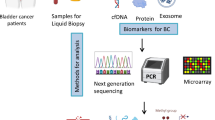
Abstract
Purpose
Urothelial carcinoma of the bladder (UCB) is clinically and genetically a highly heterogeneous disease. Treatment decisions are usually based on histopathological workup and molecular diagnostics on tissue biopsies of the primary tumor or the metastatic site. Next to completely different molecular genotypes of phenotypically similar tumors, standard biopsies do not unconditionally allow real-time insight during the natural course of disease progression. Indeed, in UCB there is an imperative need of biomarkers for improving clinical staging, detecting minimal residual disease, predicting therapy response and prognosis and finally enabling patient stratification for multimodal, individualized treatment and therapy monitoring.Liquid biopsies of blood-based circulating biomarkers have evolved from bench to bedside in some cancer entities.
Methods
In a narrative review we are summerizing the latest evidence on CTC and ctDNA in muscle-invasive and metastatic UCB.
Results
In this review, we summarize the current status, limitations and future needs of circulating tumor cells (CTC) and cell-free circulating tumor DNA (ctDNA) in UCB. Moreover, we discuss the potential clinical application of CTC and ctDNA as prognostic markers at different UCB stages and their value for target therapy guidance.
Conclusions
CTC and ctDNA are promising circulating biomarkers in UCB, but none of both has progressed from bench to bedside yet. These markers may support outcome prognostication, patient counseling follow-up monitoring, and potentially decision-making regarding chemotherapy. Further prospective clinical or randomized studies are urgently warranted.
Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2017) Cancer statistics, 2017. CA 67(1):7–30. https://doi.org/10.3322/caac.21387
Alfred Witjes J, Lebret T, Comperat EM, Cowan NC, De Santis M, Bruins HM, Hernandez V, Espinos EL, Dunn J, Rouanne M, Neuzillet Y, Veskimae E, Van der Heijden AG, Gakis G, Ribal MJ (2017) Updated 2016 EAU guidelines on muscle-invasive and metastatic bladder cancer. Eur Urol 71(3):462–475. https://doi.org/10.1016/j.eururo.2016.06.020
Soave A, Engel O, Von Amsberg G, Becker A, Dahlem R, Shariat SF, Fisch M, Rink M (2015) Management of advanced bladder cancer in the era of targeted therapies. Minerva Urol Nefrol 67(2):103–115
Soave A, Riethdorf S, Pantel K, Fisch M, Rink M (2015) Do circulating tumor cells have a role in deciding on adjuvant chemotherapy after radical cystectomy? Curr Urol Rep 16(7):46. https://doi.org/10.1007/s11934-015-0520-z
Oing C, Rink M, Oechsle K, Seidel C, von Amsberg G, Bokemeyer C (2016) Second line chemotherapy for advanced and metastatic urothelial carcinoma: vinflunine and beyond—a comprehensive review of the current literature. J Urol 195(2):254–263. https://doi.org/10.1016/j.juro.2015.06.115
von der Maase H, Sengelov L, Roberts JT, Ricci S, Dogliotti L, Oliver T, Moore MJ, Zimmermann A, Arning M (2005) Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol 23(21):4602–4608. https://doi.org/10.1200/jco.2005.07.757
Rink M (2017) The landscape of genetics and biomarkers in bladder cancer. Transl Androl Urol 6(6):1027–1030. https://doi.org/10.21037/tau.2017.11.12
Rink M, Shariat SF, Soave A (2016) Liquid biopsies in bladder cancer-did we find the Holy Grail for biomarker analyses? Transl Androl Urol 5(6):980–983. https://doi.org/10.21037/tau.2016.09.03
Pantel KA-PC, Riethdorf S (2009) Cancer micrometastases. Nat Rev Clin Oncol 6(6):335–339. https://doi.org/10.1038/nrclinonc.2009.44
Pantel K, Alix-Panabières C (2013) Real-time liquid biopsy in cancer patients: fact or fiction? Can Res 73(21):6384–6388. https://doi.org/10.1158/0008-5472.can-13-2030
Schwarzenbach H, Hoon DS, Pantel K (2011) Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer 11(6):426–437. https://doi.org/10.1038/nrc3066
Soave A, Chun FK, Hillebrand T, Rink M, Weisbach L, Steinbach B, Fisch M, Pantel K, Schwarzenbach H (2017) Copy number variations of circulating, cell-free DNA in urothelial carcinoma of the bladder patients treated with radical cystectomy: a prospective study. Oncotarget 8(34):56398–56407. https://doi.org/10.18632/oncotarget.17657
Stroun M, Lyautey J, Lederrey C, Olson-Sand A, Anker P (2001) About the possible origin and mechanism of circulating DNA apoptosis and active DNA release. Clin Chim Acta 313(1–2):139–142
Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, Carter SL, Stewart C, Mermel CH, Roberts SA, Kiezun A, Hammerman PS, McKenna A, Drier Y, Zou L, Ramos AH, Pugh TJ, Stransky N, Helman E, Kim J, Sougnez C, Ambrogio L, Nickerson E, Shefler E, Cortes ML, Auclair D, Saksena G, Voet D, Noble M, DiCara D, Lin P, Lichtenstein L, Heiman DI, Fennell T, Imielinski M, Hernandez B, Hodis E, Baca S, Dulak AM, Lohr J, Landau DA, Wu CJ, Melendez-Zajgla J, Hidalgo-Miranda A, Koren A, McCarroll SA, Mora J, Crompton B, Onofrio R, Parkin M, Winckler W, Ardlie K, Gabriel SB, Roberts CWM, Biegel JA, Stegmaier K, Bass AJ, Garraway LA, Meyerson M, Golub TR, Gordenin DA, Sunyaev S, Lander ES, Getz G (2013) Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499(7457):214–218. https://doi.org/10.1038/nature12213
Birkenkamp-Demtroder K, Nordentoft I, Christensen E, Hoyer S, Reinert T, Vang S, Borre M, Agerbaek M, Jensen JB, Orntoft TF, Dyrskjot L (2016) Genomic alterations in liquid biopsies from patients with bladder cancer. Eur Urol 70(1):75–82. https://doi.org/10.1016/j.eururo.2016.01.007
Riethdorf S, Soave A, Rink M (2017) The current status and clinical value of circulating tumor cells and circulating cell-free tumor DNA in bladder cancer. Transl Androl Urol 6(6):1090–1110. https://doi.org/10.21037/tau.2017.09.16
Pantel K, Alix-Panabieres C (2016) Liquid biopsy: potential and challenges. Mol Oncol 10(3):371–373. https://doi.org/10.1016/j.molonc.2016.01.009
Rink M, Cha E, Green D, Hansen J, Robinson B, Lotan Y, Sagalowsky A, Chun F, Karakiewicz P, Fisch M, Scherr D, Shariat S (2012) Biomolecular predictors of urothelial cancer behavior and treatment outcomes. Curr Urol Rep 13(2):122–135. https://doi.org/10.1007/s11934-012-0237-1
Alix-Panabieres C, Pantel K (2016) Clinical applications of circulating tumor cells and circulating tumor DNA as liquid biopsy. Cancer Discov 6(5):479–491. https://doi.org/10.1158/2159-8290.cd-15-1483
Joosse SA, Gorges TM, Pantel K (2015) Biology, detection, and clinical implications of circulating tumor cells. EMBO Mol Med 7(1):1–11. https://doi.org/10.15252/emmm.201303698
Azevedo R, Soares J, Peixoto A, Cotton S, Lima L, Santos LL, Ferreira JA (2018) Circulating tumor cells in bladder cancer: emerging technologies and clinical implications foreseeing precision oncology. Urol Oncol 36(5):221–236. https://doi.org/10.1016/j.urolonc.2018.02.004
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappen LW, Hayes DF (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351(8):781–791
Cohen SJ, Punt CJ, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, Picus J, Morse M, Mitchell E, Miller MC, Doyle GV, Tissing H, Terstappen LW, Meropol NJ (2008) Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol 26(19):3213–3221. https://doi.org/10.1200/jco.2007.15.8923
De Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, Doyle GV, Terstappen LW, Pienta KJ, Raghavan D (2008) Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 14(19):6302–6309. https://doi.org/10.1158/1078-0432.ccr-08-0872
Abrahamsson J, Aaltonen K, Engilbertsson H, Liedberg F, Patschan O, Ryden L, Sjodahl G, Gudjonsson S (2017) Circulating tumor cells in patients with advanced urothelial carcinoma of the bladder: association with tumor stage, lymph node metastases, FDG-PET findings, and survival. Urol Oncol 35(10):606.e609–606.e616. https://doi.org/10.1016/j.urolonc.2017.05.021
Naoe M, Ogawa Y, Morita J (2007) Detection of circulating urothelial cancer cells in the blood using the cell search system. Cancer 109(7):1439–1445
Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, Tibbe AG, Uhr JW, Terstappen LW (2004) Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res 10(20):6897–6904. https://doi.org/10.1158/1078-0432.ccr-04-0378
Riethdorf S, Fritsche H, Muller V, Rau T, Schindlbeck C, Rack B, Janni W, Coith C, Beck K, Janicke F, Jackson S, Gornet T, Cristofanilli M, Pantel K (2007) Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: a validation study of the cell search system. Clin Cancer Res 13(3):920–928
Alva A, Friedlander T, Clark M, Huebner T, Daignault S, Hussain M, Lee C, Hafez K, Hollenbeck B, Weizer A, Premasekharan G, Tran T, Fu C, Ionescu-Zanetti C, Schwartz M, Fan A, Paris P (2015) Circulating tumor cells as potential biomarkers in bladder cancer. J Urol 194(3):790–798. https://doi.org/10.1016/j.juro.2015.02.2951
Flaig TW, Wilson S, van Bokhoven A, Varella-Garcia M, Wolfe P, Maroni P, Genova EE, Morales D, Lucia MS (2011) Detection of circulating tumor cells in metastatic and clinically localized urothelial carcinoma. Urology 78(4):863–867. https://doi.org/10.1016/j.urology.2011.05.045
Guzzo T, McNeil B, Bivalacqua T, Elliott D, Sokoll L, Schoenberg M (2012) The presence of circulating tumor cells does not predict extravesical disease in bladder cancer patients prior to radical cystectomy. Urol Oncol 30(1):44–48
Karl A, Tritschler S, Hofmann S, Stief C, Schindlbeck C (2009) Perioperative search for circulating tumor cells in patients undergoing radical cystectomy for bladder cancer. Eur J Med Res 14:487–490
Rink M, Chun FK, Dahlem R, Soave A, Minner S, Hansen J, Stoupiec M, Coith C, Kluth LA, Ahyai SA, Friedrich MG, Shariat SF, Fisch M, Pantel K, Riethdorf S (2012) Prognostic role and HER2 expression of circulating tumor cells in peripheral blood of patients prior to radical cystectomy: a prospective study. Eur Urol 61(4):810–817. https://doi.org/10.1016/j.eururo.2012.01.017
Rink M, Soave A, Chun FK, Dahlem R, Hansen J, Minner S, Kluth LA, Friedrich MG, Heinzer H, Heuer R, Ahyai SA, Engel O, Eichelberg C, Shariat SF, Pantel K, Fisch M, Riethdorf S (2012) Preoperative detection of a single circulating tumour cell is independently associated with inferior outcome in patients with urothelial carcinoma of the bladder. J Urol 187(4):e356–e357
Seideman CA, Herati AS, Pan S, Cho JS, Cinman N, Tai J, Liu E, Shi E, Vira M (2009) Prognostic correlation of circulating tumor cells in the setting of urothelial and renal cell carcinoma. J Urol 181(4):108
Rink M, Chun FKH, Minner S, Friedrich M, Mauermann O, Heinzer H, Huland H, Fisch M, Pantel K, Riethdorf S (2011) Detection of circulating tumour cells in peripheral blood of patients with advanced non-metastatic bladder cancer. BJU Int 107(10):1668–1675. https://doi.org/10.1111/j.1464-410x.2010.09562.x
Soave A, Riethdorf S, Dahlem R, Minner S, Weisbach L, Engel O, Fisch M, Pantel K, Rink M (2017) Detection and oncological effect of circulating tumour cells in patients with variant urothelial carcinoma histology treated with radical cystectomy. BJU Int 119(6):854–861. https://doi.org/10.1111/bju.13782
Soave A, Riethdorf S, Dahlem R, von Amsberg G, Minner S, Weisbach L, Engel O, Fisch M, Pantel K, Rink M (2017) A nonrandomized, prospective, clinical study on the impact of circulating tumor cells on outcomes of urothelial carcinoma of the bladder patients treated with radical cystectomy with or without adjuvant chemotherapy. Int J Cancer 140(2):381–389. https://doi.org/10.1002/ijc.30445
Willis D, Kamat AM (2015) Nonurothelial bladder cancer and rare variant histologies. Hematol Oncol Clin North Am 29(2):237–252. https://doi.org/10.1016/j.hoc.2014.10.011
Zhang Z, Fan W, Deng Q, Tang S, Wang P, Xu P, Wang J, Yu M (2017) The prognostic and diagnostic value of circulating tumor cells in bladder cancer and upper tract urothelial carcinoma: a meta-analysis of 30 published studies. Oncotarget 8(35):59527–59538. https://doi.org/10.18632/oncotarget.18521
Cohen SJ, Punt CJA, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, Picus J, Morse M, Mitchell E, Miller MC, Doyle GV, Tissing H, Terstappen LWMM, Meropol NJ (2008) Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J Clin Oncol 26(19):3213–3221. https://doi.org/10.1200/jco.2007.15.8923
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ, Terstappen LWMM, Hayes DF (2004) Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med 351(8):781–791. https://doi.org/10.1056/nejmoa040766
De Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, Doyle GV, Terstappen LWWM, Pienta KJ, Raghavan D (2008) Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res 14(19):6302–6309. https://doi.org/10.1158/1078-0432.ccr-08-0872
Meeks JJ, Bellmunt J, Bochner BH, Clarke NW, Daneshmand S, Galsky MD, Hahn NM, Lerner SP, Mason M, Powles T, Sternberg CN, Sonpavde G (2012) A systematic review of neoadjuvant and adjuvant chemotherapy for muscle-invasive bladder cancer. Eur Urol 62(3):523–533. https://doi.org/10.1016/j.eururo.2012.05.048
Leow JJ, Martin-Doyle W, Rajagopal PS, Patel CG, Anderson EM, Rothman AT, Cote RJ, Urun Y, Chang SL, Choueiri TK, Bellmunt J (2014) Adjuvant chemotherapy for invasive bladder cancer: a 2013 updated systematic review and meta-analysis of randomized trials. Eur Urol 66(1):42–54. https://doi.org/10.1016/j.eururo.2013.08.033
Okegawa T, Hayashi K, Hara H, Nutahara K, Higashihara E (2010) Immunomagnetic quantification of circulating tumor cells in patients with urothelial cancer. Int J Urol 17:254–258
Gallagher D, Milowsky M, Ishill N (2009) Detection of circulating tumor cells in patients with urothelial cancer. Ann Oncol 20(2):305–308
Naoe M, Ogawa Y, Takeshita K, Iwamoto S, Miyazaki A (2008) Use of the cell search circulating tumor cell test for monitoring urothelial cancer: two case reports of metastatic urothelial cancer. South Med J 101:439–441
De Santis M, Bauernhofer T, Stoiber F, Höltl L, Sommerhuber A, Klocker J, Meran JG, Klikovich J, Colleselli D, Dittrich C, Pantel K (2016) Circulating tumor cells (CTC) and HER-2 status on CTC and primary tumor in urothelial cancer (UC) patients refractory to platinum based chemotherapy. J Clin Oncol 34(15_suppl):4520. https://doi.org/10.1200/jco.2016.34.15_suppl.4520
Alix-Panabieres C, Pantel K (2017) Characterization of single circulating tumor cells. FEBS Lett. https://doi.org/10.1002/1873-3468.12662
Riethdorf S, O’Flaherty L, Hille C, Pantel K (2018) Clinical applications of the cell search platform in cancer patients. Adv Drug Deliv Rev 125:102–121. https://doi.org/10.1016/j.addr.2018.01.011
Lima L, Neves M, Oliveira MI, Dieguez L, Freitas R, Azevedo R, Gaiteiro C, Soares J, Ferreira D, Peixoto A, Fernandes E, Montezuma D, Tavares A, Ribeiro R, Castro A, Oliveira M, Fraga A, Reis CA, Santos LL, Ferreira JA (2017) Sialyl-Tn identifies muscle-invasive bladder cancer basal and luminal subtypes facing decreased survival, being expressed by circulating tumor cells and metastases. Urol Oncol 35(12):675.e671–675.e678. https://doi.org/10.1016/j.urolonc.2017.08.012
Kuske A, Gorges TM, Tennstedt P, Tiebel AK, Pompe R, Preisser F, Prues S, Mazel M, Markou A, Lianidou E, Peine S, Alix-Panabieres C, Riethdorf S, Beyer B, Schlomm T, Pantel K (2016) Improved detection of circulating tumor cells in non-metastatic high-risk prostate cancer patients. Sci Rep 6:39736. https://doi.org/10.1038/srep39736
Sieuwerts AM, Mostert B, Bolt-de Vries J, Peeters D, de Jongh FE, Stouthard JM, Dirix LY, van Dam PA, Van Galen A, de Weerd V, Kraan J, van der Spoel P, Ramirez-Moreno R, van Deurzen CH, Smid M, Yu JX, Jiang J, Wang Y, Gratama JW, Sleijfer S, Foekens JA, Martens JW (2011) mRNA and microRNA expression profiles in circulating tumor cells and primary tumors of metastatic breast cancer patients. Clin Cancer Res 17(11):3600–3618. https://doi.org/10.1158/1078-0432.ccr-11-0255
Necchi A, Rink M, Giannatempo P, Raggi D, Xylinas E (2018) Immunotherapy for metastatic urothelial carcinoma: status quo and the future. Curr Opin Urol 28(1):1–7. https://doi.org/10.1097/mou.0000000000000457
Skibinski DA (2018) Noninvasive detection of PD-L1 on circulating tumor cells in patient blood samples. Future Oncol 14(13):1237–1240. https://doi.org/10.2217/fon-2018-0150
Mazel M, Jacot W, Pantel K, Bartkowiak K, Topart D, Cayrefourcq L, Rossille D, Maudelonde T, Fest T, Alix-Panabieres C (2015) Frequent expression of PD-L1 on circulating breast cancer cells. Mol Oncol 9(9):1773–1782. https://doi.org/10.1016/j.molonc.2015.05.009
Ilie M, Szafer-Glusman E, Hofman V, Chamorey E, Lalvee S, Selva E, Leroy S, Marquette CH, Kowanetz M, Hedge P, Punnoose E, Hofman P (2018) Detection of PD-L1 in circulating tumor cells and white blood cells from patients with advanced non-small-cell lung cancer. Ann Oncol 29(1):193–199. https://doi.org/10.1093/annonc/mdx636
Anantharaman A, Friedlander T, Lu D, Krupa R, Premasekharan G, Hough J, Edwards M, Paz R, Lindquist K, Graf R, Jendrisak A, Louw J, Dugan L, Baird S, Wang Y, Dittamore R, Paris PL (2016) Programmed death-ligand 1 (PD-L1) characterization of circulating tumor cells (CTCs) in muscle invasive and metastatic bladder cancer patients. BMC Cancer 16(1):744. https://doi.org/10.1186/s12885-016-2758-3
Jahr S, Hentze H, Englisch S, Hardt D, Fackelmayer FO, Hesch RD, Knippers R (2001) DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res 61(4):1659–1665
Ellinger J, Muller DC, Muller SC, Hauser S, Heukamp LC, von Ruecker A, Bastian PJ, Walgenbach-Brunagel G (2012) Circulating mitochondrial DNA in serum: a universal diagnostic biomarker for patients with urological malignancies. Urol Oncol 30(4):509–515. https://doi.org/10.1016/j.urolonc.2010.03.004
Vandekerkhove G, Todenhofer T, Annala M, Struss WJ, Wong A, Beja K, Ritch E, Brahmbhatt S, Volik SV, Hennenlotter J, Nykter M, Chi KN, North S, Stenzl A, Collins CC, Eigl BJ, Black PC, Wyatt AW (2017) Circulating tumor DNA reveals clinically actionable somatic genome of metastatic bladder cancer. Clin Cancer Res 23(21):6487–6497. https://doi.org/10.1158/1078-0432.ccr-17-1140
Roth C, Pantel K, Müller V, Rack B, Janni W, Schwarzenbach H (2010) Apoptosis-related deregulation of proteolytic activities and high serum levels of circulating nucleosomes and DNA in blood correlate with breast cancer progression. BMC Cancer 11(1):4
Gezer U, Holdenrieder S (2014) Post-translational histone modifications in circulating nucleosomes as new biomarkers in colorectal cancer. Vivo 28(3):287–292
Todenhofer T, Struss WJ, Seiler R, Wyatt AW, Black PC (2018) Liquid biopsy-analysis of circulating tumor DNA (ctDNA) in bladder cancer. Bladder Cancer 4(1):19–29. https://doi.org/10.3233/blc-170140
von Knobloch R, Hegele A, Brandt H, Olbert P, Heidenreich A, Hofmann R (2001) Serum DNA and urine DNA alterations of urinary transitional cell bladder carcinoma detected by fluorescent microsatellite analysis. Int J Cancer 94(1):67–72. https://doi.org/10.1002/ijc.1434
Utting M, Werner W, Dahse R, Schubert J, Junker K (2002) Microsatellite analysis of free tumor DNA in urine, serum, and plasma of patients: a minimally invasive method for the detection of bladder cancer. Clin Cancer Res 8(1):35–40
Dahse R, Utting M, Werner W, Schimmel B, Claussen U, Junker K (2002) TP53 alterations as a potential diagnostic marker in superficial bladder carcinoma and in patients serum, plasma and urine samples. Int J Oncol 20(1):107–115
Dominguez G, Carballido J, Silva J, Silva JM, Garcia JM, Menendez J, Provencio M, Espana P, Bonilla F (2002) p14ARF promoter hypermethylation in plasma DNA as an indicator of disease recurrence in bladder cancer patients. Clin Cancer Res 8(4):980–985
Ellinger J, El Kassem N, Heukamp LC, Matthews S, Cubukluoz F, Kahl P, Perabo FG, Muller SC, von Ruecker A, Bastian PJ (2008) Hypermethylation of cell-free serum DNA indicates worse outcome in patients with bladder cancer. J Urol 179(1):346–352. https://doi.org/10.1016/j.juro.2007.08.091
Jablonowski Z, Reszka E, Gromadzinska J, Wasowicz W, Sosnowski M (2011) Hypermethylation of p16 and DAPK promoter gene regions in patients with non-invasive urinary bladder cancer. Arch Med Sci 7(3):512–516. https://doi.org/10.5114/aoms.2011.23421
Valenzuela MT, Galisteo R, Zuluaga A, Villalobos M, Nunez MI, Oliver FJ, de Ruiz Almodovar JM (2002) Assessing the use of p16(INK4a) promoter gene methylation in serum for detection of bladder cancer. Eur Urol 42(6):622–628 (discussion 628–630)
Lin YL, Sun G, Liu XQ, Li WP, Ma JG (2011) Clinical significance of CDH13 promoter methylation in serum samples from patients with bladder transitional cell carcinoma. J Int Med Res 39(1):179–186. https://doi.org/10.1177/147323001103900119
Luo ZG, Li ZG, Gui SL, Chi BJ, Ma JG (2014) Protocadherin-17 promoter methylation in serum-derived DNA is associated with poor prognosis of bladder cancer. J Int Med Res 42(1):35–41. https://doi.org/10.1177/0300060513504705
Ellinger J, Bastian PJ, Ellinger N, Kahl P, Perabo FG, Buttner R, Muller SC, Ruecker A (2008) Apoptotic DNA fragments in serum of patients with muscle invasive bladder cancer: a prognostic entity. Cancer Lett 264(2):274–280. https://doi.org/10.1016/j.canlet.2008.01.038
Grivas P, Nagy RJ, Pond GR, Gupta S, Wang J, Vaishampayan UN, Pal SK, Bilen MA, Naik G, Ghatalia P, Hoimes CJ, Gopalakrishnan D, Barata P, Lanman RB, Talasaz AA (2017) Circulating tumor (ct)-DNA alterations in urothelial/bladder cancer (UC/BC): updates on a dynamic genomic landscape. J Clin Oncol 35(15_suppl):4534
Christensen E, Birkenkamp-Demtroder K, Nordentoft I, Hoyer S, van der Keur K, van Kessel K, Zwarthoff E, Agerbaek M, Orntoft TF, Jensen JB, Dyrskjot L (2017) Liquid biopsy analysis of FGFR3 and PIK3CA hotspot mutations for disease surveillance in bladder cancer. Eur Urol 71(6):961–969. https://doi.org/10.1016/j.eururo.2016.12.016
Patel KM, van der Vos KE, Smith CG, Mouliere F, Tsui D, Morris J, Chandrananda D, Marass F, van den Broek D, Neal DE, Gnanapragasam VJ, Forshew T, van Rhijn BW, Massie CE, Rosenfeld N, van der Heijden MS (2017) Association of plasma and urinary mutant DNA with clinical outcomes in muscle invasive bladder cancer. Sci Rep 7(1):5554. https://doi.org/10.1038/s41598-017-05623-3
Soave A, Schwarzenbach H, Vetterlein M, Rührup J, Engel O, Dahlem R, Fisch M, Pantel K, Riethdorf S, Rink M (2018) Detection and oncological impact of circulating tumor cells in bladder cancer patients with presence of copy number variations of circulating cell free DNA. J Clin Oncol 36(6_suppl):495
Birkenkamp-Demtroder K, Christensen E, Nordentoft I, Knudsen M, Taber A, Hoyer S, Lamy P, Agerbaek M, Jensen JB, Dyrskjot L (2018) Monitoring treatment response and metastatic relapse in advanced bladder cancer by liquid biopsy analysis. Eur Urol 73(4):535–540. https://doi.org/10.1016/j.eururo.2017.09.011
Agarwal N, Pal SK, Hahn AW, Nussenzveig RH, Pond GR, Gupta SV, Wang J, Bilen MA, Naik G, Ghatalia P, Hoimes CJ, Gopalakrishnan D, Barata PC, Drakaki A, Faltas BM, Kiedrowski LA, Lanman RB, Nagy RJ, Vogelzang NJ, Boucher KM, Vaishampayan UN, Sonpavde G, Grivas P (2018) Characterization of metastatic urothelial carcinoma via comprehensive genomic profiling of circulating tumor DNA. Cancer 124(10):2115–2124. https://doi.org/10.1002/cncr.31314
Chalfin HJ, Kates M, van der Toom EE, Glavaris S, Verdone JE, Hahn NM, Pienta KJ, Bivalacqua TJ, Gorin MA (2018) Characterization of urothelial cancer circulating tumor cells with a novel selection-free method. Urology. https://doi.org/10.1016/j.urology.2018.01.036
Small AC, Gong Y, Oh WK, Hall SJ, van Rijn CJM, Galsky MD (2012) The Emerging Role of Circulating Tumor Cell Detection in Genitourinary Cancer. J Urol 188(1):21–26. https://doi.org/10.1016/j.juro.2012.02.2558
Grunt M, Hillebrand T, Schwarzenbach H (2018) Clinical relevance of size selection of circulating DNA. Transl Cancer Res 7(Suppl 2):171–184
Schwarzenbach H (2014) The potential of circulating nucleic acids as components of companion diagnostics for predicting and monitoring chemotherapy response. Expert Rev Mol Diagn 15(2):267–275. https://doi.org/10.1586/14737159.2015.980817
Rijnders M, de Wit R, Boormans JL, Lolkema MPJ, van der Veldt AAM (2017) Systematic review of immune checkpoint inhibition in urological cancers. Eur Urol 72(3):411–423. https://doi.org/10.1016/j.eururo.2017.06.012
Desgrandchamps F, Teren M, Dal Cortivo L, Marolleau J, Bertheau P, Villette J, Cortesse A, Teillac P, Le Duc A, Hamdy F (1999) The effects of transurethral resection and cystoprostatectomy on dissemination of epithelial cells in the circulation of patients with bladder cancer. Br J Cancer 81:832–834
Kinjo M, Okegawa T, Horie S, Nutahara K, Higashihara E (2004) Detection of circulating MUC7-positive cells by reverse transcription-polymerase chain reaction in bladder cancer patients. Int J Urol 11:38–43
Pagliarulo V, Ancona P, Petitti T, Salerno A, Spadavecchia R, Di Stasi S, Cormio L, Maurizio B, Carrieri G, Ditonno P (2017) Detection and clinical significance of circulating tumor cells in patients undergoing radical cystectomy for urothelial bladder cancer. Clin Genitourin Cancer 15(4):455–462. https://doi.org/10.1016/j.clgc.2016.11.005
Soria J, Morat L, Durdux C, Housset M, Cortez A, Blaise R, Sabatier L (2002) The molecular detection of circulating tumor cells in bladder cancer using telomerase activity. J Urol 167:352–356
Gorgannezhad L, Umer M, Islam MN, Nguyen NT, Shiddiky MJA (2018) Circulating tumor DNA and liquid biopsy: opportunities, challenges, and recent advances in detection technologies. Lab Chip. 18(8):1174–1196
Author information
Authors and Affiliations
Contributions
MR: project development, data analysis, manuscript writing, and manuscript editing. HS: data collection, manuscript writing, and manuscript editing. SR: data collection, manuscript writing, and manuscript editing. AS: data collection, manuscript writing, and manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Michael Rink received honoraria by BMS and Pfizer. Dr. Michael Rink is investigator for the following companies and receives research grants by the following companies: Pfizer, MSD. All other authors have nothing to disclose.
Informed consent
All authors consented to the final draft.
Rights and permissions
About this article
Cite this article
Rink, M., Schwarzenbach, H., Riethdorf, S. et al. The current role and future directions of circulating tumor cells and circulating tumor DNA in urothelial carcinoma of the bladder. World J Urol 37, 1785–1799 (2019). https://doi.org/10.1007/s00345-018-2543-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2543-9