Abstract
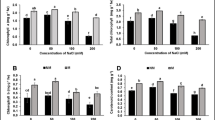
The present study investigated how contrasting citrus rootstocks, drought susceptible, Citrus jambhiri Lush cv. Jatti Khatti (JK) and drought-tolerant, X639 (C. reshni Hort. ex Tan. × Poncirous trifoliata), responded to water deficit and re-watering through reciprocal grafting. Self-graft JK showed the most declines in scion growth traits, the highest wilting score and drought injury index and a higher increase in leaf proline content compared to self-graft X639. JK/X639 witnessed a lesser decline in growth, the lowest wilting score and drought injury index among reciprocal grafts after experiencing water deficit as of normal moisture conditions. The plant combinations where X639 was used as rootstocks displayed good recovery in scion growth after re-watering. The root growth traits significantly improved in self X639/X639. The total phenol increased more in self-graft X639 and hetero-graft X639/JK after experiencing water deficit over respective normal moisture conditions. The X639, as scion or rootstock, was able to moderate lipid peroxidation significantly as compared to self-graft JK. Auto-graft X639 and hetero-graft X639/JK exhibited a higher decline in thiobarbituric acid reactive substances (TBARS) content, catalase, and glutathione reductase activities after re-watering. X639 rootstock prioritised growth, displaying the development of a vigorous root system under water scarcity, attributed to elevated relative water content, membrane stability index, and a robust antioxidant system in both self and reciprocal grafting. Conversely, JK focused on survival indicated by leaf shedding and reduced leaf area. These findings offer insights into citrus rootstock responses to water stress, valuable for citrus cultivation in regions with varying water availability.




Similar content being viewed by others
Data Availability
The data produced during this experiment are accessible through the corresponding author.
References
Aebi H (1984) Catalase in vitro. In: Methods in enzymology, vol 105, pp 121–126. Academic press
Álvarez S, Martín H, Barajas E, Rubio JA, Vivaldi GA (2020) Rootstock effects on water relations of young almond trees (cv. Soleta) when subjected to water stress and rehydration. Water. https://doi.org/10.3390/w12123319
Alves FM, Joshi M, Djidonou D, Joshi V, Gomes CN, Leskovar DI (2021) Physiological and biochemical responses of tomato plants grafted onto Solanum pennellii and Solanum peruvianum under water-deficit conditions. Plants. https://doi.org/10.3390/plants10112236
Balfagón D, Terán F, de Oliveira TDR, Santa-Catarina C, Gómez-Cadenas A (2021) Citrus rootstocks modify scion antioxidant system under drought and heat stress combination. Plant Cell Rep. https://doi.org/10.1007/s00299-021-02744-y
Balfagón D, Rambla JL, Granell A, Arbona V, Gomez-Cadenas A (2022) Grafting improves tolerance to combined drought and heat stresses by modifying metabolism in citrus scion. Environ Exp Bot. https://doi.org/10.1016/j.envexpbot.2022.104793
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39(1):205–207
Chen S, Yi L, Korpelainen H, Yu F, Liu M (2020) Roots play a key role in drought-tolerance of poplars as suggested by reciprocal grafting between male and female clones. Plant Physiol Biochem. https://doi.org/10.1016/j.plaphy.2020.05.014
da Silva MC, Pinto PI, Guerra R, Duarte A, Power DM, Marques NT (2023) Gene transcripts responsive to drought stress identified in Citrus macrophylla bark tissue transcriptome have a modified response in plants infected by Citrus tristeza virus. Sci Hortic. https://doi.org/10.1016/j.scienta.2022.111526
Deuschle K, Funck D, Hellmann H, Däschner K, Binder S, Frommer WB (2001) A nuclear gene encoding mitochondrial Δ1-pyrroline-5-carboxylate dehydrogenase and its potential role in protection from proline toxicity. Plant J. https://doi.org/10.1046/j.1365-313x.2001.01101.x
Dong T, Xi L, Xiong B, Qiu X, Huang S, Xu W, Wang J, Wang B, Yao Y, Duan C, Tang X (2021) Drought resistance in Harumi tangor seedlings grafted onto different rootstocks. Funct Plant Biol. https://doi.org/10.1071/FP20242
Dubey AK, Sharma RM (2016) Effect of rootstocks on tree growth, yield, quality and leaf mineral composition of lemon (Citrus limon. (L.) Burm). Sci Hortic. https://doi.org/10.1016/j.scienta.2016.01.013
Ejaz MK, Aurangzaib M, Iqbal R, Shahzaman M, Habib-ur-Rahman M, El-Sharnouby M, Datta R, Alzuaibr FM, Sakran MI, Ogbaga CC, Al Sabagh A (2022) The use of soil conditioners to ensure a sustainable wheat yield under water deficit conditions by enhancing the physiological and antioxidant potentials. Land 11(3):368
Engelbrecht BMJ, Tyree MT, Kursar TA (2007) Visual assessment of wilting as a measure of leaf water potential and seedling drought survival. J Trop Ecol. https://doi.org/10.1017/S026646740700421X
FAO (2023) Food and Agriculture Organizations of the United Nations. Accessed 7 August 2023 from http://www.fao.org/faostat/en/#data
Gonçalves LP, Alves TF, Martins CP, de Sousa AO, dos Santos IC, Pirovani CP, Almeida AAF, Filho MAC, Gesteira AS, Soares Filho WDS, Girardi EA (2016) Rootstock-induced physiological and biochemical mechanisms of drought tolerance in sweet orange. Acta Physiol Plant 38(7):174
Han Q, Guo Q, Korpelainen H, Niinemets Ü, Li C (2019) Rootstock determines the drought resistance of poplar grafting combinations. Tree Physiol. https://doi.org/10.1093/treephys/tpz102
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125(1):189–198
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57(12):1332–1334
Kadam DM, Dubey AK, Sharma RM, Morade A, Sharma N, Bhardwaj C (2022) Response of citrus (Citrus. sps) rootstock hybrids to PEG induced drought under hydroponic system. Indian J Agric Sci. https://doi.org/10.56093/ijas.v92i10.125062
Khoe LT, Dubey AK, Sharma RM, Awasthi OP, Meena MC, Mishra AK (2020) Citrus rootstock genotypes response to drought: Alternation in morphology, physiology and leaf mineral content. Indian J Hortic. https://doi.org/10.5958/0974-0112.2020.00032.8
Liu BH, Cheng L, Liang D, Zou YJ, Ma FW (2012) Growth, gas exchange, water-use efficiency, and carbon isotope composition of ‘Gale Gala’apple trees grafted onto 9 wild Chinese rootstocks in response to drought stress. Photosynthetica. https://doi.org/10.1007/s11099-012-0048-0
McAdam SA, Brodribb TJ, Ross JJ (2016) Shoot-derived abscisic acid promotes root growth. Plant Cell Environ. https://doi.org/10.1111/pce.12669
Morgan JA (1984) Interaction of water supply and N in wheat. Plant Physiol 76:112–117
Niu J, Zhang S, Liu S, Ma H, Chen J et al (2018) The compensation effects of physiology and yield in cotton after drought stress. J Plant Physiol. https://doi.org/10.1016/j.jplph.2018.03.001
Ozmen C, Baydu F, Hasanzadeh M, Oktem M, Babaoglu G, Kibar U, Khabbazi S, Kazan K, Ergül A (2023) Assessment of drought stress responsive genes expression profiiles and proline accumulation in a diverse set of grapevine rootstocks. Turk J Agric For. https://doi.org/10.55730/1300-011X.3089
Pedroso FK, Prudente DA, Bueno ACR, Machado EC, Ribeiro RV (2014) Drought tolerance in citrus trees is enhanced by rootstock-dependent changes in root growth and carbohydrate availability. Environ Exp Bot. https://doi.org/10.1016/j.envexpbot.2013.12.024
Premchandra GS, Saneoka H, Ogata S (1990) Cell membrane stability, an indicator of drought tolerance as affected by applied nitrogen in soybean. J Agric Sci Camb 115:63–66
Rezayian M, Niknam V, Ebrahimzadeh H (2018) Differential responses of phenolic compounds of Brassica napus under drought stress. Iran J Plant Physiol. https://doi.org/10.30495/IJPP.2018.540887
Robles JM, Botía P, Pérez-Pérez JG (2017) Sour orange rootstock increases water productivity in deficit irrigated ‘Verna’lemon trees compared with Citrus macrophylla. Agric Water Manag 186:98–107
Rodríguez-Gamir J, Primo-Millo E, Forner JB, Forner-Giner MA (2010) Citrus rootstock responses to water stress. Sci Hortic 126(2):95–102. https://doi.org/10.1016/j.scienta.2010.06.015
Sairam RK (1994) Effect of moisture stress on physiological activities of two contrasting wheat genotypes. Ind J Exp Biol 32:594–597
Sánchez-Rodríguez E, del Mar R-W, Blasco B, Leyva R, Romero L, Ruiz JM (2012) Antioxidant response resides in the shoot in reciprocal grafts of drought-tolerant and drought-sensitive cultivars in tomato under water stress. Plant Sci 188:89–96. https://doi.org/10.1016/j.plantsci.2011.12.019
Santana-Vieira DDS, Freschi L, da Hora Almeida LA, De Moraes DHS, Neves DM, dos Santos LM et al (2016) Survival strategies of citrus rootstocks subjected to drought. Sci Rep. https://doi.org/10.1038/srep38775
SAS (Statistical Analysis System) (2011) SAS version 9.3. procedure guide. SAS Inc., Cary
Silva VA, Prado FM, Antunes WC, Paiva RMC, Ferrão MAG, Andrade AC, Di Mascio P, Loureiro ME, DaMatta FM, Almeida AM (2018) Reciprocal grafting between clones with contrasting drought tolerance suggests a key role of abscisic acid in coffee acclimation to drought stress. Plant Growth Regul. https://doi.org/10.1007/s10725-018-0385-5
Silva SF, Miranda MT, Costa VE, Machado EC, Ribeiro RV (2021) Sink strength of citrus rootstocks under water deficit. Tree Physiol. https://doi.org/10.1093/treephys/tpab008
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5, 5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175(2):408–413
Varela MC, Arslan I, Reginato MA, Cenzano AM, Luna MV (2016) Phenolic compounds as indicators of drought resistance in shrubs from Patagonian shrublands (Argentina). Plant Physiol Biochem. https://doi.org/10.1016/j.plaphy.2016.03.014
Yang L, Xia L, Zeng Y, Han Q, Zhang S (2022) Grafting enhances plants drought resistance: current understanding, mechanisms, and future perspectives. Front Plant Sci 13:1015317
Yi-ling Y, Chun-hui H, Qing-qing G, Xue-yan Q, Xiao-biao X (2015) Evaluation of drought-resistance traits of citrus rootstock seedlings by multiple statistics analysis. Acta Hortic 1065(47):379–386
Zaher-Ara T, Boroomand N, Sadat-Hosseini M (2016) Physiological and morphological response to drought stress in seedlings of ten citrus. Trees. https://doi.org/10.1007/s00468-016-1372-y
Acknowledgements
We gratefully acknowledge the Director of ICAR-IARI, New Delhi for the support provided for executing the present research.
Funding
This research work received no external funding.
Author information
Authors and Affiliations
Contributions
Identified researchable areas supervision visualization, AKD, formal analysis, RMS and NS; investigation, LC; methodology, AKD, NS; supervision, AKD and RMS; writing original draft, LC; writing- AKD; review and editing, RMS, RP and LS. All authors read and provided their consent for the final version of the manuscript for publication.
Corresponding authors
Ethics declarations
Conflict of interest
No conflict of interest are present among the authors.
Ethical Approval
Not applicable.
Additional information
Handling Editor: Mikihisa Umehara.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chand, L., Sharma, N., Sharma, R.M. et al. Physio-Biochemical and Growth Response of Contrasting Reciprocal Grafting in Citrus Under Water Deficit and Rehydration. J Plant Growth Regul 43, 1230–1246 (2024). https://doi.org/10.1007/s00344-023-11179-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11179-6