Abstract
Coral communities are threatened by an increasing plethora of abiotic and biotic disturbances. Preventing the ensuing loss of coral coverage and diversity calls for a mechanistic understanding of resilience across coral species and populations that is currently lacking in coral reef science. Assessments into the dynamics of coral populations typically focus on their long-term (i.e. asymptotic) characteristics, tacitly assuming stable environments in which populations can attain their long-term characteristics. Instead, we argue that greater focus is needed on investigating the transient (i.e. short-term) dynamics of coral populations to describe and predict their characteristics and trajectories within unstable environments. Applying transient demographic approaches to evaluating and forecasting the responses of coral populations to disturbance holds promise for expediting our capacity to predict and manage the resilience of coral populations, species, and communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coral populations worldwide are sensitive to a range of biotic and abiotic pressures (Riegl 2020), many of which will be exacerbated by future climatic change and human exploitation (Wolff et al. 2018; Riegl and Glynn 2020). Subsequently, the future status of coral reef ecosystems relies on the resilience of coral populations, specifically their capacity to endure, and recover from, increasingly frequent disturbance events (Holling 1973). Accordingly, a resilience-orientated focus is becoming commonplace in reef management (Mcleod et al. 2019, 2021), necessitating an ability to predict the responses of coral populations to varying biotic and abiotic regimes (Lam et al. 2020). Yet, a limited understanding for the dynamics underpinning coral population performance and resilience still hinders our capacity to recognise and manage the climate induced reassembly and/or collapse of coral reef ecosystems (Bellwood et al. 2004; Edmunds and Riegl 2020).
Although limited, the application of demographic theory within coral research is not a novel concept (Edmunds et al. 2014). Structured population models offer insight into how the state of individuals (typically their size, age, or developmental stage; Caswell 2001) arbitrates their survival and reproduction and how these fitness components in turn shape the overall responses of populations to local biotic and abiotic conditions (Box 1; Benton et al. 2006). Understanding how patterns in the survival, progression (i.e. growth), retrogression (i.e. negative growth, shrinkage; Salguero-Gómez and Casper 2010), and reproduction of individual colonies, influence population-level characteristics will improve effective management (Riegl et al. 2018; Brito-Millán et al. 2019a; Madin et al. 2020; Pisapia et al. 2020). Appropriately, state-structured demographic approaches are being increasingly utilised to quantify the mechanisms supporting or preventing the persistence and revival of disturbed coral communities (e.g. Kayal et al. 2018; Cant et al. 2021c). Accordingly, there is a growing appreciation for how the size structure of coral populations influences their dynamics and endurance (Bak and Meesters 1998, 1999; Anderson and Pratchett 2014; Dietzel et al. 2020; Lachs et al. 2021), with recent appeals for efforts to be concentrated towards gauging the resilience of coral communities (Pisapia et al. 2020). However, we argue here that the contemporary application of state-structured demographic tools in evaluating the viability of coral populations, and their communities, does not reflect our need to anticipate their resilience.
Records of survival, size, and reproductive output, collected from tagged coral colonies over time, can enhance our understanding of population trajectories and, when collected across multiple populations, can be used to predict community reassembly under future climate scenarios. Here, subscript notation is used to reflect how the changing state of individuals corresponds with changes in individual-level characteristics. In this schematic the state of individuals refers to colony size and thus the growth (g) of individuals between size x and y is expressed as gyx
Prevailing knowledge of coral population dynamics centres around long-term (i.e. asymptotic) population characteristics (Box 2), with measures of long-term population growth rate (λ) often used for projecting the viability of coral assemblages (e.g. Foster and Foster 2013, 2018; Edmunds 2015, 2020; Riegl et al. 2017; Kodera et al. 2020; Cant et al. 2021c; Shlesinger and van Woesik 2021). Indeed, the same is true across ecological theory, with our understanding of population dynamics focused on asymptotic population characteristics (Morozov et al. 2020). Quantifying the asymptotic characteristics of various populations presents a valuable comparative tool for exploring abiotic tolerances (Beissinger and Westphal 1998). Yet, for populations to realise their asymptotic potential, they require the persistence of stable conditions over extended timescales (Caswell 2001). In reality, though, seasonality exposes populations to periodic abiotic shifts, whilst predation, human exploitation, or storm and fire events generate recurrent and instantaneous disturbances. These short-term abiotic and biotic disturbances ensure that natural environments rarely exist at a stable equilibrium (Williams et al. 2011; Hastings et al. 2018). Consequently, evaluating the resilience and viability of populations requires an understanding for how the dynamics of populations manifest across timescales more representative of the disturbance cycles to which they are exposed (Hastings 2010). Here, we discuss why greater commitment towards quantifying the short-term, or transient, dynamics of coral populations is therefore required to enhance our capacity for anticipating the future resilience of coral reef ecosystems.
The cumulative number of studies that explicitly explore the state-structured demographics of coral populations as identified by our search for the term coral* AND demograph* across the peer-reviewed literature within the SCOPUS database. The colour scale differentiates between studies that focus on only investigating patterns in colony survival, growth, and/or reproduction, and those that present estimates of either deterministic population growth (λ), stochastic asymptotic characteristics (λs), or transient population characteristics
Transient versus asymptotic population dynamics
Structured population models characterise and define the trajectories of populations according to the individual-level allocation of resources towards survival, changes in size (or the state of interest), and reproduction. Populations can be structured according to the distribution of their individuals across some state variable, be that age (Leslie 1945), developmental stage (Lefkovitch 1965), size (Easterling et al. 2000), or a combination (Caswell et al. 2018). The transition of individuals across state classes is, in turn, mediated by selection gradients that emerge from the state-specific patterns of survival, development, and reproduction (Groenendael et al. 1988; van Tienderen 2000). In state-structured demography, patterns across these vital rates can subsequently be condensed into a population projection matrix (A), with each matrix element (aij) expressing the expected transition of individuals into state class i from state class j during the time interval t to t + 1, or the per-capita a/sexual contributions of state j individuals into state class i during that same interval (Caswell 2001). This population matrix can then be used to project the size (N, number of individuals) and structure (n, number of individuals in each state) of a population:
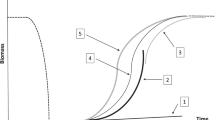
Within a stable environment, the state structure of a population is expected to converge towards its stationary equilibrium, after which the state relative composition of the population will remain constant (Caswell 2001). At its stationary equilibrium, therefore, the size of a population is expected to change at a constant rate through time, termed its asymptotic population growth rate (λ; Caswell 2001). This value of λ, for any state-structured population, is equal to the dominant eigenvalue of its corresponding population matrix A and exists on a non-negative scale reflecting the proportional growth (λ > 1), decline (λ < 1), or demographic equilibrium (λ = 1) of a population over time. For their stable structure to manifest, however, populations typically require time in a constant environment to allow the state distribution of their individuals to converge towards its equilibrium state without displacement by local disturbances (Fig. 3; Bierzychudek 1999). Yet, populations are rarely exposed to stable conditions or equilibria (Hastings 2001, 2004; Williams et al. 2011), and the vital rate patterns used in constructing population projection matrices are themselves not consistent and contingent on the environmental conditions in which they are observed (Benton and Grant 1996; Boyce et al. 2006). Moreover, as populations change in size their dynamics are subject to density–dependant regulation (Engen et al. 1998; Lande et al. 2003). The dynamics of populations are, therefore, expected to vary over time as environmental conditions change (Tuljapurkar and Orzack 1980; Tuljapurkar 1989).
At stationary equilibrium, populations are expected to change size at a constant rate, termed their asymptotic growth rate (λ). However, to display asymptotic characteristics, populations need a stable state structure and so require time at equilibrium to enable the development of this stable structure. In the absence of a stable state structure, populations will display varying growth rates as they converge towards asymptotic expectations, even within equilibrate environments. Note that, although in this schematic the populations growth rate is increasing towards its asymptotic trajectory, estimates of λ can also reflect asymptotic decline
The perpetual instability generated by regular disturbances ensures that populations rarely attain their asymptotic trajectories (Hastings 2001; Hastings et al. 2018). Natural environments persist within a non-equilibrate condition maintaining populations within a transient state during which their trajectories can radically differ from asymptotic expectations (Stott et al. 2010, 2011; Hastings et al. 2018). Anticipating the future condition and endurance of populations thus requires an awareness for their transient characteristics (Ezard et al. 2010). Transient shifts in the state structure of populations can elevate (amplification) or diminish (attenuation) their growth rates relative to asymptotic projections (Townley et al. 2007). Equally, for populations existing in a transient state, there is a natural tendency for their state structure to converge towards a stable distribution (Caswell 2001). Intuitively, with population resilience comprised of the attributes of resistance (the ability to withstand disturbances), compensation (the ability to benefit from disturbances) and recovery (the speed and ability to reattain stationary equilibria), the transient dynamics of populations therefore represent quantifiable measures of these features of resilience (Box 3; Hodgson et al. 2015; Capdevila et al. 2020).
Various measures exist for quantifying transient population characteristics. Disturbances (*) disrupt the structural composition of populations forcing them into a transient phases during which their trajectories will deviate from asymptotic expectations (λ, dashed line). The response of a population during this transient phase is contingent on their capacity for amplification (increases in population growth) and attenuation (declines in population growth). Using the measures of reactivity and maximal amplification (describing the initial, and maximum, increase in population growth rate during transient conditions) ecologists can quantify the relative amplification potential of natural populations. Alternatively, the measures of first-step attenuation and maximal attenuation can be used to describe the initial, and maximum decline in population growth rate during transient conditions. Meanwhile, damping ratio estimates describe the time required for a perturbed population to reobtain a stable equilibrium. Accordingly, by revealing the capacity for populations to grow (compensation), avoid declines (resistance), and/or recover following a disturbance event, these transient metrics present ecologists with measures of population resilience
Transient dynamics and the resilience of coral communities
Transient demographic approaches present an effective tool for explicitly evaluating the resilience of natural populations (Capdevila et al. 2020). Indeed, such approaches have proven instrumental in our understanding of the establishment of populations within new and variable environments (Iles et al. 2016; McDonald et al. 2016, 2017; Jelbert et al. 2019). Exploring the transient dynamics of coral populations can thus enhance our understanding regarding the future diversity, condition, and resilience, of coral communities. Globally, coral communities are being exposed to increasingly recurrent thermal stress events, at frequencies soon expected to exceed the recovery time required by most coral species (Hughes et al. 2018; Sully et al. 2019). Presently, considerable value is placed on classifying coral species according to their morphological and functional traits, with the subsequent categorisation of coral taxa offering proxies for the vulnerability of various coral communities to said recurrent stress events (Darling et al. 2012, 2019). Quantifying the relative affinities of different coral populations for demographic recovery presents the opportunity for justifying these assumptions; helping to distinguish the species and populations most vulnerable and or resilient to projected recurrent disturbance regimes.
Equally, the amplification and attenuation characteristics of populations simultaneously reflect their stability following a disturbance. Distinguishing between the differential attributes of compensation and resistance in different populations is important for managing the future condition of biological communities (Capdevila et al. 2020). Assessments of coral communities following repeated disturbances have demonstrated divergent shifts in species compositions (Adjeroud et al. 2009; Pratchett et al. 2011, 2020). With knowledge of the capacity for various coral populations to undergo demographic amplification or for avoiding demographic attenuation, we can begin to forecast and anticipate the reassembly of coral communities, thereby improving the effectiveness of adaptive reef management. Indeed, Sandin and McNamara (2012) and Brito-Millán et al. (2019b) demonstrate how the transient characteristics of coral populations determine the resilience of their wider reef communities to algal phase shifts. Importantly, these contributions to our understanding of transient dynamics within coral environments advocate for a consideration of spatial interactions within assessments of coral population dynamics. Spatial interactions between individual corals impacts upon their fitness characteristics (Buss and Jackson 1979; Idjadi and Karlson 2007; McWilliam et al. 2018), yet the exact role these spatial interactions play in defining the performance of coral populations remains unresolved (Alvarez-Noriega et al. 2018). Ultimately, assessing the combined influence of state-structured demography and spatial interactions (e.g. Adler et al. 2010; Chu and Adler 2015) upon the performance, and resilience, of coral populations represents an important and worthwhile endeavour.
Alternatively, a disturbance does not necessarily imply a negative impact, merely a change to a system (Pickett and White 1985). Disturbances to the structure of populations can be brought about by the cessation of a continuous pressure (e.g. Götz et al. 2008), changes in resource availability (e.g. Goldstein and Steiner 2020), or their introduction into a novel environment (Jelbert et al. 2019). With the ability of natural populations for undergoing demographic compensation underpinning their capacity for exploiting changes in their local environment (Jelbert et al. 2019), the transient dynamics of populations can offer insight into their invasive potential (Iles et al. 2016). Demographic compensation mediates the persistence of coral populations established along coastlines characterised by increased abiotic variability (Cant et al. 2021a, b). Predicting and quantifying demographic compensation within coral populations will, therefore, aid in forecasting the winners and losers in reef communities targeted by conservation and restoration initiatives, or those undergoing climate induced range shifts. To illustrate this potential, we provide an example evaluating the performance of a subtropical coral population in Eastern Australia (Box 4).
Describing how size mediated patterns in survival, growth, fragmentation, and recruitment, determine the transition of individual colonies during the time interval t to t + 1, we used an IPM to evaluate the condition and viability of a Pocillopora aliciae population. Repeated surveys of individually tagged colonies were used to document the survival, growth, fragmentation, and recruitment characteristics of individuals colonies, and how these patterns are influenced by colony size. By condensing these individual-level patterns into a probability density kernel our IPM subsequently translates the transition of P. aliciae individuals across a continuous size spectrum into measures of both asymptotic and transient population characteristics
The asymptotic and transient dynamics of Pocillopora aliciae within the Solitary Islands forecast very different outlooks for this subtropical population. (a) Estimates of long-term population growth correspond with a rapid temporal decline in population size; a trajectory that persists irrespective of population structure, as illustrated by comparisons between forecasts involving a population in which individuals are distributed evenly across the size spectra (black), a population with a structural bias towards larger sized individuals (red; >80% of individuals larger than median size), and a population with a structural bias towards smaller sized individuals (blue; >80% of individuals smaller than median size). (b) Meanwhile, the transient characteristics of the population indicate it to possess the capacity for elevating its growth rate, relative to long-term expectations (λ, dotted line), following a disturbance. However, the magnitude of this increase is dependent on initial population structure, with the increased prevalence of smaller-sized colonies reducing population reactivity (\(\overline{\rho }\)), and driving population attenuation (diminished growth rate relative to λ). Across both panels shaded regions correspond with the confidence bounds associated with each projection
Complexities in the modelling approaches used to explore the dynamics of natural populations have resulted in these techniques remaining largely overlooked within coral research (Edmunds et al. 2014). Indeed, parametrising the demographic models needed to quantify population characteristics requires considerable amounts of data (Ellner et al. 2002). Although, despite the data demanding nature of state-structured demographic models, the collection of the necessary data regarding the survival, transitions in size, fragmentation, and recruitment, of individual coral colonies is possible alongside current reef monitoring efforts (Edmunds and Riegl 2020). Furthermore, new techniques for analysing the demographic characteristics of populations structured by a continuous state variable like size (Integral Projection Models; Easterling et al. 2000) have reduced the data demands of demographic assessments (Ramula et al. 2009). As a result, IPM frameworks are growing in popularity throughout coral research (see Bruno et al. 2011; Madin et al. 2012; Zychaluk et al. 2012; Elahi et al. 2016; Kayal et al. 2018; Precoda et al. 2018; Lord et al. 2020; Cant et al. 2021c; Carlot et al. 2021). Finally, studying the dynamics, particularly the transient characteristics, of coral populations is made increasingly accessible by the development of demographic analysis packages such as popdemo (Stott et al. 2012) within open-source programs like R (R Core Team 2019).
Forecasting the resilience of coral populations presents a major challenge for the future conservation of various reef communities (Lam et al. 2020). Currently, we lack the mechanistic insight into the resilience and response of coral populations to disturbance necessary for predicting the future reassembly and persistence of coral communities given increasingly recurrent disturbances. It is not our intention to dismiss previous efforts to evaluate the dynamics of coral populations using asymptotic characteristics. The asymptotic characteristics of populations present a useful tool in exploring the relative tolerances of differing populations (Beissinger and Westphal 1998) and have proven valuable in the management of threatened populations (Johnson et al. 2010). We have illustrated, however, that for evaluating and forecasting the responses of coral assemblages to disturbance and changing environments, asymptotic characteristics do not represent measures of population resilience (Hastings et al. 2018). Instead, we advocate for greater a focus towards investigating the transient dynamics of coral populations that more accurately reflect their dynamics within natural environments (Ezard et al. 2010). Evaluating coral reef community resilience using the framework of transient demographic theory will provide a new effective lens on the temporal and spatial nature of coral reef resilience, a view that will transcend current approaches of conducting single reef assessments to determine resilience (Maynard et al. 2015; McLeod et al. 2021).
Change history
08 June 2022
A Correction to this paper has been published: https://doi.org/10.1007/s00338-022-02274-3
References
Adjeroud M, Kayal M, Penin L (2017) Importance of recruitment processes in the dynamics and resilience of coral reef assemblages. In: Rossi S, Bramanti L, Gori A, Orejas C (eds) Marine animal forests: the ecology of benthic biodiversity hotspots. Springer International Publishing, Cham, pp 549–569
Adjeroud M, Michonneau F, Edmunds PJ, Chancerelle Y, de Loma TL, Penin L, Thibaut L, Vidal-Dupiol J, Salvat B, Galzin R (2009) Recurrent disturbances, recovery trajectories, and resilience of coral assemblages on a South Central Pacific reef. Coral Reefs 28:775–780
Adler PB, Ellner SP, Levine JM (2010) Coexistence of perennial plants: An embarrassment of niches. Ecol Lett 13:1019–1029
Alvarez-Noriega M, Baird AH, Dornelas M, Madin JS, Connolly SR (2018) Negligible effect of competition on coral colony growth. Ecology 99:1347–1356
Anderson KD, Pratchett MS (2014) Variation in size-frequency distributions of branching corals between a tropical versus sub-tropical reef. Mar Ecol Progress Ser 502:117–128
Bak RPM, Meesters EH (1998) Coral population structure: the hidden information of colony size-frequency distributions. Mar Ecol Progress Ser 162:301–306
Bak RPM, Meesters EH (1999) Population structure as a response of coral communities to global change. Am Zool 39:56–65
Beissinger SR, Westphal MI (1998) On the use of demographic models of population viability in endangered species management. J Wildl Manag 62:821–841
Bellwood DR, Hughes TP, Folke C, Nyström M (2004) Confronting the coral reef crisis. Nature 429:827–833
Benton TG, Grant A (1996) How to keep fit in the real world: elasticity analyses and selection pressures on life histories in a variable environment. Am Nat 147:115–139
Benton TG, Plaistow SJ, Coulson TN (2006) Complex population dynamics and complex causation: devils, details and demography. Proc R Soc B 273:1173–1181
Bierzychudek P (1999) Looking backwards: assessing the projections of a transition matrix model. Ecol Appl 9:1278–1287
Boyce MS, Haridas CV, Lee CT, Boggs CL, Bruna EM, Coulson T, Doak D, Drake JM, Gaillard JM, Horvitz CC, Kalisz S, Kendall BE, Knight T, Mastrandrea M, Menges ES, Morris WF, Pfister CA, Tuljapurkar SD (2006) Demography in an increasingly variable world. Trends Ecol Evol 21:141–148
Brito-Millán M, Vermeji MJA, Alcantar EA, Sandin S (2019a) Coral reef assessments based on cover alone mask active dynamics of coral communities. Mar Ecol Prog Ser 630:55–68
Brito-Millán M, Werner BT, Sandin SA, McNamara DE (2019b) Influence of aggregation on benthic coral reef spatio-temporal dynamics. R Soc Open Sci 6:1–14
Bruno JF, Ellner SP, Vu I, Kim K, Harvell CD (2011) Impacts of aspergillosis on sea fan coral demography: modeling a moving target. Ecol Monogr 81:123–139
Buss LW, Jackson JBC (1979) Competitive networks: Nontransitive competitive relationships in cryptic coral reef environments. Am Nat 113:223–234
Cant J, Cook K, Reimer JD, Mezaki T, Nakamura M, O’Flaherty C, Salguero-Gómez R, Beger M (2021a) Transient amplification enhances the persistence of tropicalising coral populations in marginal high latitude environments. BioRxiv 2021a.04.23.441133
Cant J, Reimer JD, Sommer B, Cook K, Kim SW, Sims CA, Mezaki T, O’Flaherty C, Brooks M, Malcolm HA, Pandolfi JM, Salguero-Gómez R, Beger M (2021b) Coral assemblages at higher latitudes favour short-term potential over long-term performance. BioRxiv
Cant J, Salguero-Gómez R, Kim SW, Sims CA, Sommer B, Brooks M, Malcolm HA, Pandolfi JM, Beger M (2021c) The projected degradation of subtropical coral assemblages by recurrent thermal stress. J Anim Ecol 90:233–247
Capdevila P, Stott I, Beger M, Salguero-Gómez R (2020) Towards a comparative framework of demographic resilience. Trends Ecol Evol 35:776–786
Carlot J, Kayal M, Lenihan HS, Brandl SJ, Casey JM, Adjeroud M, Cardini U, Merciere A, Espiau B, Barneche DR, Rovere A, Hédouin L, Parravicini V (2021) Juvenile corals underpin coral reef carbonate production after disturbance. Glob Change Biol 27:2623–2632
Caswell H (2001) Matrix population models: construction, analysis and interpretation. Oxford University Press Inc., Oxford
Caswell H (2019) Sensitivity analysis: matrix methods in demography and ecology. Springer, Cham
Caswell H, de Vries C, Hartemink N, Roth G, van Daalen SF (2018) Age x stage-classified demographic analysis: a comprehensive approach. Ecol Monogr 88:560–584
Chu C, Adler PB (2015) Large niche differences emerge at the recruitment stage to stabilize grassland coexistence. Ecol Monogr 85:373–392
Darling ES, Alvarez-Filip L, Oliver TA, Mcclanahan TR, Côté IM (2012) Evaluating life-history strategies of reef corals from species traits. Ecol Lett 15:1378–1386
Darling ES, McClanahan TR, Maina J, Gurney GG, Graham NAJ, Januchowski-Hartley F, Cinner JE, Mora C, Hicks CC, Maire E, Puotinen M, Skirving WJ, Adjeroud M, Ahmadia G, Arthur R, Bauman AG, Beger M, Berumen ML, Bigot L, Bouwmeester J, Brenier A, Bridge TCL, Brown E, Campbell SJ, Cannon S, Cauvin B, Chen CA, Claudet J, Denis V, Donner S, Estradivari FN, Feary DA, Fenner D, Fox H, Franklin EC, Friedlander A, Gilmour J, Goiran C, Guest J, Hobbs JA, Hoey AS, Houk P, Johnson S, Jupiter SD, Kayal M, Kuo C, Lamb J, Lee MAC, Low J, Muthiga N, Muttaqin E, Nand Y, Nash KL, Nedlic O, Pandolfi JM, Pardede S, Patankar V, Penin L, Ribas-Deulofeu L, Richards Z, Roberts TE, Rodgers KS, Safuan C, Sala E, Shedrawi G, Sin T, Smallhorn-West P, Smith JE, Sommer B, Steinberg PD, Sutthacheep M, Tan C, Williams GJ, Wilson S, Yeemin T, Bruno JF, Fortin M, Krkosek M, Mouillot D (2019) Social–environmental drivers inform strategic management of coral reefs in the Anthropocene. Nature Ecol Evol 3:1341–1350
Dietzel A, Bode M, Connolly SR, Hughes TP (2020) Long-term shifts in the colony size structure of coral populations along the Great Barrier Reef. Proc R Soc B 287:1–9
Easterling MR, Ellner SP, Dixon PM (2000) Size-specific sensitivity: applying a new structured population model. Ecology 81:694–708
Edmunds PJ (2015) A quarter-century demographic analysis of the Caribbean coral, Orbicella annularis, and projections of population size over the next century. Limnol Oceanogr 60:840–855
Edmunds PJ (2020) Vital rates of small reef corals are associated with variation in climate. Limnol Oceanogr 66:901–913
Edmunds PJ, Burgess SC, Putnam HM, Baskett ML, Bramanti L, Fabina NS, Han X, Lesser MP, Madin JS, Wall CB, Yost DM, Gates RD (2014) Evaluating the causal basis of ecological success within the scleractinia: an integral projection model approach. Mar Biol 161:2719–2734
Edmunds PJ, Riegl B (2020) Urgent need for coral demography in a world where corals are disappearing. Mar Ecol Prog Ser 635:233–242
Ehrlén J, Morris WF, von Euler T, Dahlgren JP (2016) Advancing environmentally explicit structured population models of plants. J Ecol 104:292–305
Elahi R, Sebens KP, de Leo GA (2016) Ocean warming and the demography of declines in coral body size. Mar Ecol Prog Ser 560:147–158
Ellner SP, Fieberg J, Ludwig D, Wilcox C (2002) Precision of population viability analysis. Conserv Biol 16:258–261
Ellner SP, Rees M (2007) Stochastic stable population growth in integral projection models: theory and application. J Math Biol 54:227–256
Engen S, Bakke Ø, Islam A (1998) Demographic and environmental stochasticity—concepts and definitions. Biometrics 54:840–846
Engen S, Saether B (1998) Stochastic population models: some concepts, definitions and results. Oikos 83:345–352
Ezard THG, Bullock JM, Dalgleish HJ, Millon A, Pelletier F, Ozgul A, Koons DN (2010) Matrix models for a changeable world: the importance of transient dynamics in population management. J Appl Ecol 47:515–523
Foster KA, Foster G (2013) Demography and population dynamics of massive coral communities in adjacent high latitude regions (United Arab Emirates). PLoS ONE 8:1–16
Foster KA, Foster G (2018) Demographics and population dynamics project the future of hard coral assemblages in Little Cayman. Open J Mar Sci 8:196–213
Goldstein J, Steiner UK (2020) Ecological drivers of jellyfish blooms—the complex life history of a ‘well-known’ medusa (Aurelia aurita). J Anim Ecol 89:910–920
Götz A, Kerwath SE, Attwood CG, Sauer WHH (2008) Effects of fishing on population structure and life history of roman Chrysoblephus laticeps (Sparidae). Mar Ecol Prog Ser 362:245–259
Hastings A (2001) Transient dynamics and persistence of ecological systems. Ecol Lett 4:215–220
Hastings A (2004) Transients: the key to long-term ecological understanding? Trends Ecol Evol 19:39–45
Hastings A (2010) Timescales, dynamics, and ecological understanding. Ecology 91:3471–3480
Hastings A, Abbott KC, Cuddington K, Francis T, Gellner G, Lai YC, Morozov A, Petrovskii S, Scranton K, Zeeman ML (2018) Transient phenomena in ecology. Science 361:1–9
Hodgson D, McDonald JL, Hosken DJ (2015) What do you mean, “resilient”? Trends Ecol Evol 30:503–506
Holling CS (1973) Resilience and stability of ecological systems. Annu Rev Ecol Syst 4:1–23
Hughes TP (1984) Population dynamics based on individual size rather than age: a general model with a reef coral example. Am Nat 123:778–795
Hughes TP (1996) Demographic approaches to community dynamics: a coral reef example. Ecology 77:2256–2260
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM, Baird AH, Baum JK, Berumen ML, Bridge TC, Claar DC, Eakin CM, Gilmour JP, Graham NAJ, Harrison H, Hobbs JA, Hoey AS, Hoogenboom M, Lowe RJ, Mcculloch MT, Pandolfi JM, Pratchett M, Schoepf V, Torda G, Wilson SK (2018) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359:80–83
Hughes TP, Jackson JBC (1980) Do corals lie about their age? Some demographic consequences of partial mortality, fission, and fusion. Science 209:713–715
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TC, Butler IR, Byrne M, Cantin NE, Comeau S, Connolly SR, Cumming GS, Dalton SJ, Diaz-Pulido G, Eakin CM, Figueira WF, Gilmour JP, Harrison HB, Heron SF, Hoey AS, Hobbs JA, Hoogenboom MO, Kennedy EV, Kuo CY, Lough JM, Lowe RJ, Liu G, McCulloch MT, Malcolm HA, McWilliam MJ, Pandolfi JM, Pears RJ, Pratchett MS, Schoepf V, Simpson T, Skirving WJ, Sommer B, Torda G, Wachenfeld DR, Willis BL, Wilson SK (2017) Global warming and recurrent mass bleaching of corals. Nature 543:373–376
Hughes TP, Tanner JE (2000) Recruitment failure, life histories, and long-term decline of caribbean corals. Ecology 81:2250–2263
Idjadi JA, Karlson RH (2007) Spatial arrangement of competitors influences coexistence of reef-building corals. Ecology 88:2449–2454
Iles DT, Salguero-Gómez R, Adler PB, Koons DN (2016) Linking transient dynamics and life history to biological invasion success. J Ecol 104:399–408
Jelbert K, Buss D, McDonald J, Townley S, Franco M, Stott I, Jones O, Salguero-Gómez R, Buckley Y, Knight T, Silk M, Sargent F, Rolph S, Wilson P, Hodgson D (2019) Demographic amplification is a predictor of invasiveness among plants. Nat Commun 10:1–6
Johnson HE, Mills LS, Stephenson TR (2010) Population-specific vital rate contributions influence management of an endangered ungulate. Ecol Appl 20:1753–1765
Jongejans E, Skarpaas O, Shea K (2008) Dispersal, demography and spatial population models for conservation and control management. Perspect Plant Ecol Evol Syst 9:153–170
Kayal M, Lenihan HS, Brooks AJ, Holbrook SJ, Schmitt RJ, Kendall BE (2018) Predicting coral community recovery using multi-species population dynamics models. Ecol Lett 22:605–615
Kim WS, Sampayo ME, Sims AC, Sommer B, Gómez-Cabrera MC, Dalton SJ, Beger M, Malcolm HA, Ferrari R, Fraser N, Figueira W, Smith SDA, Heron SF, Baird AH, Byrne M, Eakin CM, Edgar R, Hughes TP, Kyriacou N, Liu G, Matis PA, Skirving WJ, Pandolfi JM (2019) Refugia under threat: mass bleaching of coral assemblages in high-latitude eastern Australia. Glob Change Biol 25:3918–3931
Kodera SM, Edwards CB, Petrovic V, Pedersen NE, Eynaud Y, Sandin SA (2020) Quantifying life history demographics of the scleractinian coral genus Pocillopora at Palmyra Atoll. Coral Reefs 39:1091–1105
Koons DN, Grand JB, Zinner B, Rockwell RF (2005) Transient population dynamics: relations to life history and initial population state. Ecol Model 185:283–297
Lachs L, Sommer B, Cant J, Hodge JM, Pandolfi JM, Beger M (2021) Linking population size structure, heat stress and bleaching responses in a subtropical endemic coral. Coral Reefs 40:770–790
Lam VYY, Doropoulos C, Bozec YM, Mumby PJ (2020) Resilience concepts and their application to coral reefs. Front Ecol Evol 8:1–14
Lande R, Engen S, Sther BE (2003) Stochastic population dynamics in ecology and conservation. Oxford University Press, Oxford
Lefkovitch LP (1965) The study of population growth in organisms grouped by stages. Biometrics 21:1
Leslie PH (1945) On the use of matrices in certain population mathematics. Biometrika 33:183–212
Lewontin RC, Cohen D (1969) On population growth in a randomly varying environment. PNAS 62:1056–1060
Linares C, Doak DF, Coma R, Diaz D, Zabala M (2007) Life history and viability of a long-lived marine invertebrate: the octocoral Paramuricea clavata. Ecology 88:918–928
Lord KS, Lesneski KC, Bengtsson ZA, Kuhn KM, Madin J, Cheung B, Ewa R, Taylor JF, Burmester EM, Morey J, Kaufman L, Finnerty JR (2020) Multi-year viability of a reef coral population living on mangrove roots suggests an important role for mangroves in the broader habitat mosaic of corals. Front Mar Sci 7:1–16
Mace GM, Collar NJ, Gaston KJ, Hilton-Taylor C, Akçakaya HR, Leader-Williams N, Milner-Gulland EJ, Stuart SN (2008) Quantification of extinction risk: IUCN’s system for classifying threatened species. Conserv Biol 22:1424–1442
Madin JS, Baird AH, Baskett ML, Connolly SR, Dornelas MA (2020) Partitioning colony size variation into growth and partial mortality. Biol Let 16:1–5
Madin JS, Hughes TP, Connolly SR (2012) Calcification, storm damage and population resilience of tabular corals under climate change. PLoS ONE 7:1–10
Maynard JA, McKagan S, Raymundo L, Johnson S, Ahmadia GN, Johnston L, Houk P, Williams GJ, Kendall M, Heron SF, van Hooidonk R, Mcleod E, Tracey D, Planes S (2015) Assessing relative resilience potential of coral reefs to inform management. Biol Cons 192:109–119
McDonald JL, Franco M, Townley S, Ezard THG, Jelbert K, Hodgson DJ (2017) Divergent demographic strategies of plants in variable environments. Nature Ecol Evol 1:1–6
McDonald JL, Stott I, Townley S, Hodgson DJ (2016) Transients drive the demographic dynamics of plant populations in variable environments. J Ecol 104:306–314
Mcleod E, Anthony KRN, Mumby PJ, Maynard J, Beeden R, Graham NAJ, Heron SF, Hoegh-Guldberg O, Jupiter S, MacGowan P, Mangubhai S, Marshall N, Marshall PA, McClanahan TR, Mcleod K, Nyström M, Obura D, Parker B, Possingham HP, Salm R, v., Tamelander J, (2019) The future of resilience-based management in coral reef ecosystems. J Environ Manag 233:291–301
McLeod E, Shaver EC, Beger M, Koss J, Grimsditch G (2021) Using resilience assessments to inform the management and conservation of coral reef ecosystems. J Environ Manag 277:1–8
McWilliam M, Chase TJ, Hoogenboom MO (2018) Neighbor diversity regulates the productivity of coral assemblages. Curr Biol 28:3634–3639
Mizerek TL, Madin JS, Benzoni F, Huang D, Luiz OJ, Mera H, Schmidt-Roach S, Smith SDA, Sommer B, Baird AH (2021) No evidence for tropicalization of coral assemblages in a subtropical climate change hot spot. Coral Reefs 40:1451–1461
Morozov A, Abbott K, Cuddington K, Francis T, Gellner G, Hastings A, Lai YC, Petrovskii S, Scranton K, Zeeman ML (2020) Long transients in ecology: theory and applications. Phys Life Rev 32:1–40
Pickett STA, White PS (1985) The ecology of natural disturbance and patch dynamics. Academic Press Inc., San Diego, CA
Pisapia C, Edmunds PJ, Moeller HV, Riegl BM, McWilliam M, Wells CD, Pratchett MS (2020) Projected shifts in coral size structure in the Anthropocene. In: Pisapia C (ed) Advances in marine biology. Academic Press, London, pp 31–60
Pratchett MS, McWilliam MJ, Riegl B (2020) Contrasting shifts in coral assemblages with increasing disturbances. Coral Reefs 39:783–793
Pratchett MS, Trapon M, Berumen ML, Chong-Seng K (2011) Recent disturbances augment community shifts in coral assemblages in Moorea, French Polynesia. Coral Reefs 30:183–193
Precoda K, Baird AH, Madsen A, Mizerek T, Sommer B, Su SN, Madin JS (2018) How does a widespread reef coral maintain a population in an isolated environment? Mar Ecol Prog Ser 594:85–94
R Core Team (2019) R: a language and environment for statistical computing. R Core Team, Vienna
Ramula S, Rees M, Buckley YM (2009) Integral projection models perform better for small demographic data sets than matrix population models: a case study of two perennial herbs. J Appl Ecol 46:1048–1053
Riegl B, Cavalcante G, Bauman AG, Feary DA, Steiner S, Purkis S (2017) Demographic mechanisms of reef coral species winnowing from communities under increased environmental stress. Front Mar Sci 4:1–16
Riegl B, Johnston M, Purkis S, Howells E, Burt J, Steiner SCC, Sheppard CRC, Bauman A (2018) Population collapse dynamics in Acropora downingi, an Arabian/Persian Gulf ecosystem-engineering coral, linked to rising temperature. Glob Change Biol 24:2447–2462
Riegl BM (2020) The population dynamics of the coral reef crisis—Prologue. In: Riegl BM (eds) Population dynamics of the reef crisis. Academic Press, pp xxxvii–xi
Riegl BM, Glynn PW (2020) Population dynamics of the reef crisis: consequences of the growing human population. Adv Mar Biol 87:1–30
Salguero-Gómez R, Casper BB (2010) Keeping plant shrinkage in the demographic loop. J Ecol 98:312–323
Salguero-Gómez R, Shefferson RP, Hutchings MJ (2013) Plants do not count... or do they? New perspectives on the universality of senescence. J Ecol 101:545–554
Sandin SA, McNamara DE (2012) Spatial dynamics of benthic competition on coral reefs. Oecologia 168:1079–1090
Schmidt-Roach S, Miller KJ, Andreakis N (2013) Pocillopora aliciae: a new species of scleractinian coral (Scleractinia, Pocilloporidae) from subtropical Eastern Australia. Zootaxa 3626:576–582
Shlesinger T, van Woesik R (2021) Different population trajectories of two reef-building corals with similar life-history traits. J Anim Ecol 90:1379–1389
Sommer B, Harrison PL, Beger M, Pandolfi JM (2014) Trait-mediated environmental filtering drives assembly at biogeographic transition zones. Ecology 95:1000–1009
Stott I (2016) Perturbation analysis of transient population dynamics using matrix projection models. Methods Ecol Evol 7:666–678
Stott I, Franco M, Carslake D, Townley S, Hodgson D (2010) Boom or bust? A comparative analysis of transient population dynamics in plants. J Ecol 98:302–311
Stott I, Hodgson DJ, Townley S (2012) Popdemo: an R package for population demography using projection matrix analysis. Methods Ecol Evol 3:797–802
Stott I, Townley S, Hodgson D (2011) A framework for studying transient dynamics of population projection matrix models. Ecol Lett 14:959–970
Sully S, Burkepile DE, Donovan MK, Hodgson G, van Woesik R (2019) A global analysis of coral bleaching over the past two decades. Nat Commun 10:1–5
van Tienderen PH (2000) Elasticities and the link between demographic and evolutionary dynamics. Ecology 81:666–679
Townley S, Carslake D, Kellie-Smith O, Mccarthy D, Hodgson D (2007) Predicting transient amplification in perturbed ecological systems. J Appl Ecol 44:1243–1251
Townley S, Hodgson DJ (2008) Erratum et addendum: transient amplification and attenuation in stage-structured population dynamics. J Appl Ecol 45:1836–1839
Tuljapurkar S (1989) An uncertain life: demography in random environments. Theor Popul Biol 35:227–294
Tuljapurkar SD, Orzack SH (1980) Population dynamics in variable environments I. Long-run growth rates and extinction. Theor Popul Biol 18:314–342
van Groenendael J, de Kroon H, Caswell H (1988) Projection matrices in population biology. Trends Ecol Evol 3:264–269
Williams JL, Ellis MM, Bricker MC, Brodie JF, Parsons EW (2011) Distance to stable stage distribution in plant populations and implications for near-term population projections. J Ecol 99:1171–1178
Wolff NH, Mumby PJ, Devlin M, Anthony KRN (2018) Vulnerability of the Great Barrier Reef to climate change and local pressures. Glob Change Biol 24:1978–1991
Zychaluk K, Bruno JF, Clancy D, McClanahan TR, Spencer M (2012) Data-driven models for regional coral-reef dynamics. Ecol Lett 15:151–158
Acknowledgements
The authors would like to thank D. Ceccarelli for her comments on early drafts of this manuscript. This work was supported by a NERC DTP scholarship to JC and a NERC Independent Research Fellowship (NE/M018458/1) to RS-G.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Stuart Sandin
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cant, J., Salguero-Gómez, R. & Beger, M. Transient demographic approaches can drastically expand the toolbox of coral reef science. Coral Reefs 41, 885–896 (2022). https://doi.org/10.1007/s00338-022-02250-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02250-x