Abstract
Purpose
To perform a feasibility phase study of a panel of putative protein biomarkers and determine whether it can identify and predict tumor recurrence in patients with non-muscle-invasive bladder cancer (NMIBC) on follow-up.
Methods
We prospectively analyzed the urine of 152 patients previously treated for NMIBC. Quantitative expression of plasminogen activator inhibitor-1 (PAI-1), DJ-1, apolipoprotein A-I (apoA-1), matrix metallopeptidase-9 (MMP-9), and interleukin-8 (IL-8) was assessed by enzyme-linked immunosorbent assay and compared amongst patients with and without bladder cancer recurrence at urine collection and during 3 years of follow-up. Tumor recurrence was confirmed by pathologic analysis. We performed a prediction analysis, excluding patients with recurrence at the start of the study, and assessed the influence of previous use of intravesical BCG on the level of biomarkers.
Results
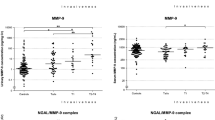
Median follow-up time was 47 months (interquartile range 39–50 months). Sixteen patients (10.5%) were diagnosed with recurrence at the start of the study, and 21 (15.4%) were diagnosed during the study. Three biomarker proteins (apoA-1, MMP-9, and IL-8) appear to hold diagnostic potential [odds ratio (OR) = 12.9; 95% CI 3.5–47.4]; while, PAI-1 and IL-8 predict recurrence (OR = 4.1; 95% CI 1.4–11.4). Previous use of intravesical BCG did not affect biomarker levels.
Conclusion
In the feasibility phase, the panel of urine biomarkers analyzed detected and predicted recurrence of NMIBC and provided reliable results in patients who had previously used intravesical BCG. Validation studies are required to confirm the panel clinical utility.

BC bladder cancer

Similar content being viewed by others
References
van Rhijn BW, Burger M, Lotan Y, Solsona E, Stief CG, Sylvester RJ, Witjes JA, Zlotta AR (2009) Recurrence and progression of disease in non-muscle-invasive bladder cancer: from epidemiology to treatment strategy. Eur Urol 56:430–442
Avritscher EBC, Cooksley CD, Grossman HB, Sabichi AL, Hamblin L, Dinney CP, Elting LS (2006) Clinical model of lifetime cost of treating bladder cancer and associated complications. Urology 68:549–553
van der Aa MN, Steyerberg EW, Sen EF, Zwarthoff EC, Kirkels WJ, van der Kwast TH, Essink-Bot ML (2008) Patients’ perceived burden of cystoscopic and urinary surveillance of bladder cancer: a randomized comparison. BJU Int 101:1106–1110
Soria F, Droller MJ, Lotan Y, Gontero P, D’Andrea D, Gust KM, Rouprêt M, Babjuk M, Palou J, Shariat SF (2018) An up-to-date catalog of available urinary biomarkers for the surveillance of non-muscle invasive bladder cancer. World J Urol 36:1981–1995
Sayyid RK, Sayyid AK, Klaassen Z et al (2018) Replacing surveillance cystoscopy with urinary biomarkers in followup of patients with non-muscle-invasive bladder cancer: patients and urologic oncologists’ perspectives. Can Urol Assoc J 12:E210–E218
Rosser CJ, Ross S, Chang M, Dai Y, Mengual L, Zhang G, Kim J, Urquidi V, Alcaraz A, Goodison S (2013) Multiplex protein signature for the detection of bladder cancer in voided urine samples. J Urol 190:2257–2262
Kumar P, Nandi S, Tan TZ et al (2015) Highly sensitive and specific novel biomarkers for the diagnosis of transitional bladder carcinoma. Oncotarget 6:13539–13549
Soukup V, Kalousová M, Capoun O, Sobotka R, Breyl Z, Pešl M, Zima T, Hanuš T (2015) Panel of urinary biomarkers for non-invasive detection of primary and recurrent urothelial urinary bladder carcinoma. Urol Int 95:56–64
Babjuk M, Oosterlinck W, Sylvester R, Kaasinen E, Böhle A, Palou-Redorta J, Rouprêt M, European Association of Urology (EAU) (2011) EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder, the 2011 update. Eur Urol 59:997–1008
Li S, Wei X, He J, Tian X, Yuan S, Sun L (2018) Plasminogen activator inhibitor-1 in cancer research. Biomed Pharmacother 105:83–94
Urquidi V, Kim J, Chang M, Dai Y, Rosser CJ, Goodison S (2012) CCL18 in a multiplex urine-based assay for the detection of bladder cancer. PLoS ONE 7:e37797
Vasseur S, Afzal S, Tardivel-Lacombe J, Park DS, Iovanna JL, Mak TW (2009) DJ-1/PARK7 is an important mediator of hypoxia-induced cellular responses. Proc Natl Acad Sci USA 106:1111–1116
Jafri H, Alsheikh-Al AA, Karas RH (2010) Baseline and on-treatment high-density lipoprotein cholesterol and the risk of cancer in randomized controlled trials of lipid-altering therapy. J Am Coll Cardiol 55:2846–2854
Shang Z, Wang J, Wang X, Yan H, Bui B, Jia C, Wang Q, Cui X, Li J, Ou T (2018) Preoperative serum apolipoprotein A-I levels predict long-term survival in non-muscle invasive bladder cancer patients. Cancer Manag Res 10:1177–1190
Chen CL, Lin TS, Tsai CH, Wu CC, Chung T, Chien KY, Wu M, Chang YS, Yu JS, Chen YT (2013) Identification of potential bladder cancer markers in urine by abundant-protein depletion coupled with quantitative proteomics. J Proteomics 85:28–43
Huang H (2018) Matrix metalloproteinase-9 (MMP-9) as a cancer biomarker and MMP9 biosensors: recent advances. Sensors (Basel) 18(10):E3249. https://doi.org/10.3390/s18103249
Reis ST, Leite KR, Piovesan LF et al (2012) Increased expression of MMP-9 and IL-8 are correlated with poor prognosis of bladder cancer. BMC Urol 13:12–18
Nutt JE, Durkan GC, Mellon JK, Lunec J (2012) Matrix metalloproteinases (MMPs) in bladder cancer: the induction of MMP9 by epidermal growth factor and its detection in urine. BJU Int 91:99–104
Waugh DJJ, Wilson C (2008) The interleukin-8 pathway in cancer. Clin Cancer Res 14:6735–6741
Urquidi V, Chang M, Dai Y, Kim J, Wolfson ED, Goodison S, Rosser CJ (2012) IL-8 as a urinary biomarker for the detection of bladder cancer. BMC Urol 12:12
Shimizu Y, Furuya H, Bryant Greenwood P, Chan O, Dai Y, Thornquist MD, Goodison S, Rosser CJ (2016) A multiplex immunoassay for the non-invasive detection of bladder cancer. J Transl Med 14:31
Frantzi M, van Kessel KE, Zwarthoff EC et al (2016) Development and validation of urine-based peptide biomarker panels for detecting bladder cancer in a multi-center study. Clin Cancer Res 22:4077–4086
Wood DP Jr (2012) Urothelial tumors of the bladder. In: Wein AJ (ed) Campbell-Walsh urology, vol 2, 1st edn. Elsevier, Philadelphia, pp 2309–2334
Khanna A, Yerram N, Zhu H, Kim S, Abouassaly R (2019) Utilization of BCG for non-muscle invasive bladder cancer in an era of BCG supply shortages. Urology 124:120–126
Boehm BE, Cornell JE, Wang H, Mukherjee N, Oppenheimer JS, Svatek RS (2017) Efficacy of bacillus Calmette-Guérin strains for treatment of nonmuscle invasive bladder cancer: a systematic review and network meta-analysis. J Urol 198:503–510
Lotan Y, Shariat SF, Schmitz-Drager BJ et al (2010) Considerations on implementing diagnostic markers into clinical decision making in bladder cancer. Urol Oncol 28:441–448
Li C, Li H, Zhang T, Li J, Liu L, Chang J (2014) Discovery of Apo-A1 as a potential bladder cancer biomarker by urine proteomics and analysis. Biochem Biophys Res Commun 446:1047–1052
Gopalakrishna A, Fantony JJ, Longo TA, Owusu R, Foo WC, Dash R, Denton BT, Inman BA (2017) Anticipatory positive urine tests for bladder cancer. Ann Surg Oncol 24:1747–1753
Seideman C, Canter D, Kim P, Cordon B, Weizer A, Oliva I, Rao J, Inman BA, Posch M, Herr H, Lotan Y (2015) Multicenter evaluation of the role of UroVysion FISH assay in surveillance of patients with bladder cancer: Does FISH positivity anticipate recurrence? World J Urol 33:1309–1313
Acknowledgements
This research was funded by FAPESP, 2016/07917-5, Sao Paulo, Brazil (Grant 64.000,00 $).
Author information
Authors and Affiliations
Contributions
VS: project development, data collection, manuscript writing. STR: project development, data analysis. NV: data collection and analysis. FPG: data collection. KRL: project development and supervision. MS: manuscript editing, project development and supervision. WCN: manuscript editing, project development and supervision.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Srougi, V., Reis, S.T., Viana, N. et al. Prospective evaluation of a urinary biomarker panel to detect and predict recurrence of non-muscle-invasive bladder cancer. World J Urol 39, 453–459 (2021). https://doi.org/10.1007/s00345-020-03188-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-020-03188-x