Abstract
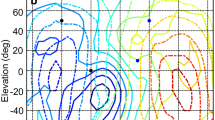
Barn owls use interaural intensity differences to localize sounds in the vertical plane. At a given elevation the magnitude of the interaural intensity difference cue varies with frequency, creating an interaural intensity difference spectrum of cues which is characteristic of that direction. To test whether space-specific cells are sensitive to spectral interaural intensity difference cues, pure-tone interaural intensity difference tuning curves were taken at multiple different frequencies for single neurons in the external nucleus of the inferior colliculus. For a given neuron, the interaural intensity differences eliciting the maximum response (the best interaural intensity differences) changed with the frequency of the stimulus by an average maximal difference of 9.4±6.2 dB. The resulting spectral patterns of these neurally preferred interaural intensity differences exhibited a high degree of similarity to the acoustic interaural intensity difference spectra characteristic of restricted regions in space. Compared to stimuli whose interaural intensity difference spectra matched the preferred spectra, stimuli with inverted spectra elicited a smaller response, showing that space-specific neurons are sensitive to the shape of the spectrum. The underlying mechanism is an inhibition for frequency-specific interaural intensity differences which differ from the preferred spectral pattern. Collectively, these data show that space-specific neurons are sensitive to spectral interaural intensity difference cues and support the idea that behaving barn owls use such cues to precisely localize sounds.














Similar content being viewed by others
Abbreviations
- ABI:
-
average binaural intensity
- HRTF:
-
head-related transfer function
- ICx:
-
external nucleus of the inferior colliculus
- IID:
-
interaural intensity difference
- ITD:
-
interaural time difference
- OT:
-
optic tectum
- RMS:
-
root mean square
- VLVp:
-
nucleus ventralis lemnisci laterale, pars posterior
References
Adolphs R (1993) Bilateral inhibition generates neuronal responses tuned to interaural level differences in the auditory brainstem of the barn owl. J Neurosci 13:3647–3668
Albeck Y (1997) Inhibition sensitive to interaural time difference in the barn owl’s inferior colliculus. Hear Res 109:102–108
Armstrong-James M, Millar J (1979) Carbon fibre microelectrodes. J Neurosci Methods 1:279–287
Arthur BJ (2002) Neural computations leading to space-specific auditory responses in the barn owl. PhD thesis, California Institute of Technology
Blauert J (1997) Spatial hearing: the psycophysics of human sound localization (revised edition). MIT Press, Cambridge, Massachusetts
Carlile S (1990) The auditory periphery of the ferret. I. Directional response properties and the pattern of interaural level differences. JASA 88:2180–2195
Chung W, Carlile S, Leong P (2000) A performance adequate computational model for auditory localization JASA 107:432–445
Coles RB, Guppy A (1988) Directional hearing in the barn owl (Tyto alba) J Comp Physiol A 163:117–133
Datum MS, Palmieri F, Moiseff A (1996) An artificial neural network for sound localization using binaural cues JASA 100:372–383
Egnor SER (2000) Cellular mechanisms for resolving phase ambiguity in the owl’s inferior colliculus. PhD thesis, California Institute of Technology
Euston DR, Takahashi TT (2002) From spectrum to space: the contribution of level difference cues to spatial receptive fields in the barn owl inferior colliculus. J Neurosci 22:284–293
Gill D, Troyansky L, Nelken I (2000) Auditory localization using direction-dependent spectral information. Neurocomputing 32–33:767–773
Gold JI, Knudsen EI (1999) Hearing impairment induces frequency-specific adjustments in auditory spatial tuning in the optic tectum of young owls. J Neurophysiol 82:2197–2209
Gold JI, Knudsen EI (2000) Abnormal auditory experience induces frequency-specific adjustments in unit tuning for binaural localization cues in the optic tectum of juvenile owls. J Neurosci 20:862–877
Gorman RP, Sejnowski JT (1988a) Analysis of hidden units in a layered network trained to classify sonar targets. Neural Netw 1:75–89
Gorman RP, Sejnowski TJ (1988b) Learned classification of sonar targets using a massively parallel network. IEEE Trans Acoust Speech Sign Proc 36:1135–1140
Keller CH, Hartung K, Takahashi TT (1998) Head-related transfer functions of the barn owl: measurement and neural responses. Hear Res 118:13–34
Kulkarni A, Colburn HS (1998) Role of spectral detail in sound-source localization. Nature 396:747–749
LeCun Y, Boser B, Denker JS, Henderson D, Howard RE, Hubbard W, Jackel LD (1989) Backpropagation applied to handwritten zip code recognition. Neural Comput 1:541–551
Lippmann RP (1989) Review of neural networks for speech recognition. Neural Comput 1:1–38
Manley GA, Köppl C, Konishi M (1988) A neural map of interaural intensity differences in the brain stem of the barn owl. J Neurosci 8:2665–2676
Marrese CA (1987) Preparation of strongly adherent platinum black coatings Anal Chem 59:217–218
Mazer JA (1995) Integration of parallel processing streams in the inferior colliculus of the barn owl. PhD thesis, California Institute of Technology
Middlebrooks JC, Green DM (1991) Sound localization by human listeners. Annu Rev Psychol 42:135–159
Moiseff A (1989a) Bi-coordinate sound localization by the barn owl. J Comp Physiol A 164:637–644
Moiseff A (1989b) Binaural disparity cues available to the barn owl for sound localization. J Comp Physiol A 164:629–636
Moiseff A, Konishi M (1981) Neuronal and behavioral sensitivity to binaural time differences in the owl. J Neurosci 1:40–48
Moiseff A, Konishi M (1983) Binaural characteristics of units in the owl’s brainstem auditory pathway: precursors of restricted spatial receptive fields. J Neurosci 3:2553–2562
Nandy D, Ben-Arie J (2001) Neural models for auditory localization based on spectral cues. Neurol Res 23:489–500
Neti C, Young ED, Schneider MH (1992) Neural network models of sound localization based on directional filtering by the pinna. JASA 92:3140–3156
Olsen JF, Knudsen EI, Esterly SD (1989) Neural maps of interaural time and intensity differences in the optic tectum of the barn owl. J Neurosci 9:2591–2605
Payne RS (1971) Acoustic location of prey by barn owls (Tyto alba). J Exp Biol 54:535–573
Pittman CU Jr, Jiang W, Yue ZR, Leon y Leon CA (1999) Surface area and pore size distribution of microporous carbon fibers prepared by electrochemical oxidation. Carbon 37:85–96
Poganiatz I, Wagner H (2001) Sound-localization experiments with barn owls in virtual space: influence of broadband interaural level difference on headturning behavior. J Comp Physiol A 187:225–233
Rumelhart D, Hinton G, Williams R (1986) Learning internal representations by error propagation. In: Rumelhart D, McClelland J (eds) Parallel distributed processing: explorations in the microstructure of cognition, vol 1. Foundations. MIT Press, Cambridge
Takahashi T, Konishi M (1986) Selectivity for interaural time difference in the owl’s midbrain. J Neurosci 6:3413–3422
Acknowledgements
Thanks are due to Masakazu Konishi for guidance and support, to Kip Keller, Klaus Hartung, and Terry Takahashi for the generous use of their data, to Chris Malek, Gene Akutagawa, Mike Walsh, Herb Adams, and Janet Baer for technical help, to the National Science Foundation and the National Institutes of Health (DC00134) for funding, and to Masakazu Konishi, José Luis Peña, and Sharad Shanbhag for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arthur, B.J. Sensitivity to spectral interaural intensity difference cues in space-specific neurons of the barn owl. J Comp Physiol A 190, 91–104 (2004). https://doi.org/10.1007/s00359-003-0476-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-003-0476-1