Abstract
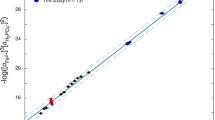
The Henry’s Law constant (k) for phosphine in seawater was determined by multiple phase equilibration combined with headspace gas chromatography. The effects of pH, temperature, and salinity on k were studied. The k value for phosphine in natural seawater was 6.415 at room temperature (approximately 23°C). This value increases with increases in temperature and salinity, but no obvious change was observed at different pH levels. At the same temperature, there was no significant difference between the k for phosphine in natural seawater and that in artificial seawater. This implies that temperature and salinity are major determining factors for k in marine environment. Double linear regression with Henry’s Law constants for phosphine as a function of temperature and salinity confirmed our observations. These results provide a basis for the measurement of trace phosphine concentrations in seawater, and will be helpful for future research on the status of phosphine in the oceanic biogeochemical cycle of phosphorus.
Similar content being viewed by others
References
Devai I, DeLaune R D, Devai G, Patrick W H, Czegeny I. 1999. Phosphine production potential of various wastewater and sewage sludge sources. Anal. Lett., 32(7): 1 447–1 457.
Devai I, Delaune R D. 1995. Evidence for phosphine production and emission from louisiana and florida marsh soils. Org. Geochem., 23(3): 277–279.
Devai I, Felfoldy L, Wittner I, Plosz S. 1988. Detection of phosphine-new aspects of the phosphorus cycle in the hydrosphere. Nature, 333(6171): 343–345.
Dewulf J, Drijvers D, Vanlangenhove H. 1995. Measurement of henrys law constant as function of temperature and salinity for the low-temperature range. Atmos. Environ., 29(3): 323–331.
Elkins J W. 1980. Determination of dissolved nitrous oxide in aquatic systems by gas chromatography using electroncapture detection and multiple phase equilibration. Analytical Chemistry, 52(2): 263–267.
Feng Z H, Song X X, Yu Z M. 2008a. Distribution characteristics of matrix-bound phosphine along the coast of China and possible environmental controls. Chemosphere, 73(4): 519–525.
Feng Z H, Song X X, Yu Z M. 2008b. Seasonal and spatial distribution of matrix-bound phosphine and its relationship with the environment in the Changjiang River Estuary, China. Marine Pollution Bulletin, 56(9): 1 630–1 636.
Frank R, Rippen G. 1987. Verhalten von phosphin in der Atmosphäre (Fate of phosphine in the atmosphere). Lebensmitteltechnik, 17: 409–411.
Gassmann G, van Beusekom J E E, Glindemann D. 1996. Offshore atmospheric phosphine. Naturwissenschaften, 83(3): 129–131.
Gassmann G. 1994. Phosphine in the fluvial and marine hydrosphere. Mar. Chem., 45(3): 197–205.
Geng J J, Niu X J, Wang X R, Edwards M, Glindemann D. 2010. The presence of trace phosphine in Lake Taihu water. Int. J. Environ. Anal. Chem., 90(9): 737–746.
Glindemann D, Bergmann A, Stottmeister U, Gassmann G. 1996. Phosphine in the lower terrestrial troposphere. Naturwissenschaften, 83(3): 131–133.
Glindemann D, Edwards M, Kuschk P. 2003. Phosphine gas in the upper troposphere. Atmos. Environ., 37(18): 2 429–2 433.
Glindemann D, Eismann F, Bergmann A, Kuschk P, Stottmeister U. 1998. Phosphine by bio-corrosion of phosphide-rich iron. Environ. Sci. Pollut. Res., 5(2): 71–74.
Hanrahan G, Salmassi T M, Khachikian C S, Foster K L. 2005. Reduced inorganic phosphorus in the natural environment: significance, speciation and determination. Talanta, 66(2): 435–444.
Lau K, Rogers T N, Chesney D J. 2010. Measuring the aqueous Henry’s Law constant at elevated temperatures using an extended EPICS technique. J. Chem. Eng. Data, 55(11): 5 144–5 148.
Li J B, Zhang G L, Zhang J, Liu S M, Ren J L, Hou Z X. 2009. Phosphine in the lower atmosphere of Qingdao-a coastal site of the Yellow Sea (China). Water Air and Soil Pollution, 204(1–4): 117–131.
Liu J A, Yahui C H Z, Kuschk P, Eismann F, Glindemann D. 1999. Phosphine in the urban air of Beijing and its possible sources. Water Air and Soil Pollution, 116(3–4): 597–604.
McAullif C. 1971. GC determination of solutes by multiple phase equilibration. Chemical Technology, (Jan): 46–51.
Mohebbi V, Naderifar A, Behbahani R M, Moshfeghian M. 2012. Determination of Henry’s law constant of light hydrocarbon gases at low temperatures. J. Chem. Thermodyn., 51: 8–11.
Niu X J, Geng J J, Wang X R, Wang C H, Gu X H, Edwards M, Glindemann D. 2004. Temporal and spatial distributions of phosphine in Taihu Lake, China. Science of the Total Environment, 323(1–3): 169–178.
Rice C P, Chernyak S M, McConnell L L. 1997. Henry’s law constants for pesticides measured as a function of temperature and salinity. J. Agr. Food Chem., 45(6): 2 291–2 298.
Roels J, Verstraete W. 2004. Occurrence and origin of phosphine in landfill gas. Science of the Total Environment, 327(1–3): 185–196.
Rolf S. 1999. Compilation of Henry’s Law Constants for Inorganic and Organic Species of Potential Importance in Environmental Chemistry(Version 3). http://www.mpchmainz.mpg.de/-sander/res/henry.html. Accessed on 2011-6-28.
Ruiz-Bevia F, Fernandez-Torres M J. 2010. Determining the Henry’s Law constants of THMs in seawater by means of purge-and-trap gas chromatography (PT-GC): the influence of seawater as sample matrix. Anal. Sci., 26(6): 723–726.
Song X X, Morrison R J, Feng Z H, Liu D Y, Harrison J J, Yu Z M. 2011. Matrix-bound phosphine in sediments from Lake Illawarra, New South Wales, Australia. Marine Pollution Bulletin, 62(8): 1 744–1 750.
Wang Y H, Wang B Q. 2001. Determination of Henry’s law constants for dimethyl sulfide in seawater. Chinese Journal of Chromatography, 19(4): 358–360. (in Chinese with English abstract)
Wilhelm E, Battino R, Wilcock R J. 1977. Low-pressure solubility of gases in liquid water. Chemical Reviews, 77(2): 219–262.
Yu Z M, Song X X. 2003. Matrix-bound phosphine: a new form of phosphorus found in sediment of Jiaozhou Bay. Chinese Science Bulletin, 48(1): 31–35.
Zhu R B, Glindemann D, Kong D M, Sun L G, Geng J J, Wang X R. 2007. Phosphine in the marine atmosphere along a hemispheric course from China to Antarctica. Atmos. Environ., 41(7): 1 567–1 573.
Zhu R B, Kong D M, Sun L G, Geng J J, Wang X R, Glindemann D. 2006. Tropospheric phosphine and its sources in coastal Antarctica. Environ. Sci. Technol., 40(24): 7 656–7 661.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Nos. 30970522, 40576058) and the National Natural Science Foundation of China for Creative Research Groups (No. 41121064)
Rights and permissions
About this article
Cite this article
Fu, M., Yu, Z., Lu, G. et al. Henry’s law constant for phosphine in seawater: determination and assessment of influencing factors. Chin. J. Ocean. Limnol. 31, 860–866 (2013). https://doi.org/10.1007/s00343-013-2212-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-013-2212-1