Abstract
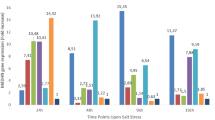
The present investigation aimed to determine the salt tolerance level of six already selected elite clones of Eucalyptus tereticornis. The response of individual shoot cultures was evaluated on basal MS medium supplemented with different concentrations of NaCl (0, 100, 200, 300, 400, and 500 mM) for 28 days. The increase in NaCl concentration affected the survival of microshoots along with inhibition in shoot and root growth in all the clones. A better performance of clone ‘KE8’ was evident with higher survival of microshoots (65.18%) in comparison to clones ‘CE2’ (36.29%), and ‘Y8’ (10.37%) on medium containing 500 mM NaCl. The highest salt tolerance index (0.30) was recorded for clone ‘KE8’, followed by 0.24 for clone ‘CE2’ and 0.10 for clone ‘Y8’ on medium containing 500 mM NaCl. The 50% growth inhibition dose of NaCl was also found to be 479 mM for clone ‘KE8’, as compared to 385 mM and 206 mM for clones ‘CE2’ and ‘Y8’, respectively. Various physiological and biochemical parameters were investigated in microshoots of putatively higher salt-tolerant clone ‘KE8’, moderately salt-tolerant clone ‘CE2’ and less salt-tolerant clone ‘Y8’ at different intervals of culture period (7, 14, 21, 28 days). The relative water content and pigment levels (chlorophyll and carotenoid) declined significantly during culture on medium containing 500 mM NaCl with increasing culture period. Furthermore, the decline was higher in the case of clone ‘Y8’ than ‘CE2’ followed by ‘KE8’. A significant increase in the activity of antioxidant enzymes (superoxide dismutase, catalase, and peroxidase), accumulation of proline and lower levels of malondialdehyde content were recorded in clone ‘KE8’ as compared to the other clones ‘CE2’ and ‘Y8’. The present study suggested that clone ‘KE8’ is more tolerant to salt stress, which can be commercially grown in saline soils.





Similar content being viewed by others
References
Abdelaziz M, Xuan T, Mekawy A, Wang H, Khanh T (2018) Relationship of salinity tolerance to Na+ exclusion, proline accumulation, and antioxidant enzyme activity in rice seedlings. Agriculture 8:166
AbdElgawad H, Zinta G, Hegab MM, Pandey R, Asard H, Abuelsoud W (2016) High salinity induces different oxidative stress and antioxidant responses in maize seedlings organs. Front Plant Sci 7:276
Acosta-Motos JR, Ortuño MF, Bernal-Vicente A, Diaz-Vivancos P, Sanchez-Blanco MJ, Hernandez JA (2017) Plant responses to salt stress: adaptive mechanisms. Agronomy 7:18
Aebi H (1984) Catalase in vitro. In: Methods in enzymology, vol 105. Elsevier, Amsterdam, pp 121–126
Aggarwal D, Kumar A, Reddy MS (2010) Shoot organogenesis in elite clones of Eucalyptus tereticornis. Plant Cell Tissue Organ Cult 102:45–52
Alam M, Juraimi AS, Rafii M, Abdul Hamid A (2015) Effect of salinity on biomass yield and physiological and stem-root anatomical characteristics of purslane (Portulaca oleracea L.) accessions. Biomed Res Int. https://doi.org/10.1155/2015/105695
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1–15
Ayala-Astorga GI, Alcaraz-Meléndez L (2010) Salinity effects on protein content, lipid peroxidation, pigments, and proline in Paulownia imperialis (Siebold & Zuccarini) and Paulownia fortunei (Seemann & Hemsley) grown in vitro. Electron J Biotechnol 13:13–14
Bai X, Dai L, Sun H, Chen M, Sun Y (2019) Effects of moderate soil salinity on osmotic adjustment and energy strategy in soybean under drought stress. Plant Physiol Biochem 139:307–313
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Cha-um S, Somsueb S, Samphumphuang T, Kirdmanee C (2013) Salt tolerant screening in eucalypt genotypes (Eucalyptus spp.) using photosynthetic abilities, proline accumulation, and growth characteristics as effective indices. In Vitro Cell Dev Biol Plant 49:611–619
Chakraborty S, Newton AC (2011) Climate change, plant diseases and food security: an overview. Plant Pathol 60:2–14
Chelli-Chaabouni A, Mosbah AB, Maalej M, Gargouri K, Gargouri-Bouzid R, Drira N (2010) In vitro salinity tolerance of two pistachio rootstocks: Pistacia vera L. and P. atlantica Desf. Environ Exp Bot 69:302–312
Cheng Y-E, Dong M-Y, Fan X-W, Nong L-L, Li Y-Z (2018) A study on cassava tolerance to and growth responses under salt stress. Environ Exp Bot 155:429–440
de Azevedo Neto AD, Prisco JT, Enéas-Filho J, de Abreu CEB, Gomes-Filho E (2006) Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ Exp Bot 56:87–94
de Morais MB, Barbosa-Neto AG, Willadino L, Ulisses C, Junior TC (2019) Salt stress induces increase in starch accumulation in duckweed (Lemna aequinoctialis, Lemnaceae): biochemical and physiological aspects. J Plant Growth Regul 38(2):683–700
Demirkiran A, Marakli S, Temel A, Gozukirmizi N (2013) Genetic and epigenetic effects of salinity on in vitro growth of barley. Genet Mol Biol 36:566–570
Genc Y, Mcdonald GK, Tester M (2007) Reassessment of tissue Na+ concentration as a criterion for salinity tolerance in bread wheat. Plant Cell Environ 30:1486–1498
Gondim FA, Gomes-Filho E, Costa JH, Alencar NLM, Prisco JT (2012) Catalase plays a key role in salt stress acclimation induced by hydrogen peroxide pretreatment in maize. Plant Physiol Biochem 56:62–71
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7:1456–1466
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hernández JA (2019) Salinity Tolerance in plants: trends and perspectives. Int J Mol Sci 20:2408
Hishida M, Ascencio-Valle F, Fujiyama H, Orduño-Cruz A, Endo T, Larrinaga-Mayoral J (2014) Antioxidant enzyme responses to salinity stress of Jatropha curcas and J. cinerea at seedling stage. Russ J Plant Physiol 61:53–62
Hossain Z, Mandal AKA, Datta SK, Biswas AK (2007) Development of NaCl-tolerant line in Chrysanthemum morifolium Ramat. through shoot organogenesis of selected callus line. J Biotechnol 129:658–667
India’s State of Forest Report (2019) Forest Survey of India, Ministry of Environment Forest and Climate Change, Government of India
Jamil A, Riaz S, Ashraf M, Foolad M (2011) Gene expression profiling of plants under salt stress. Crit Rev Plant Sci 30:435–458
Janků M, Luhová L, Petřivalský M (2019) On the origin and fate of reactive oxygen species in plant cell compartments. Antioxidants 8:105
Koca H, Bor M, Özdemir F, Türkan I (2007) The effect of salt stress on lipid peroxidation, antioxidative enzymes and proline content of sesame cultivars. Environ Exp Bot 60:344–351
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Machado R, Serralheiro R (2017) Soil salinity: effect on vegetable crop growth. Management practices to prevent and mitigate soil salinization. Horticulturae 3:30
McEwan A, Marchi E, Spinelli R, Brink M (2019) Past, present and future of industrial plantation forestry and implication on future timber harvesting technology. J For Res 31:339–351
McEwen Jr CM (1971) Monoamine oxidase [rabbit serum]. In: Methods in enzymology, vol 17. Elsevier, Amsterdam, pp 686-692
Mhamdi A, Queval G, Chaouch S, Vanderauwera S, Van Breusegem F, Noctor G (2010) Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models. J Exp Bot 61:4197–4220
Mousavi S et al (2019) Physiological, epigenetic and genetic regulation in some olive cultivars under salt stress. Sci Rep 9:1–17
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Mustafa G, Akhtar MS (2019) Crops and methods to control soil salinity. In: Salt stress, microbes, and plant interactions: mechanisms and molecular approaches. Springer, Singapore, pp 237–251
Niknam V, Bagherzadeh M, Ebrahimzadeh H, Sokhansanj A (2004) Effect of NaCl on biomass and contents of sugars, proline and proteins in seedlings and leaf explants of Nicotiana tabacum grown in vitro. Biol Plant 48:613–615
Oyiga BC, Sharma R, Shen J, Baum M, Ogbonnaya F, Léon J, Ballvora A (2016) Identification and characterization of salt tolerance of wheat germplasm using a multivariable screening approach. J Agron Crop Sci 202:472–485
Passamani LZ et al (2017) Salt stress induces changes in the proteomic profile of micropropagated sugarcane shoots. PLoS ONE 12:e0176076
Patel BB, Patel BB, Dave RS (2011) Studies on infiltration of saline-alkali soils of several parts of Mehsana and Patan districts of North Gujarat. J Appl Technol Environ Sanitation 1:87–92
Radanielson AM, Angeles O, Li T, Ismail AM, Gaydon DS (2018) Describing the physiological responses of different rice genotypes to salt stress using sigmoid and piecewise linear functions. Field Crops Res 220:46–56
Rahnama H, Ebrahimzadeh H (2005) The effect of NaCl on antioxidant enzyme activities in potato seedlings. Biol Plant 49:93–97
Rahneshan Z, Nasibi F, Moghadam AA (2018) Effects of salinity stress on some growth, physiological, biochemical parameters and nutrients in two pistachio (Pistacia vera L.) rootstocks. J Plant Interact 13:73–82
Rai MK, Kalia RK, Singh R, Gangola MP, Dhawan A (2011) Developing stress tolerant plants through in vitro selection—an overview of the recent progress. Environ Exp Bot 71:89–98
Ramage MH et al (2017) The wood from the trees: the use of timber in construction. Renew Sust Energ Rev 68:333–359
Sharma DK, Singh A (2017) Current trends and emerging challenges in sustainable management of salt-affected soils: a critical appraisal. In: Bioremediation of salt affected soils: an Indian perspective. Springer, Cham, pp 1–40
Singh D, Kaur S, Kumar A (2020) In vitro drought tolerance in selected elite clones of Eucalyptus tereticornis Sm. Acta Physiol Plant 42:17
Sixto H, Grau J, Alba N, Alia R (2005) Response to sodium chloride in different species and clones of genus Populus L. Forestry 78:93–104
Smart RE, Bingham GE (1974) Rapid estimates of relative water content. Plant Physiol 53:258–260
Tavakkoli E, Rengasamy P, McDonald GK (2010) High concentrations of Na+ and Cl– ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J Exp Bot 61:4449–4459
Vijayan K, Chakraborti S, Ghosh P (2003) In vitro screening of mulberry (Morus spp.) for salinity tolerance. Plant Cell Rep 22:350–357
Woodward AJ, Bennett IJ (2005) The effect of salt stress and abscisic acid on proline production, chlorophyll content and growth of in vitro propagated shoots of Eucalyptus camaldulensis. Plant Cell Tissue Organ Cult 82:189–200
Zhang L, Ma H, Chen T, Pen J, Yu S, Zhao X (2014) Morphological and physiological responses of cotton (Gossypium hirsutum L.) plants to salinity. PLoS ONE 9:112807
Acknowledgements
DS is thankful to Thapar Institute of Engineering and Technology, Patiala for providing financial support in the form of Teaching Associateship. The authors would like to acknowledge TIFAC-CORE for providing necessary facilities for the work.
Author information
Authors and Affiliations
Contributions
DS conducted the experiments, analyzed, compiled the data and wrote the initial manuscript. AK helped in planning the experiments and finalizing the manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, D., Kumar, A. In Vitro Screening and Characterization of Selected Elite Clones of Eucalyptus tereticornis Sm. for Salt Stress. J Plant Growth Regul 40, 694–706 (2021). https://doi.org/10.1007/s00344-020-10138-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-020-10138-9