Abstract
Background
Prostate cancer (PCa) is the most common cancer in men in the US and androgen deprivation therapy (ADT) is the most frequently used systemic therapy for PCa. Data suggest that ADT is associated with an increased risk of new-onset diabetes mellitus (NODM) and cardiovascular complications. As the incidence and mortality of PCa are highest among the African American (AA) population, it is important to evaluate the difference in the incidence of NODM and ischemic heart disease (IHD) between AA men compared to Caucasian men.
Methods
This is a retrospective cohort study utilizing the TriNetX database to assess NODM and IHD risk, risk difference, and risk ratio (RR) after recent ADT initiation in an AA cohort and a Caucasian cohort of patients with PCa. Propensity score matching (PSM) was performed by age, BMI, and confounding comorbidities.
Results
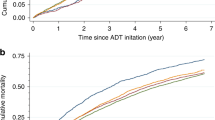
After matching, the cohort included 1159 AA patients and 843 Caucasian patients with NODM after ADT initiation. The IHD cohort included 1269 AA patients and 1248 Caucasian patients. The risk of incidence of NODM is higher among AA men at 11.6% risk compared to Caucasian men at 7.4%. The risk difference is 4.1% (95% CI = 3.4, 4.9) p = 0.000. The RR is 1.56 (95% CI = 1.43, 1.70). In contrast, risk difference and risk ratio of IHD was not significant between AA and Caucasian groups.
Conclusion
ADT exposure increases the risk of NODM in men with PCa, especially among AA men compared with Caucasian men. Men receiving ADT should be monitored routinely for signs and symptoms of metabolic syndrome and diabetes. Targeted close monitoring of AA men on ADT would be critical to prevent and treat metabolic complications with potential of reducing disparities in PCa morbidity.


Similar content being viewed by others
References
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73(1):17–48. https://doi.org/10.3322/caac.21763
Sasse AD, Sasse E, Carvalho AM, Macedo LT (2012) Androgenic suppression combined with radiotherapy for the treatment of prostate adenocarcinoma: a systematic review. BMC Cancer 12(1):54. https://doi.org/10.1186/1471-2407-12-54
Nguyen PL et al (2015) Adverse effects of androgen deprivation therapy and strategies to mitigate them. Eur Urol 67(5):825–836. https://doi.org/10.1016/j.eururo.2014.07.010
Ketchandji M, Kuo YF, Shahinian VB, Goodwin JS (2009) Cause of death in older men after the diagnosis of prostate cancer. J Am Geriatr Soc 57(1):24–30. https://doi.org/10.1111/J.1532-5415.2008.02091.X
Virani SS et al (2020) Heart disease and stroke statistics—2020 update: a report from the American Heart Association. Circulation 141(9):e139–e596
Grossmann M et al (2008) Low testosterone levels are common and associated with insulin resistance in men with diabetes. J Clin Endocrinol Metab 93(5):1834–1840
Smith MR et al (2002) Changes in body composition during androgen deprivation therapy for prostate cancer. J Clin Endocrinol Metab 87(2):599–603
Smith MR, Lee H, Nathan DM (2006) Insulin sensitivity during combined androgen blockade for prostate cancer. J Clin Endocrinol Metab 91(4):1305–1308
Wang H, Sun X, Zhao L, Chen X, Zhao J (2016) Androgen deprivation therapy is associated with diabetes: evidence from meta-analysis. J Diabetes Investig 7(4):629–636
Saylor PJ et al (2012) Changes in biomarkers of inflammation and angiogenesis during androgen deprivation therapy for prostate cancer. Oncologist 17(2):212–219
Osei K, Schuster DP (1994) Ethnic differences in secretion, sensitivity, and hepatic extraction of insulin in Black and White Americans. Diabet Med 11(8):755–762. https://doi.org/10.1111/j.1464-5491.1994.tb00349.x
Magi-Galluzzi C et al (2011) TMPRSS2-ERG gene fusion prevalence and class are significantly different in prostate cancer of Caucasian, African–American and Japanese patients. Prostate 71(5):489–497. https://doi.org/10.1002/pros.21265
Geerkens MJM, Pouwels NSA, Beerlage HP (2020) The effectiveness of lifestyle interventions to reduce side effects of androgen deprivation therapy for men with prostate cancer: a systematic review. Qual Life Res 29:843–865
Funding
National Cancer Institute, 5P30CA056036.
Author information
Authors and Affiliations
Contributions
SG contributed to protocol/project development, data collection or management, data analysis, and manuscript writing/editing. WKK, EM, ES, CZ-J, and K-YW were involved in protocol/project development and manuscript writing/editing. H-SJ was involved in protocol/project development, data collection or management, and manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
There was no COI to declare.
Informed consent
This study is exempt by Thomas Jefferson University’s IRB and do not require informed consent as a retrospective cohort using de-identified patient data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gomaa, S., Kelly, W.K., Mitchell, E. et al. Racial disparities in new-onset diabetes mellitus in prostate cancer patients on androgen deprivation therapy: a retrospective analysis of TriNetX data. World J Urol 41, 2351–2357 (2023). https://doi.org/10.1007/s00345-023-04531-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-023-04531-8