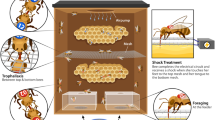
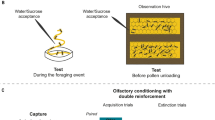
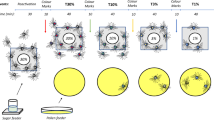
Abstract
Trophallaxis among adult worker honeybees is the transfer of liquid food by mouth from one individual to another. Within the colony, nectar foragers perform offering contacts (as food-donors) to transfer the contents of their crops to recipient nest-mates and, in addition, they also perform begging contacts (as food-receivers). The biological relevance of these last interactions remains unknown. Previous evidence suggests that begging may be involved in the exchange of information on food resources that occurs naturally between employed foragers and nest-mates. This work was aimed to reveal possible connections between the information obtained while foraging and the begging behavior displayed inside the nest. Experiments were intended to (1) analyze whether chemosensory information obtained while foraging, i.e., odors and sucrose concentrations, affects begging behavior, and (2) determine whether resource uncertainty enhances begging contacts. Results showed that: (1) most begging contacts lasted less than 1 s, a duration which only allows receiving food samples from nest-mates; (2) the diversity of odors and sucrose concentrations at the feeding place enhances the occurrence of begging contacts; and (3) an increased resource uncertainty enhances the forager begging behavior. In addition, results suggest that foragers may direct their begging contacts frequently to other employed nectar foragers.



Similar content being viewed by others
Abbreviations
- RF:
-
rate feeder
- T:
-
treatment
References
Beutler R (1950) Zeit und Raum im Leben der Sammelbiene. Naturwissenschaften 37:102–105
Beutler R, Schöntag A (1940) Über die Nektarabscheidung einiger Nutzpflanzen. Z Vergl Physiol 28:254–285
Bitterman ME (1988) Vertebrate-invertebrate comparisons. NATO ASI Series-Intel. Evol Biol 17:251–275
Bitterman ME, Menzel R, Fietz A, Schäfer S (1983) Classical conditioning of proboscis extension in honeybees (Apis mellifera). J Comp Psychol 97:107–119
Brandstetter M, Crailsheim K, Heran H (1988) Provisioning of food in the honeybee before foraging. BIONA Rep 6:129–148
Butler CG (1951) The importance of perfume on the discovery of food by the worker honeybee Apis mellifera L. Proc R Soc Lond Ser B 138:403–413
Crailsheim K (1998) Trophallactic interactions in the adult honeybee (Apis mellifera L.). Apidologie 29:97–112
De Marco RJ, Farina WM (2001) Changes in food source profitability affect the trophallactic and dance behavior of forager honeybees. Behav Ecol Sociobiol 50:441–449
Doolittle GM (1907) Where do the field-bees deposit their loads? Am Bee J 42:653–654
Farina WM (1996) Food-exchange by foragers in the hive: a means of communication among honey bees? Behav Ecol Sociobiol 38:59–64
Farina WM, Wainselboim A (2001) Thermographic recordings show that honeybees may receive nectar from foragers even during short trophallactic contacts. Insectes Soc 48:360–362
Free JB (1956) A study of the stimuli which release the food begging and offering responses of worker honeybees. Br J Anim Behav 4:94–101
Free JB (1957) The transmission of food between worker honeybees. Br J Anim Behav 5:41–47
Free JB (1959) The transfer of food between the adult members of a honeybee community. Bee World 40:193–201
Frisch K von (1934) Über den Geschmackssinn der Bienen. Z Vergl Physiol 21:1–156
Frisch K von (1946) Die ´Sprache' der Bienen und ihre Nutzanwendung in der Land-wirtschaft. Experientia 2:397–404
Frisch K von (1967) The dance language and orientation of bees. Harvard University Press, Cambridge, MA
Gerber B, Geberzahn F, Hellstern J, Klein O, Kowalsky D, Wustenberg D, Menzel R (1996) Honey bees transfer olfactory memories built during flower visits to a proboscis extension paradigm in the laboratory. Anim Behav 52:1079–1085
Greggers U, Menzel R (1993) Memory dynamics and foraging strategies of honeybees. Behav Ecol Sociobiol 32:17–29
Hammer M, Menzel R (1995) Learning and memory in the honeybee. J Neurosci 15:1617–1630
Heinrich B (1975) Energetics of pollination. Annu Rev Ecol Syst 6:139–170
Istomina-Tsvetkova KP (1960) Contribution to the study of trophic relations in adult worker bees. XVII Int Beekeep Congr Bologna-Roma 2:361–368
Korst PJAM, Velthuis HHW (1982) The nature of trophallaxis in honeybees. Insectes Soc 29:209–221
Lindauer M (1961) Communication among social bees. Harvard University Press, Cambridge, MA
Maurizio A (1960) Bienenbotanik. In: Büdel A, Herold E (eds) Biene und Bienenzucht. München, pp 68–104
Menzel R (1985) Learning in honeybees in an ecological and behavioral context. In: Hölldobler B, Lindauer M (eds) Experimental behavioral ecology. Fischer, Stuttgart, pp 55–74
Menzel R (1990) Learning, memory, and "cognition" in honey bees. In: Kesner RP, Olten DS (eds) Neurobiology of comparative cognition. Erlbaum, Hillsdale, NJ, pp 237–292
Menzel R (1999) Memory dynamics in the honeybee. J Comp Physiol 185:323–340
Menzel R, Müller U (1996) Learning and memory in honeybees: from behavior to neural substrates. Annu Rev Neurosci 19:379–404
Michener CD (1974) The social behavior of the bees. Harvard University Press, Cambridge, MA
Nixon HL, Ribbands CR (1952) Food transmission within the honeybee community. Proc R Soc Lond Ser B 140:43–50
Núñez JA (1966) Quantitative Beziehungen zwischen den Eigenschaften von Futterquellen und dem Verhalten von Sammelbienen. Z Vergl Physiol 53:142–164
Núñez JA (1970) The relationship between sugar flow and foraging and recruiting behavior of honeybees (Apis mellifera L.). Anim Behav 18:527–538
Pyke GH (1978) Optimal foraging in bumblebees and coevolution with their plants. Oecologia 36:281–293
Ribbands CR (1952) Division of labour in the honeybee community. I. The response of crop-attached bees to the scent of their crop. Proc R Entomol Soc Lond B 140:32–42
Ribbands CR (1954) Communication between honeybees, I: the response of crop-attached bees to the scent of their crop. Proc R Entomol Soc Lond A 29:10–12
Ribbands CR (1955) The scent perception of the honeybee. Proc R Entomol Soc Lond B 143:367–379
Rösch GA (1925) Untersuchungen über die Arbeitsteilung im Bienenstaat, Teil: die Tätigkeit im normalen Bienenstaatte und ihre Beziehung zum Alter der Arbeitsbienen. Z Vergl Physiol 2:571–631
Takeda K (1961) Classical conditioned response in the honey bee. J Insect Physiol 6:168–179
Waddington KD, Holden LR (1979) Optimal foraging: on flower selection by bees. Am Nat 114:179–196
Wilson EO (1971) The insect societies. Harvard University Press, Cambridge, MA
Zar JH (1996) Biostatistical analysis, 3rd edn. Prentice-Hall, New Jersey
Acknowledgements
We are deeply indebted to Prof. Dr. J. A. Núñez for his permanent encouragement, fruitful discussions and valuable comments. Prof. Núñez also designed the 3-feeder patch employed during the experiments. We thank Koos Biesmeijer (Utrecht, The Netherlands), Giovanni Galizia (Berlin, Germany), and Martin Giurfa (Toulouse, France) for suggestions and valuable comments on early versions of the manuscript. We also thank M. Gil and J. Goyret for advisements on statistics. R.D.M. gratefully acknowledges helpful assistance with the experiments by Maria Ariadna Szapiro. This study was partially supported by funds from ANPCYT (PICT 98-03103) and from the University of Buenos Aires (JX53) to W.M.F., and from CONICET to both authors. The present experiments comply with the Principles of animal care (publication No. 86-23, -revised 1985) of the National Institute of Health and the corresponding national current laws.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Dr. Josué A. Núñez on the occasion of his 78th birthday.
Rights and permissions
About this article
Cite this article
De Marco, R.J., Farina, W.M. Trophallaxis in forager honeybees (Apis mellifera): resource uncertainty enhances begging contacts?. J Comp Physiol A 189, 125–134 (2003). https://doi.org/10.1007/s00359-002-0382-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-002-0382-y