Abstract
Research on Mesophotic Coral Ecosystems (MCEs; 30–150 m) has grown exponentially in the last few decades, highlighting their rich diversity and extensive distribution. However, they are still largely underexplored compared to shallow-water coral reefs and frequently remain under-protected and under-represented in marine spatial planning. One reason for the imbalance between the high ecological value of MCEs and the limited levels of protection may be that baseline data on MCEs are largely missing to date, yet are crucial to provide evidence-based information for management actions. Here, we present data on the alpha and beta diversity of the benthic communities within MCEs in the Chagos Archipelago, Indian Ocean. Using imagery collected from Remotely Operated Vehicle surveys, benthic invertebrate megafauna were surveyed along the entire depth gradient from shallow to lower mesophotic depths (15–160 m). The diversity of the benthic communities decreased with increasing depth, from shallow water to the lower mesophotic zone. Nevertheless, the deepest parts of MCEs in the Archipelago displayed higher species richness values than several other shallow subtropical regions. In addition, the benthic communities showed high dissimilarity along the depth gradient, indicating that the key driver of community composition change with depth is species turnover (species replacement), revealing the uniqueness of MCEs. This study presents novel findings on MCEs in the central Indian Ocean, demonstrating that they host a high and unique benthic diversity, and highlighting the need to protect these ecosystems to preserve the overall biodiversity of coral reefs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mesophotic coral ecosystems (MCEs; light-dependent coral ecosystems found at depths between 30 to 150 m (Hinderstein et al. 2010; Kahng et al. 2014)), have received increasing attention from the scientific community over the past few decades. However, they are still largely underexplored compared to their shallow-water counterparts (Bongaerts and Smith 2019; Pyle and Copus 2019; Soares et al. 2020; Stefanoudis et al. 2022). The increasing interest in MCEs was initially driven by the potential refuge MCEs could offer to threatened shallow coral reefs, the so-called deep reef refugia hypothesis (DRRH) (Glynn 1996), and the evolution of technology, allowing exploration of these deeper, cooler waters (Bongaerts et al. 2010). More recently, the interest has been driven by the desire to investigate the distinct biological and environmental characteristics of MCEs compared to shallow reefs, and the functional role that MCEs play in coral reef ecosystems (Kramer et al. 2020; Carpenter et al. 2022; Pérez-Rosales, et al. 2022a).
Recent research has revealed that MCEs are more biodiverse and widespread than previously expected, with 80% of all coral reef habitat falling within MCEs (based on depth alone) and they are an important component for maintaining overall reef biodiversity (Englebert et al. 2017; Shoham and Benayahu 2017; Muir and Pichon 2019; Pyle and Copus 2019; Pérez-Rosales et al. 2022). Yet the protection status of MCEs remains poor (Hernandez‐Agreda et al. 2022). In fact, despite meeting the international criteria used to identify priority areas for conservation policies, and hence being good candidates for future Marine Protected Area (MPA) designation (Soares et al. 2020), MCEs and deeper reefs (i.e. rariphotic zone 150–300 m, sensu Baldwin et al. (2018) are considered under-protected and under-represented in marine spatial planning (Rocha et al. 2018; Stefanoudis et al. 2022). Indeed, the few protected mesophotic and deeper reefs were often included incidentally due to geopolitical boundaries (e.g., a country protects its entire Exclusive Economic Zone (EEZ) and MCEs happen to fall within this boundary), rather than purposely (Soares et al. 2019; Turner et al. 2019; Stefanoudis et al. 2022). Several reviews suggest that MCEs are among the least protected ecosystems of the world’s oceans, while considered as the richest in terms of endemic and rare species (Hinderstein et al. 2010; Bridge and Guinotte 2013; Loya et al. 2016; Turner et al. 2019). This may partly be due to the lack of an holistic understanding of the patterns and drivers of diversity with depth, although the nature of spatial patterns of biodiversity has been one of the fundamental goals of ecological research for centuries (Lesser et al. 2019), and biodiversity data are crucial to provide evidence-based information for management and conservation actions for MCEs (Socolar et al. 2016).
Diversity can be described at different scales. Alpha diversity identifies diversity at a local scale, while beta diversity reveals the variation in species between communities. More specifically, alpha diversity (sensu Whittaker (1960)) measures the number of species within a community, and beta diversity (sensu Whittaker (1972)) defines the variation in species composition across a gradient, e.g. along the depth gradient or between communities at different sites (Miyazawa et al. 2020). Beta diversity can be partitioned into two components: nestedness, i.e. when the species composition of the sample is a subset of a richer community, hence indicating a species loss; and species turnover, i.e. species replacement between communities, demonstrating the uniqueness of the communities within an ecosystem (Baselga 2010). These two components allow for a quantitative understanding of the variation in biodiversity gradients and the assessment of community change across environmental gradients (Pérez-Rosales, et al. 2022a). To date, studies of alpha and beta diversity have mainly focussed on terrestrial areas (Wolters et al. 2006), or shallow-water environments (McDevitt-Irwin et al. 2021). Within MCEs, where the calculation of alpha and beta diversity indices remains scarce, the majority of studies has investigated fishes or scleractinian corals (Rocha et al. 2018; Pérez-Rosales, et al. 2022b), with little consideration of the diversity of the overall benthic community. Yet, research has shown the importance of assessing diversity within communities as a whole, rather than within single taxonomic groups, as species interactions may influence patterns of diversity (McDevitt-Irwin et al. 2021).
The aim of the present study is to investigate patterns of diversity within MCEs over the depth gradient (15 m to 160 m) and between sites in the Chagos Archipelago, located in a particularly understudied region, the central Indian Ocean. The quantification of alpha and beta diversity indices will provide fundamental data on the patterns of species diversity and potential taxonomic overlap among communities over the depth gradient, and between shallow-water reefs and MCEs. Such data are essential to understand the role of MCEs in the functioning of coral reef ecosystems, ii. unravel the ecological contribution of MCEs to the coral reef system as a whole; and iii. develop conservation and management plans for these ecosystems.
Methods
Study area
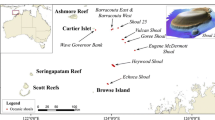
This study was conducted in the Chagos Archipelago (6° S, 71°30’ E), central Indian Ocean. The Archipelago is a fully no-take MPA established in 2010, covering 640,000 km2 and incorporating shallow coral reefs, seagrass meadows, submerged banks and seamounts (Hays et al. 2020). The Archipelago has been uninhabited since the 1970s (with the exception of one atoll with strict environmental regulations), therefore human disturbance from pollution, agriculture and reef fisheries is minimal (Head et al. 2019; Hays et al. 2020). In this study, MCEs were investigated at two oceanographically contrasting sites around Egmont Atoll (6° 40’ S, 71 21′ E; Fig. 1): Ile Des Rats (IDR, North-West coast) open to the wider ocean and Manta Alley (MA, North-East coast) somewhat sheltered from the Great Chagos Bank (Fig. 1). Both sites occur on steep slopes with rocky outcrops and strong currents prevailing from east to west and west to east, depending on the movement of the tide.
Location of study sites within the Chagos Archipelago. A: Chagos Archipelago with depth gradients extracted from GEBCO data between 10 and 200 m depth. B: central Indian Ocean with the Chagos Marine Protected Area delimited in dark pink. C: Egmont Atoll displaying bathymetry data acquired using multibeam (between 10 and 200 m depth) and the two study sites: Ile Des Rats (teal diamond) and Manta Alley (orange diamond)
Biological data
A Remotely Operated Vehicle (ROV, Saab Seaeye Falcon model) with a GoPro Hero 4 (2.7k, 24 fps, wide field of view) attached to it was used to collect benthic imagery from MCEs at the two study sites, IDR and MA. An information overlay on the ROV SD camera displayed time, depth, heading, pitch and roll. Full ROV specifications and details of the biological data collection are provided in detail in Diaz et al., (2023a), but briefly, HD still images were extracted from the GoPro video transects when the ROV approached the seabed and was positioned at an oblique angle (altitude of <1.5 m). The presence of a strong current at both sites, precluded linear benthic video transects.
The entire MCE depth gradient was surveyed, with 6 depth zones chosen a priori to the study based on the literature, covering the shallow reef and the lower mesophotic zones: 15–20 m; 30–40 m; 60–70 m; 80–90 m; 110–120 m; 150–160 m. Three transects were surveyed per depth zone (average transect duration = 30 mins 3 s ± 6 mins 58 s), parallel to the slope at each of the two study sites. Ninety images were collected per depth zone (n=30 images/transect) with a total of 1,080 images from the two study sites. All data were collected during research expeditions in November 2019 (transects 1, 2, 3 for IDR; transect 1 for MA) and March 2020 (transects 2 and 3 for MA).
Still images collected along each transect were manually annotated by a single observer using BIIGLE 2.0, an online annotation platform (Langenkämper et al. 2017), which enables identification and enumeration of organisms. The image analysis procedures are provided in detail in Diaz et al., (2023a). All specimens were identified according to both relevant literature and taxonomist’s support (Tables S1 and acknowledgements). During the study, a morphospecies catalogue was created for the region (Diaz et al. 2023b) following the global standardised marine taxon reference image database created by Howell et al., (2019); a total of 582 OTUs were identified in this study. Only one observer annotated the images to reduce the annotation bias. In addition, quality assurance was carried out by re-annotating 5% of the images by the same observer, as recommended in Schoening et al. (2016). A summary of the total number of morphospecies and individuals per site and depth zone are shown in Table S2.
The ROV was not equipped with paired lasers during the surveys, thus calculation of the field of view within each image was not possible. To standardise data collection, the pilots positioned the ROV at a similar distance from the seabed, with the same angle of view, for each image collected. The mean time taken to complete a single transect was 30 min 3 s (± 6 min 58 s), with no significant differences observed in the time taken to complete a transect with depth zone and site (Kruskal–Wallis test, p-value > 0.05. Transect by depth zone: chi-squared = 10.244, df = 5, p-value = 0.069; transect by site: chi-squared = 0.0022733, df = 1, p-value = 0.962). Thus, given the consistency in the speed of the ROV, it can be assumed a comparable area of seabed was surveyed within each transect and depth zone. To enable comparisons between sites and depth zones, this study focuses on the number of images surveyed and counts of benthic organisms rather than an area-based analysis. Furthermore, as suggested by Colwell et al. (2012) and Roswell et al. (2021), data were standardised by individuals and sampling coverage per depth zone using the iNext package in R software (Hsieh, Ma and Chao, 2020) (see next section).
Statistical analyses
To assess the variation in alpha and beta diversity of MCEs over the depth gradient and between the study sites, data were analysed using diversity indices with the iNEXT (iNterpolation/EXTrapolation) (Hsieh, Ma and Chao, 2020) and betapart packages (Baselga and Orme 2012) in R software (Team R Core 2020).
Alpha diversity analyses
To allow comparisons among sites and depth zones, despite possible differences between survey areas covered, a combination of asymptotic and non-asymptotic diversity estimations was applied, based on rarefaction and extrapolation analyses and Hill numbers, using the iNext package in R. Individual-based rarefaction and extrapolation (R/E) curves, as well as sample coverage-based R/E curves, were plotted, with non-overlapping 95% confidence intervals interpreted as a significant difference, as per García-Martínez, et al., (2017) and Cumming et al. (2007). Individual-based R/E curves were used as they provide unbiased estimates of species diversity and enable comparison of morphospecies diversity between depth zones and sites, specifically when they reach an asymptote.
The protocol proposed by Chao et al. (2020) was followed to assess and compare alpha diversity and sample completeness among our surveys. Firstly, Hill numbers, also known as the effective number of equally abundant species qD, were used (Hill 1973; Chao et al. 2014), and the three most widely used orders (i.e. q = 0, q = 1 and q = 2) were assessed here. 0D corresponds to species richness, which is an indication of the number of species present in the community without considering their abundances and thus gives disproportionate weight to rare species. 1D indicates the measure of Shannon diversity (i.e. exponential of Shannon entropy), which weighs each species according to its abundance in the community and can be interpreted as the number of ‘common’ species present in the assemblage. Finally, 2D represents Simpson diversity (i.e. inverse of Simpson concentration index), which discounts all but the dominant species and thus can be interpreted as the number of highly ‘abundant’ (or dominant) species in the community (Chao et al. 2020).
To allow for comparisons among the investigated benthic communities, the steps proposed by Chao et al. (2020), were followed as listed below. Firstly, sample completeness of each survey was estimated via a bootstrap method (n = 500) for q = 0, 1, 2 to compare the diversity detected, or sample completeness, between each depth zone and site (Table S3). Secondly, the observed and extrapolated diversities at each site and depth zone were estimated by computing individual-based and sample-size based R/E curves for Hill numbers of q = 0, 1, 2; with extrapolation to a sampling effort equal to double the number of individuals (extrapolation beyond this is unreliable due to prediction bias; (Chao et al. 2014)). Thirdly, the observed and extrapolated Hill numbers were compared to assess undetected diversity and compare diversity between the depth zones and sites (Table S4). However, these results must be taken with caution as they were extracted prior to sample completeness standardisation. Finally, diversity estimates were calculated at a standardised coverage Cmax (e.g. equally complete samples) to statistically compare the benthic diversities along the depth gradient and between sites, which corresponds to the sample coverage of the depth zone exhibiting the lowest coverage when samples are extrapolated to double the number of samples per depth zone at a site (Table S5), as used in El-Khaled et al. (2022). Here, the Cmax value was 96.4% (Fig. S1; Table S5), corresponding to the lowest sample completeness for q = 0, 1, 2.
Beta diversity analyses
Beta diversity was measured as the variation in morphospecies with depth and between sites. Jaccard’s dissimilarity was calculated, including its two components, turnover (i.e. species replacement) and nestedness (i.e. subset of a richer community), using the entire benthic community presence-absence data for each depth zone and site. Jaccard dissimilarity varies between 0 and 1, with values close to 1 revealing a high difference in the composition of benthic communities between two depth zones, while a low dissimilarity (close to 0) denotes similar benthic communities. These indices were calculated using the betapart package and plotted in a heatmap, as well as a barplot with ggplot2 package in R (Baselga and Orme 2012; Team R Core 2020).
Results
Alpha diversity
A total of 582 distinct morphospecies (OTUs) were identified from the benthic communities at Egmont Atoll, between 15 and 160 m, covering 17 higher taxa (ranging from phylum to genus; Table S2). All morphospecies identified can be consulted in the freely accessible catalogue created for the region (Diaz et al., 2023b). Species richness (q = 0) was observed to decrease with depth at both sites, with lower diversity observed at 110–120 m and 150–160 m compared to the shallower depth zones (Fig. 2a). Although only conservative minimum estimates could be obtained for species richness (as individual-based R/E curves did not reach an asymptote; Fig. 2a), at the fixed sample coverage of Cmax = 96.4%, benthic morphospecies richness was significantly lower for the two deepest zones compared to the shallower depth zones (Fig. S1c, Table S5; non-overlapping 95% confidence intervals (CIs) for the two deepest depth zones, overlapping CIs for 15 m to 90 m depth zones).
Overview of alpha diversity analysis based on Hill numbers. a) individual-based Rarefaction/Extrapolation (R/E) curves for q = 0. b) individual-based R/E curves for q = 1. c) individual-based R/E curves for q = 2. Shaded areas represent the 95% confidence intervals for each curve. Shades of blue represent the different surveyed depth zones at Ile Des Rats (IDR), and the shades of orange are the depth zones at Manta Alley (MA). Solid lines are the rarefaction curves while the dashed lines represent the extrapolation curves up to double the respective sample size
Estimates of Shannon and Simpson diversity reached asymptotes for all depths at both sites and overall showed significant decreases in diversity over the depth gradient, from 15 to 160 m (Fig. 2b, c; Fig. S1b, c; non-overlapping CIs). The exception was at IDR, where both Shannon and Simpson diversity were significantly higher at 30–40 m compared to 15–20 m (Fig. 2b, c). At both sites, Shannon diversity between 60–70 m and 150–160 m was significantly lower than observed at shallower depths; however, there was no significant difference in Shannon diversity between 80–90 m and 110–120 m (Fig. 2b). Simpson diversity differed between the two sites (Fig. 2c): at MA, Simpson diversity was significantly different between 15–20 m and 30–40 m and the deeper depths; however, the values did not differ significantly between the depth zones from 60–70 m to 110–120 m; while at IDR, the 80–90 m depth zone showed a significantly lower Simpson diversity, compared to both 60–70 and 110–120 m (Table S5). Finally, the values were significantly lower at 150–160 m compared to shallower depth zones for both sites (Fig. 2c; Fig. S1b, c; Table S5).
In this study, the diversity curves did not all reach an asymptote, indicating that total diversity was not captured (Fig. 2). This undetected diversity for each depth zone at each site is detailed in Table S4. Nevertheless, coverage-based sample completeness profiles revealed that between 93.8% and 97.3% of benthic morphospecies diversity was detected, depending on the depth zones and sites (Table S3), showing that while a few species were undetected, an acceptable standardized value for comparing species richness between communities was sampled (Moreno and Halffter 2000).
Beta diversity
Values of beta diversity between depth zones for the entire benthic community were similar between both study sites (Fig. 3a and d). Total beta diversity values were high between the depth zones, throughout the entire depth gradient for both sites (from 15 to 160 m; Jaccard 0.61 to > 0.96; Fig. 3, Table S6, Fig. S1), indicating a limited number of shared morphospecies between depth zones. Adjacent depth zones displayed less dissimilarity compared to depths located further apart (Jaccard 0.65 for 15–20 m/30–40 m, to 0.81 for 110–120 m/150–160 m; 0.85 for 80–90 m/150–160 m to 0.95 for 15–20 m/150–160 m, respectively; Fig. 3a and d, Table S7, Fig. S1).
Changes in beta diversity and its two components–turnover and nestedness – with depth for the entire benthic community at both study sites Ile Des Rats (IDR, a to c) and Manta Alley (MA, d to f), using Jaccard’s dissimilarity index from 0 to 1. Values close to 0 indicate a high level of similarity between the two communities being compared and values close to 1 indicate a low level of similarity
Turnover was the largest component of beta diversity, thus it can be considered as the main driver of differentiation between benthic communities, indicating that the change in morphospecies diversity along the depth gradient, from 15 to 160 m, was mostly due to species replacement (Jaccard > 0.5 to > 0.9; Fig. 3b and e; Table S7, Fig. S1). On the contrary, nestedness had only a limited influence on the overall dissimilarity with depth (Jaccard 0.01 to < 0.2; Fig. 3c and f; Table S7, Fig. S1); showing that, while species diversity broadly decreased with depth (Fig. 2), deeper communities were not just a subset of richer communities observed at shallower depths. The differences between the turnover and nestedness components, compared to total dissimilarity, are detailed in Fig. S1.
Between sites, for the same depth zone, beta diversity did not show a strong pattern (Fig. 4). Overall, dissimilarity was less pronounced between sites in shallow-water communities (15–20 m) compared to communities at mesophotic depths (30–160 m), with 60–70 m and 150–160 m zones showing the highest benthic dissimilarity at a local scale. Similarly to the beta diversity calculated along the depth gradient, turnover was the main component of beta dissimilarity between sites, indicating benthic dissimilarity is mainly driven by species replacement at a local scale.
Jaccard’s dissimilarity and its two components – turnover and nestedness – between the two study sites (Ile des Rats and Manta Alley) for a depth zone. Jaccard’s dissimilarity index ranges from 0 to 1, with values close to 0 indicating a high level of similarity between the two communities and values close to 1 indicating a low level of similarity
Discussion
This study provides novel, baseline data on the diversity of benthic communities within MCEs in the central Indian Ocean (Egmont Atoll, Chagos Archipelago). Using alpha and beta diversity measures, the results show that benthic communities within MCEs at Egmont Atoll host a high and unique biodiversity, highlighting the importance of considering MCEs within conservation and management plans. Here, diversity values were comparable to shallow-water coral reefs (Edgar et al. 2017), which are considered to be one of the most diverse ecosystems in the world (Bryant et al. 1998), and communities displayed a high species turnover along the depth gradient.
Species richness values estimated at Egmont Atoll (calculated at Cmax and ranging from 57 to 220 across the depth gradient; see Methods and Table S5) were analogous to values from a global study of inshore reef richness for invertebrates, both in the Chagos Archipelago (species richness of 120) and the wider tropical-subtropical regions (species richness ranged from 40 to 230) (Edgar et al. 2017). Furthermore, even the deepest zone (150–160 m) in the current study, displaying the lowest diversity values, had higher species richness than some shallow (~ 10 m) subtropical regions (i.e. 67.59 for IDR and 56.58 for MA compared to 40 in Edgar et al. (2017)).
When accounting for scleractinian corals only, the species richness and Shannon index values calculated in this study were similar to those measured in the Red Sea, within shallow-water reefs and the upper mesophotic zone (Table S2; (Kramer et al. 2019)), but slightly lower than coral species richness values in American Samoa (Montgomery et al. 2021). In addition, for one of our two study sites, total benthic biodiversity was higher at 30–40 m than 15–20 m (IDR, Fig. 2a, b, c). This trend has also been noted in the Caribbean for the entire benthic community (increase in species richness from 15 to 53 m, (Liddell and Ohlhorst 1988)), but also for scleractinian corals in French Polynesia (Pérez-Rosales, et al. 2022b) and octocorals in the Gulf of Aqaba (Shoham and Benayahu 2017). According to Pérez-Rosales et al. (2022a, b), this peak in coral richness could be generated by the range overlap of assemblages typical of shallow and mesophotic communities within this depth range, as observed in (Diaz et al., 2023a).
While comparison with other studies was challenging due to the lack of a standardised methodology to measure diversity, as well as the differences in terms of investigated taxa between studies, the high benthic diversity encountered within the entire mesophotic gradient in the Chagos Archipelago can still be acknowledged. Here, more than 90% of the total benthic megafauna diversity was detected (Table S3, see Table S4 for undetected diversity), which is considered as an acceptable standardized value for comparing species richness between communities (Moreno and Halffter 2000). Fundamentally, the sampling effort of this study obtained an efficient inventory of the benthic species community across the shallow to lower mesophotic depth gradient at Egmont Atoll. Hence, the authors suggest that future studies should estimate diversity indices using rarefied and extrapolated methods, to obtain a standardised and robust comparison between studies (Roswell et al. 2021).
Overall, the diversity of the benthic communities at Egmont Atoll decreased from the shallow water down to the lower mesophotic zone (from 15 to 160 m) at the two study sites, when accounting for rare, abundant, and highly abundant morphospecies (Hill numbers for species richness, Shannon and Simpson indices; Fig. 2). While studies of the entire benthic community down to mesophotic depths are scarce, this pattern has been observed in several locations, such as the Caribbean (Stefanoudis et al. 2019), the Western Indian Ocean (Stefanoudis et al. 2023) and the Pacific Ocean (Pyle et al. 2016). The decrease in benthic diversity with depth has also been described worldwide for scleractinian corals, in the Caribbean (Appeldoorn et al. 2019), the Indo-Pacific (Bridge et al. 2012; Rowley et al. 2019; Turak and DeVantier 2019; Laverick et al. 2020; Montgomery et al. 2021; Osuka et al. 2021) and the western Atlantic regions (Muir and Pichon 2019). On the contrary, sponge (Slattery and Lesser 2012; Eyal et al. 2019; Rowley et al. 2019; Diaz et al. 2023a), gorgonian and antipatharian (Bridge et al. 2012; Bo 2019; Rowley et al. 2019; Diaz et al. 2023a)) diversity in MCEs were shown to increase with depth. Similar trends in specific phylum change are presented in Table S2.
Benthic diversity and community composition change along the depth gradient may be explained by several environmental factors, as observed in a parallel study by (Diaz, et al. 2023a), which identified fifteen environmental variables contributing significantly to the distribution and community composition of MCEs over the depth gradient. Decreasing photosynthetically active radiation (PAR) and temperature were identified as the two main drivers of variation in community composition with depth at Egmont Atoll (Diaz et al, 2023a). Similarly, a global study across a variety of taxa identified PAR and nutrient availability (both dissolved and particulate organic matter) to be correlated with a community break observed at 60 m (Lesser et al. 2019). In the Caribbean (i.e. Bermuda and Little Cayman), temperature and light were the main factors correlated with changes in overall benthic morphotype richness and scleractinian corals with depth, respectively (Stefanoudis et al. 2019; Carpenter et al. 2022). A decrease in benthic diversity with depth can therefore be explained by several factors, but mostly by the decrease in PAR, temperature, and nutrient availability. These factors are important drivers of primary production, reflected here in the distribution of photosynthetic corals and algae.
Nevertheless, while morphospecies richness decreased with depth overall for the two study sites; for a standardised sample coverage (i.e. Cmax) and within the four shallowest depth zones (from 15 to 90 m), this decrease was not significant (Fig. 2d). However, Shannon and Simpson indices (i.e., accounting for abundant and highly abundant morphospecies), revealed significant differences between depth zones with an overall decrease in diversity indices with depth (except between 80–90 m and 110–120 m for q = 1, Fig. 2e; and between 60–70 m and 110–120 m at IDR, 80–90 m and 110–120 m at MA for q = 2, Fig. 2f). The relatively constant total community richness at the species (or morphospecies) level over a broad bathymetric range is suggested to be due to major shifts of the larger taxonomic categories (Liddell and Ohlhorst 1988). Indeed, whilst diversity values were relatively constant with depth (Fig. 2), numerous depth-specialist species were observed along the depth gradient at Egmont Atoll in a parallel study, with a gradual decrease in scleractinian corals with depth, while octocorals, hydrozoans, antipatharians, hydrozoans and sponges increased (Table S3; (Diaz et al. 2023a)). Depth specialist species included Astrogorgia sp, Leptoseris sp, and members of the Nephtheidae and Hydrozoa.
Shallow and mesophotic coral ecosystems observed at Egmont Atoll displayed a high dissimilarity in terms of benthic communities along the depth gradient (Fig. 3a, d), as well as among sites (Fig. 4). Turnover was found to be the main component of overall dissimilarity along the depth gradient and locally between sites, indicating that species replacement was the main driver of the differences in the benthic communities (Fig. 3). In addition, Jaccard dissimilarity displayed higher values between sites in mesophotic depth zones (30–160 m), compared to the shallower sites (15–20 m) (Fig. 4). The results presented in this study hence reveal the uniqueness of MCEs at Egmont Atoll.
A change in community composition was observed along the depth gradient (Table S2). Thus, rather than the deeper communities being a subset of the richer shallower communities, they are composed of a different assemblage of species. Similar trends in dissimilarity along the depth gradient have been observed in other studies investigating beta diversity for the entire benthic community, in the Caribbean and western Indian Ocean (Semmler et al. 2017; Stefanoudis et al. 2019, 2023; Osuka et al. 2021). In addition, high species turnover has also been observed for scleractinian corals in the Pacific and western Atlantic regions (Rocha et al. 2018; Montgomery et al. 2021; Pérez-Rosales, et al. 2022b). More recently, a study investigating the “whole community” using eDNA metabarcoding in Hawai’i found significant differences between reefs at 0–45 m and 60–90 m, for 18S-eukaryotes and COI-metazoans, with turnover identified as the highest component of the beta diversity (Hoban et al. 2023). However, a review by Muir and Pichon (2019) suggests that at mesophotic depths shallower than 60 m, coral communities are a subset of shallow-water reefs, with a shift in coral fauna occurring from 60 m. This shift in community composition at 60 m has been described in several locations and defined as a “community break” (Lesser et al. (2019)). Here, turnover was nevertheless high even between the shallowest depth zones (15-20 m and 30-40 m), supporting previous studies suggesting that ecological connectivity between the shallow and mesophotic zones is limited (Rocha et al. 2018; Soares et al. 2020; Hoban et al. 2023; Stefanoudis et al. 2023) and driven by a change in benthic species along the depth gradient, rather than a loss of species (Fig. 3; Table S7, Fig. S1). Hence, these findings add evidence to the uncertainty surrounding the DRRH and the reseeding potential of MCEs should shallow-water reefs at Egmont Atoll, and likely in the wider Chagos Archipelago, be subject to disturbance, due to the high dissimilarity in benthic communities observed with depth. In addition, mesophotic reefs at Egmont Atoll have been found to be vulnerable to increases in temperature, with severe coral bleaching occurring between 60–90 m, in the absence of shallow coral bleaching, in late 2019 (Diaz et al. 2023c), challenging one of the assumptions of the DRRH that deep reefs are less impacted by climate change than their shallow-water counterparts (Glynn 1996).Whilst mesophotic coral bleaching was occurring during data collection, every organism, including bleached scleractinian corals, were annotated, and included within the analysis, thus the bleaching is unlikely to have affected the diversity results presented here.
Alpha and beta diversity indices were assessed through the visual annotation of underwater images, where all organisms were identified to the highest taxonomic resolution possible and distinct morphospecies were assigned an Operational Taxonomic Unit (OTU), as standard taxonomic approaches are not often applicable to image data. A morphospecies may then correspond to species, genus, family or higher taxonomic levels. Whilst images were annotated by the same observer (who also re-annotated 5% of the images for quality assurance), this method lacks taxonomic resolution, which may consequently have had a negative effect on the diversity results presented here and led to the grouping of several species within one morphospecies, or on the contrary, separation of one species into several colour morphs.
During the current study, it was not possible to collect the physical samples typically required to enable traditional taxonomic investigation. Taxon differentiation was thus missing at the fine scale, molecular and genetic levels, likely leading to lower (or higher) observed diversity values and differences in diversity than actually occurs in reality. MCEs are still relatively poorly understood in taxonomic terms, thus, high-resolution images with close-ups in addition to physical sample collection and taxonomic expertise are crucial to resolve their true diversity and the variation in taxa along the depth gradient.
This study investigated alpha and beta diversities for benthic communities, along a large depth gradient, for the first time in the central Indian Ocean. These indices are crucial indicators to provide evidence-based information for management and conservation actions. Indeed, despite meeting the international criteria used to identify priority areas for conservation policies (Soares et al. 2020), MCEs are still considered under-protected and under-represented in marine spatial planning (Stefanoudis et al. 2022), and they are usually incidentally included in MPAs due to geopolitical boundaries, rather than purposefully (Turner et al. 2019; Stefanoudis et al. 2022). MCEs, while encompassed within the fully protected Chagos MPA since 2010, have not been included in research and specific protection until 2018, where an objective to improve their research and protection by 2023 was mentioned in the conservation management planning workshop (Blue Belt Programme 2018). However, the authors are not aware of any specific management steps being taken regarding the protection of MCEs within the Chagos MPA, other than the research presented here.
Conclusions
The current study provides baseline data on the benthic diversity within MCEs in a remote, pristine area. High species turnover along the depth gradient and among sites indicates that the MCEs of the Chagos Archipelago are of limited refuge for threatened shallow-water coral reefs and are a significant ecosystem in terms of diversity and endemicity. Thus, following the call of numerous scientists worldwide (e.g. Rocha et al. 2018; Soares et al. 2020; Stefanoudis et al. 2022), the authors advocate for protection of these critical mesophotic ecosystems, in order to preserve overall biodiversity and the associated benefits.
Data availability
Raw data are available on Dryad, https://doi.org/10.5061/dryad.pzgmsbcs8. The benthic species catalogue is currently on Zenodo (Diaz et al. 2023b). All scripts and data necessary to replicate the analysis of this study are available on GitHub: https://github.com/ClaraDiaz974/Mesophotic_benthic_diversity.
References
Appeldoorn RS, Alfaro M, Ballantine DL, Bejarano I, Ruíz HJ, Schizas NV, Schmidt WE, Sherman CE, Weil E (2019) Puerto Rico. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer International Publishing, Cham, pp 111–129. https://doi.org/10.1007/978-3-319-92735-0_7
Baldwin CC, Tornabene L, Robertson DR (2018) Below the Mesophotic. Sci Rep 8(1):4920. https://doi.org/10.1038/s41598-018-23067-1
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19(1):134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A, Orme CDL (2012) Betapart: an R package for the study of beta diversity betapart package. Methods Ecol Evol 3(5):808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Blue Belt Programme (2018) British Indian Ocean Territory (BIOT) conservation management planning workshop 5–6th March 2018 workshop report.
Bo M, Montgomery AD, Opresko DM, Wagner D, Bavestrello G (2019) Antipatharians of the mesophotic zone: four case studies. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer Nature Switzerland
Bongaerts P, Smith TB (2019) Beyond the “deep reef refuge” hypothesis: a conceptual framework to characterize persistence at depth. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer Nature Switzerland
Bongaerts P, Ridgway T, Sampayo EM, Hoegh-Guldberg O (2010) Assessing the ‘deep reef refugia’ hypothesis: focus on Caribbean reefs. Coral Reefs 29(2):309–327. https://doi.org/10.1007/s00338-009-0581-x
Bongaerts P, Riginos C, Brunner R, Englebert N, Smith SR, Hoegh-Guldberg H (2017) ‘Deep reefs are not universal refuges: reseeding potential varies among coral species’,. Sci Adv. https://doi.org/10.1126/sciadv.1602373
Bridge TCL, Fabricius KE, Bongaerts P, Wallace CC, Muir PR, Done TJ, Webster JM (2012) Diversity of Scleractinia and Octocorallia in the mesophotic zone of the Great Barrier Reef, Australia. Coral Reefs 31(1):179–189. https://doi.org/10.1007/s00338-011-0828-1
Bridge T C L and Guinotte J M (2013) ‘Mesophotic Coral Reef Ecosystems in the Great Barrier Reef World Heritage Area in the Great Barrier Reef World’, Great Barrier Reef Marine Park Authority [Preprint], (109). https://hdl.handle.net/11017/2776.
Bryant DL, Burke L, McManus J, Spalding M (1998) Reefs at risk. World Resources Institute.
Carpenter GE, Chequer AD, Weber S, Mass T, Goodbody‐Gringley G (2022) Light and photoacclimatization drive distinct differences between shallow and mesophotic coral communities. Ecosphere 13(8):e4200. https://doi.org/10.1002/ecs2.4200
Chao A, Gotelli NJ, Hsieh TC, Sander EL, Ma KH, Colwell RK, Ellison AM (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84(1):45–67. https://doi.org/10.1890/13-0133.1
Chao A, Kubota Y, Zelený D, Chiu C-H, Li C-F, Kusumoto B, Yasuhara M, Thorn S, Wei C-L, Costello MJ, Colwell RK (2020) Quantifying sample completeness and comparing diversities among assemblages. Ecol Res 35(2):292–314. https://doi.org/10.1111/1440-1703.12102
Colwell RK, Chao A, Gotelli NJ, Lin S-Y, Mao CX, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5(1):3–21. https://doi.org/10.1093/jpe/rtr044
Cumming G, Fidler F, Vaux DL (2007) Error bars in experimental biology. J Cell Biol 177(1):7–11. https://doi.org/10.1083/jcb.200611141
de Oliveira M, Soares JT, de Araújo S, Ferreira MC, Santos BA, Boavida JRH, Costantini F, Rossi S (2020) Why do mesophotic coral ecosystems have to be protected? Sci Total Environ 726:138456. https://doi.org/10.1016/j.scitotenv.2020.138456
de Oliveira M, Soares TC, Tavares L, de Bastos Macêdo Carneiro, P (2019) Mesophotic ecosystems: distribution, impacts and conservation in the South Atlantic. Divers Distrib 25(2):255–268. https://doi.org/10.1111/ddi.12846
Diaz C, Howell KL, Robinson E, Hosegood P, Bolton A, Ganderton P, Arber P, Attrill MJ, Foster NL (2023a) Light and temperature drive the distribution of mesophotic benthic communities in the central Indian Ocean. Divers Distributions 29(12):1578–1593. https://doi.org/10.1111/ddi.13777
Diaz C, Foster NL, Howell KL (2023b) Shallow and mesophotic benthos reference image database for the Chagos Archipelago, central Indian Ocean’, Zenodo. Available at: https://doi.org/10.5281/ZENODO.7948517
Diaz C, Foster NL, Attrill MJ, Bolton A, Ganderton P, Howell KL, Hosegood P (2023c) Mesophotic coral bleaching associated with changes in thermocline depth. Nat Commun. https://doi.org/10.1038/s41467-023-42279-2
Edgar GJ, Alexander TJ, Lefcheck JS, Bates AE, Kininmonth SJ, Thomson RJ, Emmett Duffy J, Costello MJ, Stuart-Smith RD (2017) Abundance and local-scale processes contribute to multi-phyla gradients in global marine diversity. Sci Adv. https://doi.org/10.1126/sciadv.1700419
El-Khaled YC, Daraghmeh N, Tilstra A, Roth F, Huettel M, Rossbach FI, Casoli E, Koester A, Beck M, Meyer R, Plewka J, Schmidt N, Winkelgrund L, Merk B, Wild C (2022) Fleshy red algae mats act as temporary reservoirs for sessile invertebrate biodiversity. Commun Biol. https://doi.org/10.1038/s42003-022-03523-5
Englebert N, Bongaerts P, Muir PR, Hay KB, Pichon M, Hoegh-Guldberg O (2017) Lower mesophotic coral communities (60-125 m depth) of the northern great barrier reef and coral sea. PLoS ONE 12(2):e0170336. https://doi.org/10.1371/journal.pone.0170336
Eyal G, Cohen I, Eyal-Shaham L, Ben-Zvi O, Tikochinski Y, Loya Y (2019) Photoacclimation and induction of light-enhanced calcification in the mesophotic coral Euphyllia paradivisa. Royal Soc Open Sci 6(2):180527. https://doi.org/10.1098/rsos.180527
García-Martínez MÁ, Valenzuela-González JE, Escobar-Sarria F, López-Barrera F, Castaño-Meneses G (2017) The surrounding landscape influences the diversity of leaf-litter ants in riparian cloud forest remnants. PLoS ONE 12(2):e0172464. https://doi.org/10.1371/journal.pone.0172464
Glynn PW (1996) Coral reef bleaching: facts, hypotheses and implications. Glob Change Biol 2(6):495–509. https://doi.org/10.1111/j.1365-2486.1996.tb00063.x
Hays GC, Koldewey HJ, Andrzejaczek S, Attrill MJ, Barley S, Bayley DTI, Benkwitt CE, Block B, Schallert RJ, Carlisle AB, Carr P, Chapple TK, Collins C, Diaz C, Dunn N, Dunbar RB, Eager DS, Engel J, Embling CB, Esteban N, Ferretti F, Foster NL, Freeman R, Gollock M, Graham NAJ, Harris JL, Head CEI, Hosegood P, Howell KL, Hussey NE, Jacoby DMP, Jones R, Pilly SS, Lange ID, Letessier TB, Levy E, Lindhart M, McDevitt-Irwin JM, Meekan M, Meeuwig JJ, Micheli F, Mogg AOM, Mortimer JA, Mucciarone DA, Nicoll MA, Nuno A, Perry CT, Preston SG, Rattray AJ, Robinson E, Roche RC, Schiele M, Sheehan EV, Sheppard A, Sheppard C, Smith AL, Soule B, Spalding M, Stevens GMW, Steyaert M, Stiffel S, Taylor BM, Tickler D, Trevail AM, Trueba P, Turner J, Votier S, Wilson B, Williams GJ, Williamson BJ, Williamson MJ, Wood H, Curnick DJ (2020) A review of a decade of lessons from one of the world’s largest MPAs: conservation gains and key challenges. Marine Biol. https://doi.org/10.1007/s00227-020-03776-w
Head CEI, Bayley DTI, Rowlands G, Roche RC, Tickler DM, Rogers AD, Koldewey H, Turner JR, Andradi-Brown DA (2019) Coral bleaching impacts from back-to-back 2015–2016 thermal anomalies in the remote central Indian Ocean. Coral Reefs 38(4):605–618. https://doi.org/10.1007/s00338-019-01821-9
Hernandez‐Agreda A, Sahit FM, Englebert N, Guldberg O-H, Bongaerts P (2022) Hidden in the deep: distinct benthic trajectories call for monitoring of mesophotic reefs. Conserv Lett 15(2):e12875
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54(2):427–432. https://doi.org/10.2307/1934352
Hinderstein LM, Marr JCA, Martinez FA, Dowgiallo MJ, Puglise KA, Pyle RL, Zawada DG, Appeldoorn R (2010) Theme section on “mesophotic coral ecosystems: characterization, ecology, and management.” Coral Reefs 29(2):247–251. https://doi.org/10.1007/s00338-010-0614-5
Hoban ML, Bunce M, Bowen BW (2023) Plumbing the depths with environmental DNA ( eDNA ): metabarcoding reveals biodiversity zonation at 45–60 m on mesophotic coral reefs. Mol Ecol 32(20):5590–5608. https://doi.org/10.1111/mec.17140
Howell KL, Davies JS, Louise Allcock A, Braga-Henriques A, Buhl-Mortensen P, Carreiro-Silva M, Dominguez-Carrió C, Durden JM, Foster NL, Game CA, Hitchin B, Horton T, Hosking B, Jones DOB, Mah C, Marchais CL, Menot L, Morato T, Pearman TRR, Piechaud N, Ross RE, Ruhl HA, Saeedi H, Stefanoudis PV, Taranto GH, Thompson MB, Taylor JR, Tyler P, Vad J, Victorero L, Vieira RP, Woodall LC, Xavier JR, Wagner D (2019) A framework for the development of a global standardised marine taxon reference image database (SMarTaR-ID) to support image-based analyses. PLoS ONE 14(12):e0218904. https://doi.org/10.1371/journal.pone.0218904
Hsieh T, Ma K and Chao A (2020) ‘iNEXT: Interpolation and Extrapolation for Species Diversity (2.0.20)’, Comprehensive R Archive Network (CRAN) [Preprint].
Kahng SE, Copus JM, Wagner D (2014) Recent advances in the ecology of mesophotic coral ecosystems (MCEs). Curr Opin in Environ Sustain 7:72–81. https://doi.org/10.1016/j.cosust.2013.11.019
Kramer N, Eyal G, Tamir R, Loya Y (2019) Upper mesophotic depths in the coral reefs of Eilat, Red Sea, offer suitable refuge grounds for coral settlement. Sci Rep 9(1):1–12. https://doi.org/10.1038/s41598-019-38795-1
Kramer N, Tamir R, Eyal G, Loya Y (2020) Coral Morphology portrays the spatial distribution and population size-structure along a 5–100m depth gradient. Front Marine Sci. https://doi.org/10.3389/fmars.2020.00615
Langenkämper D, Zurowietz M, Schoening T, Nattkemper TW (2017) Biigle 2.0-browsing and anotating large marine image collections. Front Mar Sci 4:83
Laverick JH, Tamir R, Eyal G, Loya Y (2020) A generalized light‐driven model of community transitions along coral reef depth gradients. Glob Ecol Biogeography 29(9):1554–1564. https://doi.org/10.1111/geb.13140
Lesser MP, Slattery M, Laverick JH, Macartney KJ, Bridge TC (2019) Global community breaks at 60 m on mesophotic coral reefs. Glob Ecol Biogeography 28(10):1403–1416. https://doi.org/10.1111/geb.12940
Liddell WD, Ohlhorst SL (1988) Hard substrata community patterns, 1–120 m, North Jamaica. Palaios 3(4):413. https://doi.org/10.2307/3514787
Loya Y, Eyal G, Treibitz T, Lesser MP, Appeldoorn R (2016) Theme section on mesophotic coral ecosystems: advances in knowledge and future perspectives. Coral Reefs 35(1):1–9. https://doi.org/10.1007/s00338-016-1410-7
McDevitt-Irwin JM, Kappel C, Harborne AR, Mumby PJ, Brumbaugh DR, Micheli F (2021) Coupled beta diversity patterns among coral reef benthic taxa. Oecologia 195(1):225–234. https://doi.org/10.1007/s00442-020-04826-2
Miyazawa E, Montilla LM, Agudo-Adriani EA, Ascanio A, Mariño-Briceño G, Croquer A (2020) On the importance of spatial scales on beta diversity of coral assemblages: a study from Venezuelan coral reefs. PeerJ 8:e9082. https://doi.org/10.7717/peerj.9082
Montgomery AD, Fenner D, Donahue MJ, Toonen RJ (2021) Community similarity and species overlap between habitats provide insight into the deep reef refuge hypothesis. Sci Reports 11(1):23787. https://doi.org/10.1038/s41598-021-03128-8
Morais J, Santos BA (2018) ‘Limited potential of deep reefs to serve as refuges for tropical Southwestern Atlantic corals’,. Ecosphere. https://doi.org/10.1002/ecs2.2281
Moreno CE, Halffter G (2000) Assessing the completeness of bat biodiversity inventories using species accumulation curves. J Appl Ecol 37(1):149–158. https://doi.org/10.1046/j.1365-2664.2000.00483.x
Muir PR, Pichon M (2019) Biodiversity of Reef-Building, Scleractinian Corals. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic coral ecosystems. Springer International Publishing, Cham, p 589
Osuka KE, McClean C, Stewart BD, Bett BJ, Le Bas T, Howe J, Samoilys M (2021) Characteristics of shallow and mesophotic environments of the Pemba Channel, Tanzania: implications for management and conservation. Ocean Coast Manag 200:105463. https://doi.org/10.1016/j.ocecoaman.2020.105463
Pérez‐Rosales G, Pichon M, Rouzé H, Villéger S, Torda G, Bongaerts P, Carlot J, Parravicini V, Hédouin L (2022a) Mesophotic coral ecosystems of French Polynesia are hotspots of alpha and beta generic diversity for scleractinian assemblages. Divers Distrib 28(7):1391–1403. https://doi.org/10.1111/ddi.13549
Pérez-Rosales G, Hernández-Agreda A, Bongaerts P, Rouzé H, Pichon M, Carlot J, Torda G, Parravicini V, Hédouin L (2022b) Mesophotic depths hide high coral cover communities in French Polynesia. Sci Total Environ 844:157049. https://doi.org/10.1016/j.scitotenv.2022.157049
Pyle RL, Copus JM (2019) Mesophotic coral ecosystems: introduction and overview. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic coral ecosystems. Springer International Publishing, Cham, pp 3–27
Pyle RL, Boland R, Bolick H, Bowen BW, Bradley CJ, Kane C, Kosaki RK, Langston R, Longenecker K, Montgomery A, Parrish FA, Popp BN, Rooney J, Smith CM, Wagner D, Spalding HL (2016) A comprehensive investigation of mesophotic coral ecosystems in the Hawaiian Archipelago. PeerJ 4:e2475. https://doi.org/10.7717/peerj.2475
Edward Roberts T, Tom CL, Bridge MJ, Caley JS, MadinBaird AH (2019) Resolving the depth zonation paradox in reef‐building corals. Ecology 100(8):e02761. https://doi.org/10.1002/ecy.2761
Rocha LA, Pinheiro HT, Shepherd B, Papastamatiou YP, Luiz OJ, Pyle RL, Bongaerts P (2018) Mesophotic coral ecosystems are threatened and ecologically distinct from shallow water reefs. Science 361(6399):281–284. https://doi.org/10.1126/science.aaq1614
Roswell M, Dushoff J, Winfree R (2021) A conceptual guide to measuring species diversity. Oikos 130(3):321–338. https://doi.org/10.1111/oik.07202
Rowley SJ, Edward Roberts T, Coleman RR, Spalding HL, Joseph E, Dorricott MKL (2019) Pohnpei, federated States of Micronesia. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer International Publishing, Cham, pp 301–320
Schoening T, Osterloff J, Nattkemper TW (2016) ‘RecoMIA-recommendations for marine image annotation: lessons learned and future directions.’ Front Mar Sci. https://doi.org/10.3389/fmars.2016.00059
Semmler RF, Hoot WC, Reaka ML (2017) Are mesophotic coral ecosystems distinct communities and can they serve as refugia for shallow reefs? Coral Reefs 36(2):433–444. https://doi.org/10.1007/s00338-016-1530-0
Shoham E, Benayahu Y (2017) Higher species richness of octocorals in the upper mesophotic zone in Eilat (Gulf of Aqaba) compared to shallower reef zones. Coral Reefs 36(1):71–81. https://doi.org/10.1007/s00338-016-1528-7
Slattery M and Lesser M P (2012) Mesophotic coral reefs: a global model of community structure and function, In: Proceedings of the 12th International Coral Reef Symposium, Cairns, Australia, 9–13 July 2012 [Preprint], (9C Ecology of mesophotic coral reefs).
Socolar JB, Gilroy JJ, Kunin WE, Edwards DP (2016) How should beta-diversity inform biodiversity conservation? Trends Ecol Evol 31(1):67–80. https://doi.org/10.1016/j.tree.2015.11.005
Stefanoudis PV, Rivers M, Smith SR, Schneider CW, Wagner D, Ford H, Rogers AD, Woodall LC (2019) Low connectivity between shallow, mesophotic and rariphotic zone benthos. Royal Society Open Science 6(9):190958. https://doi.org/10.1098/rsos.190958
Stefanoudis PV, Talma S, Fassbender N, Swanborn D, Ochieng CN, Mearns K, Komakoma JD, Otwoma LM, Mbije NE, Osuka KE, Samoilys M, Shah N, Samaai T, Trotzuk E, Tuda A, Zivane F, Wagner D, Woodall LC (2022) Stakeholder‐derived recommendations and actions to support deep‐reef conservation in the Western Indian Ocean. Conserv Lett 16(1):e12924. https://doi.org/10.1111/conl.12924
Stefanoudis P, Fassbender N, Samimi-Namin K, Adam P-A, Ebrahim A, Harlay J, Koester A, Samoilys M, Sims H, Swanborn D, Talma S, Winter S, Woodall L (2022) Trait-based approaches reveal that deeper reef ecosystems in the Western Indian Ocean are functionally distinct. SSRN Electron J. https://doi.org/10.2139/ssrn.4121008
Team R Core (2020) ‘R: A language and environment for statistical computing’, R foundation for statistical computing, Vienna Austria [Preprint]. Available at: https://www.r-project.org/.
Turak E, DeVantier L (2019) Reef-building corals of the upper mesophotic zone of the central Indo-West Pacific. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer International Publishing, Cham, pp 621–651
Turner JA, Andradi-Brown DA, Gori A, Bongaerts P, Burdett HL, Ferrier-Pagès C, Voolstra CR, Weinstein DK, Bridge TCL, Costantini F, Gress E, Laverick J, Loya Y, Goodbody-Gringley G, Rossi S, Taylor ML, Viladrich N, Voss JD, Williams J, Woodall LC, Eyal G (2019) Key Questions for Research and Conservation of Mesophotic Coral Ecosystems and Temperate Mesophotic Ecosystems. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic Coral Ecosystems. Springer International Publishing, Cham, pp 989–1003
Whittaker RH (1960) vegetation of the Siskiyou Mountains, Oregon and California. Ecol Monogr 30(3):279–338. https://doi.org/10.2307/1943563
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21(2–3):213–251. https://doi.org/10.2307/1218190
Wolters V, Bengtsson J, Zaitsev AS (2006) Relationship among the species richness of different taxa. Ecology 87(8):1886–1895. https://doi.org/10.1890/0012-9658(2006)87[1886:RATSRO]2.0.CO;2
Acknowledgements
We thank the United Kingdom Foreign and Commonwealth Office and the British Indian Ocean Territory Administration for allowing us to undertake this research. We thank the crew of the Tethys Supporter for their assistance throughout the cruises. We thank Edward Robinson for general help and for generating Egmont map. Thanks to Peter Ganderton and Peter Arber for their technical support throughout the cruises. We finally thank Phil Alderslade, Yehuda Benayahu, Michel Pichon, Jeremy Horowitz, Mathilde Godefroid and Gonzalo Perez-Rosales for their assistance during specimen identification on images.
Funding
This research has been funded by the Garfield Weston Foundation and the Bertarelli Foundation.
Author information
Authors and Affiliations
Contributions
Clara Diaz: Conceptualization, Data collection and curation, Formal analysis, Writing—original draft preparation. Kerry Howell: Funding acquisition, Conceptualization, Data collection, Writing—reviewing and editing. Phil Hosegood: Funding acquisition, Project leading, Writing—reviewing and editing. Adam Bolton: Multibeam data collection and analysis. Nicola Foster: Funding acquisition, Conceptualization, Data collection, formal analysis, supervision of first author, Writing—reviewing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Diaz, C., Howell, K.L., Hosegood, P. et al. Diverse and ecologically unique mesophotic coral ecosystems in the central Indian Ocean. Coral Reefs (2024). https://doi.org/10.1007/s00338-024-02535-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00338-024-02535-3