921–932
Windmiller B (1996) Tracking techniques useful for field studies of anuran orientation and movement. Herpet Rev 27:13–15
Wollerman L (1999) Acoustic interference limits call detection in a Neotropical frog Hyla ebraccata. Anim Behav 57:529–536
Wollerman L, Wiley RD (2002) Background noise from a natural chorus alters female discrimination of male calls in a Neotropical frog. Anim Behav 63:15–22
Zar J (1999) Biostatistical analysis. Prentice-Hall, Upper Saddle River, NJ
Acknowledgments
We thank the Missouri Department of Conservation and the University of Missouri-Columbia for allowing studies and capture permits. Carl Gerhardt generously provided advice and material support. Vincent Marshall, Noah Gordon, Oliver Beckers, and Sarah Bush provided valuable advice and comments on the present study. Sarah Humfeld and Alison Welch aided in coordinating field activities and in providing material support. Eric Rozier provided many helpful comments and assistance with the statistical analysis. We would also like to thank the University of Missouri-Columbia undergraduate students who greatly aided in both animal collection and experimental execution: Paul Kimball, Stephanie Kleyman, and Leah Reynolds. This research was supported by a National Science Foundation Collaborative Research in Computational Neuroscience (CRCNS-0422073) grant to ASF and a National Science Foundation grant (IOB-0445286) to JS. All experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Illinois.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christie, K., Schul, J. & Feng, A.S. Phonotaxis to male’s calls embedded within a chorus by female gray treefrogs, Hyla versicolor . J Comp Physiol A 196, 569–579 (2010). https://doi.org/10.1007/s00359-010-0544-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-010-0544-2