Abstract
As climate changes increase heat stress on tropical ecosystems, the long-term persistence of coral reefs requires rapid coral recovery following coral bleaching events. Using the extent of coral cover return to a pre-bleaching baseline as a benchmark, recovery of fast-growing and stress-tolerant coral growth forms suggests that reefs can bounce back between repeated disturbances if given adequate time and protection from anthropogenic disturbances. However, long-term recovery dynamics of coral communities following severe bleaching and mass mortality are limited, particularly for fringing reefs along inhabited coastlines where human stressors may compromise recovery potential. Here, we examine the dynamics and drivers of coral recovery in Seychelles, where 12 reefs returned to pre-bleaching coral cover levels after a severe bleaching event caused > 95% coral mortality. Six reefs with initially low cover ( < 25%) recovered within 7–12 yr and, after 16 yr, exceeded pre-bleaching cover levels by 132–305%. In contrast, six reefs with initially high cover (20–60%) remained at 48–93% of pre-bleaching levels, with recovery projected to take 17–29 yr. Abiotic and historic conditions constrained recovery rates, with the slowest recovery times observed on deep and wave-exposed reefs with high pre-bleaching coral cover. Reefs with high juvenile coral densities and low nitrogen levels recovered fastest, possibly due to the interplay between nutrient enrichment, algal proliferation, and coral recruitment. Our findings emphasize the importance of understanding small-scale variation in recovery potential, whereby recovery times were governed by natural limits on growth rates and modified by coral recruitment and nutrient enrichment. Ultimately, climate-impacted reefs can recover to moderate coral cover levels, but, if bleaching causes repeated high coral mortality, short recovery windows will prevent a return to historic levels of coral dominance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate-driven thermal stress events that cause coral bleaching events are accelerating in frequency, threatening the persistence of coral-dominated reefs across the tropics (Pandolfi et al. 2003; Heron et al. 2016). As global temperatures have risen from 1980 to 2016, coral bleaching recovery windows have shortened from 27 to 5.9 yr (Hughes et al. 2018a), and are likely to become even shorter as severe bleaching events are expected to occur annually by 2050 (van Hooidonk et al. 2016). Although examples of resilient reefs that regenerate coral cover suggest that certain conditions, such as isolation from human stressors, facilitate recovery from bleaching (Sheppard et al. 2008; Gilmour et al. 2013), the conditions that promote or depress recovery rates are not well understood, particularly for fringing reefs along inhabited coastlines where chronic anthropogenic stressors are pervasive. As such, it is unclear under what conditions reefs may be able to recover rapidly in the face of diminishing recovery windows.
The ability of coral reefs to return to coral-dominated states following declines from acute disturbances, including bleaching, is typically measured by the degree of recovery towards pre-disturbance coral cover (Connell 1997; Osborne et al. 2011; Johns et al. 2014). Although differential bleaching susceptibility and recovery potential of coral growth forms (Darling et al. 2013) mean that reassembly of community composition is expected to lag behind cover recovery (Johns et al. 2014), return times to pre-disturbance coral cover (i.e. 100% recovery) may be considered an early indicator of recovery. Short return times enhance the probability of coral-dominated states under recurrent bleaching, but should also increase the potential for a return to pre-bleaching functioning (Alvarez-Filip et al. 2013). However, return times can vary considerably among reefs (Osborne et al. 2011; Johns et al. 2014) and regions (Connell 1997; Baker et al. 2008; Graham et al. 2011), implying that recovery potential is highly context dependent. As such, our understanding of plausible recovery times under recurrent bleaching scenarios requires analysis of long-term benthic changes according to local conditions of resilient reefs.
Regional and local differences in coral recovery are likely due to reef-specific abiotic and biotic conditions that are conducive to coral growth, and to degrading influences of local anthropogenic stressors. For example, high wave energy limits coral growth and larval settlement, thus placing natural constraints on coral cover (Gove et al. 2015) which likely also influence recovery times. Ecological feedbacks between corals, algae, grazers and nutrients underscore the importance of biotic processes in determining coral recovery, with coral recruitment and survival dependent on grazing control of algal competitors (McCook et al. 2001; Birrell et al. 2008), particularly under nutrient regimes that stimulate algal productivity (Burkepile and Hay 2009; Burkepile et al. 2013). These feedbacks may be disrupted by anthropogenic influences where, for example, sedimentation directly inhibits coral growth (Fabricius et al. 2005) and nitrogen enrichment promotes macroalgal overgrowth (Lapointe 1997). Overexploitation of grazers can promote algal overgrowth (Mumby et al. 2006) and magnify nutrient effects (Burkepile and Hay 2006) and so, after bleaching, may slow recovery rates by limiting coral recruitment (Elmhirst et al. 2009). These factors imply that benthic recovery will vary spatially and thus impact ecosystem functions in different ways among reefs and between regions, but that natural recovery processes could be accelerated to increase the resilience of coral reefs within expected recovery windows.
To date, empirical studies of coral recovery have typically contended with several types of disturbance on reefs that are partially protected from human stressors. For example, comparative analyses of reef recovery rates on the Great Barrier Reef (GBR) suggest that water quality (Ortiz et al. 2018; MacNeil et al. 2019) and thermal heating (Osborne et al. 2017) have compromised GBR recovery potential. The GBR has suffered severe bleaching following the 2016 El Niño (Hughes et al. 2018b), but previous studies have focused on recovery following moderate coral decline by other disturbances (e.g. cyclones, crown-of-thorns starfish), and the role of protection networks (Mellin et al. 2016). Furthermore, detection of recovery mechanisms requires reef-scale ecological metrics (e.g. coral recruitment, nutrient loads) that are often unavailable at the appropriate temporal and spatial scale.
Here, we assess the factors that promote or depress long-term coral recovery following a severe bleaching event. We utilize long-term monitoring data of reef sites in the inner Seychelles that experienced > 90% coral mortality after bleaching in 1998, where 12 reefs gradually regained coral cover and habitat complexity over 2005–2014 (Graham et al. 2015). These monitoring data identified reef-specific conditions that increased the likelihood of regime shifts to macroalgal states (Graham et al. 2015), but equivalent reef-level variation in recovery dynamics at resilient reefs has not yet been examined. We used logistic growth models in a hierarchical Bayesian modelling framework to demonstrate variability in recovery trajectories, and quantified variation in projected recovery times according to historic reef states, abiotic and biotic influences, and anthropogenic stressors.
Methods
Identification of recovering reefs
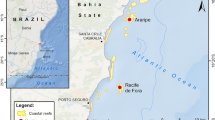
We examined the benthic recovery dynamics at 12 reef sites in the inner Seychelles (Fig. 1). Reefs were defined as recovering from the 1998 bleaching event by Graham et al. (2015), based on the relative abundance and trajectories of hard coral and macroalgae from 1994–2014. Recovering reefs had greater cover of hard corals than macroalgae, increased in hard coral cover from 2005 to 2011, and met one of the three following trajectory criteria: (1) declining Euclidean distance between pre-disturbance (1994) and post-disturbance benthic condition (2005, 2008, 2011); (2) the rate of hard coral cover increase was stable or greater than that of macroalgal cover change; (3) the decline in coral cover between 1994–2011 was lower than that of 1994–2005, and corresponding change in macroalgal cover was negligible (Graham et al. 2015). Because Seychelles reefs experienced a second mass bleaching event in 2016 (Hughes et al. 2018a), our analysis focuses on the recovery period 2005–2014.
Map of recovering reef sites on Mahe (a) and Praslin (b) in Seychelles, with Indian Ocean location (c) and change in benthic habitat composition over 2005–2014 (d, e). Points are mean percent cover of coral growth forms (d) and structural complexity (e) across all recovering reefs ( ± 2 standard errors). 1994 pre-bleaching baseline total coral cover and structural complexity indicated as dashed lines
Benthic community data
Coral cover and structural complexity estimates were collected using visual point counts. Surveys were conducted in 1994 (i.e. pre-bleaching) and, in the recovery period, every 3 yr from 2005–2014 (2005, 2008, 2011, 2014). At each reef site, one diver (SJ in 1994, SKW in all subsequent years) visually assessed benthic cover and structural complexity in point counts of 7 m radius. Percent cover of major coral growth forms (branching, massive, encrusting) was estimated in each point count, and structural complexity was visually assessed on a 6-point scale (Polunin and Roberts 1993). Point counts were repeated for eight (2011, 2014) or sixteen replicates (1994, 2005, 2008) at each reef site. Coral visual assessments were supplemented with genera-level surveys conducted in 2008, 2011 and 2014. At each reef site, we used eight replicate 10-m line intercept transects to record the percent cover of major coral genera.
Predictors of benthic recovery
We examined reef-level variation in benthic recovery dynamics using a suite of abiotic, biotic and anthropogenic covariates that are thought to influence benthic recovery dynamics. Abiotic processes place natural constraints on coral abundances (Williams et al. 2015) and, as such, may depress or promote recovery rates. For example, recovery may be compromised in shallow water locations where corals are more vulnerable to bleaching (Safaie et al. 2018). Locations subjected to high intensity wave action may have lower natural cover of fast-growing branching corals (Gove et al. 2015) that are vulnerable to breakage and dislodgement during severe storms (Madin and Connolly 2006) and, as such, may also require longer recovery periods. To measure these processes, we used UVC survey depth (5–10 m) and a long-term wave climatology metric as abiotic predictor covariates. Wave energy (Joules) estimates were derived from hourly wind speed and direction values (Seychelles National Meteorological Service) and accounted for the uninterrupted fetch distance across which waves are generated (Ekebom et al. 2003; Chollett and Mumby 2012). For each reef, fetch distances were based upon a 55-m resolution map extending to 500 km for 32 compass directions, and wind speed and direction values were averaged over 1998–2011 (Graham et al. 2015). Thus, our wave energy metric represents reef-level variation in physical exposure in the post-bleaching recovery period.
Abiotic constraints may be modified by herbivorous grazing pressure, which enhances coral recruitment by clearing larval settlement space and suppressing growth of competing turf and macroalgae (McCook et al. 2001; Mumby et al. 2006). In Seychelles, reefs with higher herbivore biomass were also less likely to transition to macroalgal states after 1998 bleaching, but it is unclear if this effect also influenced recovery of resilient reefs. We used observations of herbivorous fish biomass from diver surveys conducted at the same reef sites. Before each benthic survey conducted in 2005, one diver (NAJG) surveyed the abundance and length (cm) of 37 species of all diurnally active, reef-associated herbivorous fish (croppers, scrapers, excavators and browsers), using point counts of 7 m radius. Abundances and lengths were converted to biomass (kg ha−1) using published length ~ weight relationships (Froese and Pauly 2015), and averaged across replicates to give the mean herbivorous fish biomass at each reef. As a measure of coral recruitment rates at each reef in the recovery period, we estimated juvenile coral densities in 2011. Juveniles were corals < 10 cm in diameter, estimated using 33 × 33 cm quadrats for 8 replicates placed within each benthic point count survey.
Recovery rates may be depressed on reefs nearby to sources of anthropogenic run-off, owing to unbalanced nutrient loads which may indirectly impact coral recruitment through proliferation of algal competitors (McCook et al. 2001; Fabricius et al. 2005; D’Angelo and Wiedenmann 2014). We estimated the nitrogen concentration (%) of Sargassum fronds collected at each reef site in 2014 (Graham et al. 2015). Nitrogen concentrations reflect differences in nitrogen availability among reefs due to spatial variation in terrestrial nutrient inputs.
Finally, we used pre-disturbance surveys to account for potential unmeasured reef-level variation in the capacity of each reef to reach highly complex, coral-dominated states (hereafter ‘historic’ predictors). Furthermore, because we defined recovery as a return to pre-bleaching coral cover, we expected coral-dominated reefs to have the longest recovery times. Initial benthic conditions were the pre-bleaching estimate of hard coral cover and structural complexity (i.e. in 1994), averaged across replicates at each reef site. All predictor covariates were scaled and centred to a mean of 0 and standard deviation of 1 and examined for collinearity before model fitting.
Coral recovery models
We examined reef-level variation in hard coral recovery trajectories using a hierarchical Bayesian logistic model. Observed hard coral cover (y) was modelled as a logistic function where the recovery rate r, asymptote a_max, and the curve inflexion point xmid predict the coral cover in each post-disturbance survey year i. Survey year was rescaled to represent recovery years following 1998 bleaching (e.g. first survey year 2005 = 7 recovery years).
We fitted two logistic model parameterizations, for either a standard 3-parameter logistic model with one maximum asymptote term (1) or a more flexible 4-parameter logistic with minimum (a_min) and maximum asymptote terms (a_max) (2). Furthermore, because the logistic asymptote may be defined by the observed recovery rate or fixed to the substrate area available for coral colonization, we also fitted models with either unconstrained asymptotes (i.e. a_max is estimated by the model) or asymptotes set to the amount of hard substrate area available for colonization at each reef in 1994 (i.e. a_max = total hard coral + rock + rubble). Reef-level recovery dynamics were modelled in a hierarchical structure that allowed r, a_min, and a_max to vary by each reef site s, for gamma-Poisson-distributed observations (McElreath 2017),
with mean models:
Models were estimated by Markov chain Monte Carlo (MCMC) using the No-U-Turn-Sampler implemented in Stan, sampling for 3,000 iterations across 3 chains with warmup of 1,500. Prior values were drawn from a normal distribution with mean = 6 and standard deviation = 1 (N(6, 1)) for r, from N(3.6, 1) for a_min and a_max ( = 36% cover on linear scale), and N(−0.9, 1) for xmid. We compared model fits among logistic models (1, 2) and asymptote values (unconstrained or fixed to available substrate area) with the Widely Applicable Information Criterion (WAIC) (McElreath 2017), which supported the four-parameter logistic model with a_max fixed to the available substrate area (Electronic Supplementary Material, ESM Table S1). We ensured chain convergence by assessing trace plots, and by checking that the Gelman–Rubin diagnostic (\(\widehat{R}\)) was < 1.01 and the number of effective samples was sufficiently high (Electronic Supplementary Material, ESM Table S2). We also evaluated model fits by checking correspondence between fitted and observed values (Electronic Supplementary Material, ESM Fig. S2). Finally, we sampled posterior predictions of recovery year using a model of 7,000 iterations across 1 chain.
Reef-level variability in coral recovery
We examined coral recovery trajectories at each reef by drawing predicted cover values from the posterior distribution for each year in the recovery period 2005–2014. Observed reef-level trajectories were visualized on a common scale by rescaling each predicted coral cover value relative to its baseline (i.e. 1994), such that coral recovery was expressed as a proportion of its pre-bleaching cover (e.g. coral recovery = 100% when predicted coral cover = coral cover in 1994). We then examined variation in recovery times by identifying, for each reef, the year when mean predicted coral recovery reached the pre-bleaching baseline. For reefs that failed to recover by the last visual census (2014), we projected coral recovery trajectories forward in time until the baseline was reached.
Next, we sought to understand variation in the expected year of recovery (y_recovery) according to abiotic, biotic, and anthropogenic covariates. We fitted a Bayesian linear model to recovery year and eight fixed covariates,
with prior values drawn from the distribution N(0, 2) for fixed covariates and at the average recovery year across reefs (N(17, 5)) for the intercept. Model parameters were estimated by MCMC sampling of 7,000 iterations, with a warmup of 1,500 across 3 chains. Model convergence was assessed with posterior predictive checks, effective samples, and \(\widehat{R}\) (Electronic Supplementary Material, ESM Table S3). We further ensured that parameter effect sizes were robust to potential estimation biases caused by outlying predictor values at individual reefs. Using a jackknife resampling approach, we compared parameter posterior distributions across predictive models fitted to datasets that dropped each reef in turn (i.e. 12 models, each with n = 11). Possible overfitting of recovery year models fitted to relatively few reefs (n = 12) and a high number of predictors (8) was minimized by use of weakly uninformative covariate priors and half-Cauchy variance prior (Cauchy(0, 2)) (McNeish 2016).
We assessed covariate influences with standardized effect sizes, represented by the posterior distribution median with 95% (strong inference) and 50% (weak inference) confidence intervals, and visualized median recovery times across the observed gradient of each covariate. We also used heat maps to visualize the combined effect of any strong biotic and human covariates on median coral recovery time, thus allowing us to assess how local reef conditions might be manipulated to enhance coral recovery after bleaching.
All analyses were conducted in R 3.4.1 (R Development Core Team 2018), logistic model parameterizations were provided by SSlogis and SSfpl, and Bayesian models were fitted in Stan with the rethinking package (McElreath 2017). We provide our R code and model predictions at http://github.com/jpwrobinson/coral-bleach-recovery.
Results
Of the 12 reefs that recovered from mass bleaching in 1998, pre-bleaching coral cover averaged 26%, with seven low cover reefs (10–20%) and five high cover reefs (20–60%). In 2005, 7 yr after bleaching, mean coral cover across all 12 reefs was 11%, which steadily increased to return to mean pre-bleaching cover (27%) by 2014 (Fig. 1d). However, mean trends obscured considerable variation in reef-level recovery trajectories. In 2005, four reefs had recovered 72–127% of pre-bleaching coral cover, whereas eight reefs remained below 50% of pre-bleaching cover (Fig. 2a). From 2005 to 2014 (i.e. 7–16 yr after bleaching), reefs steadily recovered towards pre-bleaching conditions (Electronic Supplementary Material, ESM Fig. S2). By 2014, six ‘overshoot’ reefs had exceeded baseline cover by 132–305% to reach fully recovered states within 7–12 yr, while six reefs failed to recover to 1994 conditions and remained at 48–93% baseline cover, with recovery predicted to occur within 17–29 yr (Fig. 2b). Thus, despite recovery trajectories across reefs (Electronic Supplementary Material, ESM Fig. S2) and return to average pre-bleaching coral cover (Fig. 1d), time to recovery was highly variable across reefs, ranging from 7 to 29 yr. Recovery was primarily driven by branching Acropora corals rather than massive or encrusting forms (Fig. 1d; Electronic Supplementary Material, ESM Figs. S3, S4).
Reef-level variability in the extent and timing of coral recovery. a Hard coral cover as a proportion of the pre-bleaching coral cover at each reef in 1994, for overshoot (green) and failed (purple) recovery trajectories. Lines are posterior predictions of hard coral at each reef from 2005–2014 conditioned on reef-specific predictor covariates, for the median prediction (solid lines) and 100 draws (thin lines) from the posterior distribution. b Time to recovery for each reef, defined as the year at which predicted coral cover equalled pre-bleaching cover (i.e. 1994). Points are median recovery year with 95% credible intervals, with lines indicating median recovery trajectories. Corresponding percent cover trajectories are displayed in Electronic Supplementary Material, ESM Fig. S2. Recovery models assume no further mortality to corals, but we note that the 2016 mass bleaching event caused mass mortality at these reefs (Wilson et al. 2019) and therefore visualized recovery trajectories were not completed
Reef-level influences on recovery year
Recovery years were strongly associated with reef-level explanatory covariates. For abiotic and historic covariates, recovery was slowest at exposed and deep reefs with high pre-bleaching coral cover (Figs. 3, 4). For instance, recovery year was delayed by 1.8 yr for every 10% increase in historic coral cover, by 2.7 yr for every 5 m increase in depth, and by 2.4 yr for every 0.5 J hr−1 increase in wave energy (Fig. 4). Initial structural complexity, which was relatively similar among reefs (3–3.7) relative to post-bleaching habitat structure (1.9–3.2), was weakly associated with recovery year (effect size = 0.37; 95% CIs − 1.30, 2.08).
Abiotic, biotic, human, and historic influences on recovery year. Effect sizes are the median of the posterior distribution for each parameter, with 95% and 50% credible intervals drawn from 1,000 samples. Predictors were standardized to the same scale and effect sizes were robust to jackknife resampling (Electronic Supplementary Material, ESM Fig. S6)
Predicted change in recovery year across gradients in abiotic and historic conditions, for a depth b initial complexity c initial coral cover, and d wave energy. Thick lines are median posterior estimate sampled across the range of each abiotic covariate, holding all other covariate effects to their means (0), with uncertainty represented with by 100 samples from the posterior distribution (thin lines). Observed data distribution is indicated along the x-axis with points coloured by their recovery trajectory (overshoot = green, failed = purple)
Coral juvenile densities and nitrogen load were the strongest positive influences on recovery year, with recovery times minimized on reefs with low nutrient enrichment and high juvenile coral density (Figs. 3, 5) (Electronic Supplementary Material, ESM Fig. S5). After accounting for abiotic effects, recovery times < 10 yr were expected for reefs with both low nitrogen load (nitrogen < 0.7%) and high coral recruitment ( > 40 juveniles m−2) (Fig. 5). Surveyed reefs, however, did not meet these criteria, with predicted relationships primarily driven by slow recovery times of nutrient-enriched reefs with low juvenile coral densities, and by fast recovery times of one nutrient-poor reef (7 yr, 0.6% nitrogen) and one high-density juvenile coral reef (9 yr, 58.8 juveniles m−2). Despite these outliers, covariate effect sizes were not biased by observations from individual reefs (Electronic Supplementary Material, ESM Fig. S6) and abiotic and biotic covariates were uncorrelated (Electronic Supplementary Material, ESM Fig. S1), indicating that variability in recovery times was attributable to the combined effect of several covariates rather than to collinearity or the condition of outlying reefs. Recovery times were slower on protected reefs (effect size = 1.56; 95% CIs − 0.07, 3.18) and on those with abundant herbivores, with recovery extending by 4.1 yr from the lowest to highest levels of grazing biomass (42–509 kg ha−1) (Electronic Supplementary Material, ESM Fig. S5b).
Combined influence of juvenile coral density and nitrogen concentration on recovery year. Colours indicate the median recovery year across the range of each predictor covariate, holding all other influences to their means (0). The range of observed coral densities and nitrogen concentrations are represented by grey points. Note that additional abiotic and historic influences on recovery times mean that points do not necessarily correspond with predicted recovery year
Discussion
Over a decade after severe coral bleaching, recovering Seychelles reefs varied substantially in their extent of coral recovery, with six reefs returning to pre-bleaching coral cover within 7–12 yr and six reefs failing to reach pre-bleaching cover before the next major bleaching event in 2016. Projected recovery times ranged from 7–29 yr and were linked to reef-level variation in several abiotic, biotic, and anthropogenic factors. Abiotic and historic conditions placed natural constraints on recovery, with the fastest recovery times predicted for shallow reefs with low initial coral cover and low daily wave exposure. After accounting for abiotic limits, recovery times were also faster under conditions of high coral recruitment and low nitrogen enrichment, implying that improving juvenile coral survival and mitigation of nutrient run-off may enhance coral recovery.
Reef recovery was driven by regeneration of fast-growing branching Acropora corals, which on most reefs have replaced massive growth forms (Wilson et al. 2012, 2019). Such compositional turnover suggests that these reefs have not recovered their pre-bleaching functions and, for example, recovery of structural complexity was incomplete by 2014. Other ecosystem functions, such as community calcification rates, have not fully returned (Januchowski-Harty et al. 2017). Although we were unable to examine long-term abundance shifts among individual coral species, community turnover is expected to continue after pre-bleaching cover levels have been reached (Johns et al. 2014). Studies which model recovery trajectories for individual coral species will help to identify future community compositions for heat-stressed reefs (Ortiz et al. 2018). Compositional shifts towards branching Acropora corals, combined with either overshoot or failure to return to pre-bleaching coral cover, mean that Seychelles’ reefs failed to resemble pre-bleaching states after 16 yr of recovery. As bleaching events continue to accentuate boom-bust dynamics in the keystone habitat structure of coral reefs (Wilson et al. 2019), the likelihood of full recovery cycles is diminishing (Hughes et al. 2018a). In Seychelles, the 2016 coral bleaching event caused extensive coral mortality (Wilson et al. 2019), meaning the recovery dynamics we have documented in this study need to recommence.
Recovery times did, however, vary predictably with natural abiotic limits, with the slowest recovery times occurring on deep reefs with high wave exposure. Such influences likely reflect constraints on coral growth rates where, for example, light attenuation at depth slows coral growth rates (Huston 1985; Pratchett et al. 2015). However, the influence of depth on bleaching responses is multifaceted, with evidence that shallow reefs often incur great incidences of bleaching (Bridge et al. 2013) and are more likely to undergo regime shifts to macroalgae after bleaching (Graham et al. 2015). Thus, deep cool water reefs may be refuges to heat stress (Safaie et al. 2018). As such, the fast coral growth at shallow reefs may only be realized under conditions which promote coral growth (e.g. high juvenile coral density), while reduced stress in deeper waters may help reefs to retain high coral cover, at the cost of slower recovery times. For wave energy, exposure gradients determine spatial patterns in coral cover (Done 1982; Williams et al. 2015; Gove et al. 2015) and extreme events can cause physical dislodgement of coral colonies (Madin and Connolly 2006). We extend these concepts to show that wave energy may also negatively influence coral recovery rates, with reefs exposed to high daily wave action also slower to recover. Such physical constraints on coral recovery likely occurred because branching corals, which are most susceptible to removal by wave action (Madin and Connolly 2006), dominated recovery dynamics. Thus, abiotic filtering of community composition can enhance or delay reef recovery rates. For example, reefs with highest wave exposure were characterized by low levels of branching corals, and failed to reach pre-bleaching total coral cover (Mahe E Patch, 66% recovery; Ste Anne Patch, 62% recovery). Together, these predictions of abiotic constraints can guide expectations of coral recovery after bleaching events, which are particularly needed for reefs where in situ monitoring data are unavailable. For example, remote sensing of depth and wave energy could be paired with thermal stress maps and predictors of bleaching vulnerability (e.g. Safaie et al. 2018) to forecast long-term resilience to heat stress across large spatial scales.
Coral recruitment and nutrient enrichment exceeded abiotic limits on coral recovery times, likely due to their opposing influences on competition between calcifiers and algal taxa. High rates of coral recruitment (i.e. juvenile coral densities) should positively correlate with future adult coral abundances (Birrell et al. 2008) and thus shorten recovery times. In Seychelles, reef-level variation in coral recruitment appears to be driven by benthic habitat properties rather than larval supply. Survivorship of juvenile corals to adults is lower on unstable rubble reefs that are frequently disturbed by wave action (Chong-Seng et al. 2014), and juvenile coral recovery from severe bleaching is moderated by habitat complexity (Dajka et al. 2019). Nitrogen enrichment, however, may have had an additional, indirect influence on recovery times by stimulating algal growth. Nutrients may exert bottom-up control of benthic composition through animal excretion (Graham et al. 2018), seasonal and upwelling sources (Williams et al. 2015), and by anthropogenic-driven eutrophication (Fabricius et al. 2005). Our results demonstrate that high nutrient loads attributable to terrestrial run-off also slowed coral recovery, which is consistent with previous observations that low C/N ratios (i.e. high nitrogen concentrations) increased the probability of a macroalgal regime shift on Seychelles reefs (Graham et al. 2015). Indeed, juvenile coral densities decreased with increasing nutrient enrichment at 10 of 12 reefs (though were uncorrelated in our predictive models), possibly because high nutrient loads inhibit coral recruitment (Koop et al. 2001) and promote growth of competing algal organisms (Burkepile and Hay 2009), which likely combine to raise post-recruitment coral mortality (Chong-Seng et al. 2014). Nutrient concentrations can also benefit coral growth, though this is contingent on high herbivory levels (Burkepile and Hay 2006) and the ratio of nitrogen to phosphorus (D’Angelo and Wiedenmann 2014). Given that several nutrient-based mechanisms potentially underpin considerable reef-level variability in recovery times, experimental investigation of the relationships between coral recruitment, nutrient enrichment, and bleaching recovery is urgently needed. Such research will help to unravel biotic feedbacks and thus identify conditions that accelerate coral recovery.
Local biotic and anthropogenic influences on recovery times should anchor expectations according to abiotic constraints and reveal potential processes that might be manipulated to enhance recovery after bleaching. Across regions, large-scale oceanographic influences such as temperature and productivity constrain natural baselines of coral cover (Williams et al. 2015) and so may contribute to regional differences in recovery times (Baker et al. 2008; Graham et al. 2011). At smaller spatial scales, biotic processes that promote coral recruitment may be enhanced by manipulation of ecological feedbacks to enhance coral recovery (Nyström et al. 2012; Ladd et al. 2018), such as reducing terrestrial run-off. However, the influences of nutrient enrichment on coral condition are complex, owing to confounding sources from terrestrial inputs (Fabricius et al. 2005), upwellings and animal excretion (Graham et al. 2018), variable impacts according to the type and balance of nutrients (D’Angelo and Wiedenmann 2014), and uncertainties surrounding nutrient cycling and their long-term persistence in seawater (Fabricius et al. 2005). Isolation of nutrient pathways which enhance coral growth and recruitment is a critical avenue for further research.
Herbivore biomass had a weak, positive effect on recovery year, contradicting expectations that high grazing pressure will enhance recovery through top-down control of algae. Given that reefs with herbivore biomass > 177 kg ha−1 were less likely to regime shift in Seychelles (Graham et al. 2015), grazing pressure may be a relatively weak influence on coral recovery as herbivore biomass has increased on Seychelles reefs since the 1998 bleaching event (Robinson et al. 2019), and thus thresholds that prevent macroalgal overgrowth have been exceeded on many reefs. Alternatively, because grazing effects on benthic communities are tightly linked to the size structure and functional composition of herbivore assemblages (Nash et al. 2015; Steneck et al. 2018), grazing influences on coral recovery may not be detectable with coarse biomass metrics which combine distinct functional groups. The longer recovery times on reefs protected from fishing was also unexpected. Both herbivore and management effects may, however, be somewhat confounded by pre-bleaching coral cover. Compared to fished reefs, protected reefs supported 65 kg ha−1 greater herbivore biomass and 15.5% higher pre-bleaching coral cover, meaning that protected reefs require longer recovery times to reach coral-dominated states. Extending our recovery analyses to other regions will help resolve uncertainties around management and herbivory effects.
Recovery times from severe bleaching events inform expectations for the long-term persistence of coral-dominated reefs in a warming climate. Here, similar recovery trajectories meant that reefs that recovered before the next mass bleaching event (i.e. within 16 yr) were those with pre-bleaching cover < 25%. Reefs that failed to recover were generally those with exceptional coral cover ( > 30%). Such patterns are consistent with evidence that recurring mass bleaching events, particularly those that occur within 16 yr of each other and cause severe coral mortality, will prevent coral reefs returning to historic coral-dominated states (Birkeland 2004; Hughes et al. 2018a). Furthermore, given that pre-bleaching cover was highly variable (11–55%), our findings underscore the uncertainty associated with using historic conditions as a recovery benchmark. For example, pre-bleaching disturbances may have limited potential coral cover, which would explain why overshoot reefs were able to far exceed their baseline cover after bleaching.
Our long-term analysis of coral recovery dynamics uncovered substantial reef-scale variability in recovery times after mass bleaching, whereby reefs either failed to recover or exceeded their baseline state and recovery ranged from 7 to 29 (projected) years. The number of recovery years was strongly constrained by abiotic conditions (depth, wave energy) and pre-bleaching coral cover, indicating that predictions of bleaching recovery times can be informed by abiotic and historic conditions at the scale of individual reefs. The strongest influences on recovery times were post-bleaching juvenile coral densities and nitrogen concentrations, suggesting that recovery might be enhanced by limiting nutrient run-off and promoting coral recruitment and survivorship. However, coral-dominated reefs are unlikely to persist under recurrent bleaching events that cause extensive coral mortality, if recovery windows from such severe bleaching shorten to less than 10 yr (Hughes et al. 2018a). Further investigation of feedbacks between nutrients, algal growth, and coral recruitment is necessary to understand how manipulation of biotic processes can accelerate reef recovery after climate-driven bleaching events.
References
Alvarez-Filip L, Carricart-Ganivet JP, Horta-Puga G, Iglesias-Prieto R (2013) Shifts in coral-assemblage composition do not ensure persistence of reef functionality. Sci Rep 3:3486
Baker A, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: an ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:435
Birkeland C (2004) Ratcheting down the coral reefs. Bioscience 54:1021–1027
Birrell CL, McCook LJ, Willis BL, Diaz-Pulido GA (2008) Effects of benthic algae on the replenishment of corals and the implications for the resilience of coral reefs. Oceanogr Mar Biol Annu Rev 46:41
Bridge TCL, Hughes TP, Guinotte JM, Bongaerts P (2013) Call to protect all coral reefs. Nat Clim Chang 3:528
Burkepile DE, Hay ME (2006) Herbivore vs. nutrient control of marine primary producers: context-dependent effects. Ecology 87:3128–3139
Burkepile DE, Hay ME (2009) Nutrient versus herbivore control of macroalgal community development and coral growth on a Caribbean reef. Mar Ecol Prog Ser 389:71–84
Burkepile DE, Allgeier JE, Shantz AA, Pritchard CE, Lemoine NP, Bhatti LH, Layman CA (2013) Nutrient supply from fishes facilitates macroalgae and suppresses corals in a Caribbean coral reef ecosystem. Sci Rep 3:1493
Chollett I, Mumby PJ (2012) Predicting the distribution of Montastraea reefs using wave exposure. Coral Reefs 31:493–503
Chong-Seng KM, Graham NAJ, Pratchett MS (2014) Bottlenecks to coral recovery in the Seychelles. Coral Reefs 33:449–461
Connell JH (1997) Disturbance and recovery of coral assemblages. Coral Reefs 16:S101–S113
D’Angelo C, Wiedenmann J (2014) Impacts of nutrient enrichment on coral reefs: new perspectives and implications for coastal management and reef survival. Current Opinion in Environmental Sustainability 7:82–93
Dajka J-C, Wilson SK, Robinson JPW, Chong-Seng KM, Harris A, Graham NAJ (2019) Uncovering drivers of juvenile coral density following mass bleaching. Coral Reefs https://doi.org/10.1007/s00338-019-01785-w
Darling ES, McClanahan TR, Côté IM (2013) Life histories predict coral community disassembly under multiple stressors. Glob Chang Biol 19:1930–1940
Done TJ (1982) Patterns in the distribution of coral communities across the central Great Barrier Reef. Coral Reefs 1:95–107
Ekebom J, Laihonen P, Suominen T (2003) A GIS-based step-wise procedure for assessing physical exposure in fragmented archipelagos. Estuar Coast Shelf Sci 57:887–898
Elmhirst T, Connolly SR, Hughes TP (2009) Connectivity, regime shifts and the resilience of coral reefs. Coral Reefs 28:949–957
Fabricius K, De’ath G, McCook L, Turak E, Williams DM (2005) Changes in algal, coral and fish assemblages along water quality gradients on the inshore Great Barrier Reef. Mar Pollut Bull 51:384–398
Froese R, Pauly D (2015) Fishbase. www.fishbase.org.
Gilmour JP, Smith LD, Heyward AJ, Baird AH, Pratchett MS (2013) Recovery of an isolated coral reef system following severe disturbance. Science 340:69–71
Gove JM, Williams GJ, McManus MA, Clark SJ, Ehses JS, Wedding LM (2015) Coral reef benthic regimes exhibit non-linear threshold responses to natural physical drivers. Mar Ecol Prog Ser 522:33–48
Graham NAJ, Nash KL, Kool JT (2011) Coral reef recovery dynamics in a changing world. Coral Reefs 30:283–294
Graham NAJ, Jennings S, MacNeil MA, Mouillot D, Wilson SK (2015) Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 518:94–97
Graham NAJ, Wilson SK, Carr P, Hoey AS, Jennings S, MacNeil MA (2018) Seabirds enhance coral reef productivity and functioning in the absence of invasive rats. Nature 559:250–253
Heron SF, Maynard JA, van Hooidonk R, Eakin CM (2016) Warming Trends and Bleaching Stress of the World’s Coral Reefs 1985–2012. Sci Rep 6:38402
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM, Baird AH, Baum JK, Berumen ML, Bridge TC, Claar DC, Eakin CM, Gilmour JP, Graham NAJ, Harrison H, Hobbs J-PA, Hoey AS, Hoogenboom M, Lowe RJ, McCulloch MT, Pandolfi JM, Pratchett M, Schoepf V, Torda G, Wilson SK (2018a) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359:80–83
Hughes TP, Kerry JT, Baird AH, Connolly SR, Dietzel A, Eakin CM, Heron SF, Hoey AS, Hoogenboom MO, Liu G, McWilliam MJ, Pears RJ, Pratchett MS, Skirving WJ, Stella JS, Torda G (2018b) Global warming transforms coral reef assemblages. Nature 556:492–496
Huston M (1985) Variation in coral growth rates with depth at Discovery Bay, Jamaica. Coral Reefs 4:19–25
Januchowski-Hartley FA, Graham NAJ, Wilson SK, Jennings S, Perry CT (2017) Drivers and predictions of coral reef carbonate budget trajectories. Proc R Soc B Biol Sci 284(1847):20162533. https://doi.org/10.1098/rspb.2016.2533
Johns KA, Osborne KO, Logan M (2014) Contrasting rates of coral recovery and reassembly in coral communities on the Great Barrier Reef. Coral Reefs 33:553–563
Koop K, Booth D, Broadbent A, Brodie J, Bucher D, Capone D, Coll J, Dennison W, Erdmann M, Harrison P, Hoegh-Guldberg O, Hutchings P, Jones GB, Larkum AW, O’Neil J, Steven A, Tentori E, Ward S, Williamson J, Yellowlees D (2001) ENCORE: the effect of nutrient enrichment on coral reefs. Synthesis of results and conclusions. Mar Pollut Bull 42:91–120
Ladd MC, Miller MW, Hunt JH, Sharp WC, Burkepile DE (2018) Harnessing ecological processes to facilitate coral restoration. Front Ecol Environ 16:239–247
Lapointe BE (1997) Nutrient thresholds for bottom-up control of macroalgal blooms on coral reefs in Jamaica and southeast Florida. Limnol Oceanogr 42:1119–1131
MacNeil MA, Mellin C, Matthews S, Wolff NH, McClanahan TR, Devlin M, Drovandi C, Mengersen K, Graham NAJ (2019) Water quality mediates resilience on the Great Barrier Reef. Nature Ecology & Evolution 3:620–627
Madin JS, Connolly SR (2006) Ecological consequences of major hydrodynamic disturbances on coral reefs. Nature 444:477–480
McCook L, Jompa J, Diaz-Pulido G (2001) Competition between corals and algae on coral reefs: a review of evidence and mechanisms. Coral Reefs 19:400–417
McElreath R (2017) Statistical rethinking: A Bayesian course with examples in R and Stan. CRC Press
McNeish D (2016) On Using Bayesian Methods to Address Small Sample Problems. Struct Equ Modeling 23:750–773
Mellin C, MacNeil AM, Cheal AJ, Emslie MJ, Julian Caley M (2016) Marine protected areas increase resilience among coral reef communities. Ecol Lett 19:629–637
Mumby PJ, Dahlgren CP, Harborne AR, Kappel CV, Micheli F, Brumbaugh DR, Holmes KE, Mendes JM, Broad K, Sanchirico JN, Buch K, Box S, Stoffle RW, Gill AB (2006) Fishing, trophic cascades, and the process of grazing on coral reefs. Science 311:98–101
Nash KL, Graham NAJ, Jennings S, Wilson SK, Bellwood DR (2015) Herbivore cross-scale redundancy supports response diversity and promotes coral reef resilience. J Appl Ecol 53:646–655
Nyström M, Norström AV, Blenckner T, de la Torre-Castro M, Eklöf JS, Folke C, Österblom H, Steneck RS, Thyresson M, Troell M (2012) Confronting Feedbacks of Degraded Marine Ecosystems. Ecosystems 15:695–710
Ortiz J-C, Wolff NH, Anthony KRN, Devlin M, Lewis S, Mumby PJ (2018) Impaired recovery of the Great Barrier Reef under cumulative stress. Sci Adv 4:eaar6127
Osborne K, Dolman AM, Burgess SC, Johns KA (2011) Disturbance and the dynamics of coral cover on the Great Barrier Reef (1995–2009). PLoS ONE 6:e17516
Osborne K, Thompson AA, Cheal AJ, Emslie MJ, Johns KA, Jonker MJ, Logan M, Miller IR, Sweatman HPA (2017) Delayed coral recovery in a warming ocean. Glob Chang Biol 23:3869–3881
Pandolfi JM, Bradbury RH, Sala E, Hughes TP, Bjorndal KA, Cooke RG, McArdle D, McClenachan L, Newman MJH, Paredes G, Warner RR, Jackson JBC (2003) Global trajectories of the long-term decline of coral reef ecosystems. Science 301:955–958
Polunin NVC, Roberts CM (1993) Greater biomass and value of target coral-reef fishes in two small Caribbean marine reserves. Marine Ecology-Progress Series 100:167–167
Pratchett MS, Anderson KD, Hoogenboom MO, Widman E, Baird AH, Pandolfi JM, Edmunds PJ, Lough JM (2015) Spatial, temporal and taxonomic variation in coral growth—implications for the structure and function of coral reef ecosystems. Oceanogr Mar Biol Annu Rev 53:215–295
R Development Core Team (2018). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, https://www.R-project.org.
Robinson JP, Wilson SK, Robinson J, Gerry C, Lucas J, Assan C, Govinden R, Jennings S, Graham NAJ (2019) Productive instability of coral reef fisheries after climate-driven regime shifts. Nature Ecol Evol 3:183–190
Safaie A, Silbiger NJ, McClanahan TR, Pawlak G, Barshis DJ, Hench JL, Rogers JS, Williams GJ, Davis KA (2018) High frequency temperature variability reduces the risk of coral bleaching. Nat Commun 9:1671
Sheppard CRC, Harris A, Sheppard ALS (2008) Archipelago-wide coral recovery patterns since 1998 in the Chagos Archipelago, central Indian Ocean. Mar Ecol Prog Ser 362:109–117
Steneck RS, Mumby PJ, MacDonald C, Rasher DB, Stoyle G (2018) Attenuating effects of ecosystem management on coral reefs. Sci Adv 4:eaao5493
van Hooidonk R, Maynard J, Tamelander J, Gove J, Ahmadia G, Raymundo L, Williams G, Heron SF, Planes S (2016) Local-scale projections of coral reef futures and implications of the Paris Agreement. Sci Rep 6:39666
Williams GJ, Gove JM, Eynaud Y, Zgliczynski BJ, Sandin SA (2015) Local human impacts decouple natural biophysical relationships on Pacific coral reefs. Ecography 38:751–761
Wilson SK, Robinson JPW, Chong-Seng K, Robinson J, Graham NAJ (2019) Boom and bust of keystone structure on coral reefs. Coral Reefs. https://doi.org/10.1007/s00338-019-01818-4
Wilson SK, Graham NAJ, Fisher R, Robinson J, Nash K, Chong-Seng K, Polunin NVC, Aumeeruddy R, Quatre R (2012) Effect of macroalgal expansion and marine protected areas on coral recovery following a climatic disturbance. Conserv Biol 26:995–1004
Acknowledgements
We thank Aaron MacNeil for statistical advice, Simon Jennings for contributing 1994 survey data, and two anonymous reviewers for helpful suggestions. This study was funded by grants from the Royal Society (UF140691), Leverhulme Trust, and Australian Research Council. Many thanks to Seychelles Fishing Authority, Seychelles Marine Parks Authority, Nature Seychelles, and Global Vision International for field assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Morgan S. Pratchett
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Robinson, J.P.W., Wilson, S.K. & Graham, N.A.J. Abiotic and biotic controls on coral recovery 16 years after mass bleaching. Coral Reefs 38, 1255–1265 (2019). https://doi.org/10.1007/s00338-019-01831-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-019-01831-7