Abstract
Over the past decades numerous studies have reported declines in stony corals and, in many cases, phase shifts to fleshy macroalgae. However, long-term studies documenting changes in other benthic reef organisms are scarce. Here, we studied changes in cover of corals, algal turfs, benthic cyanobacterial mats, macroalgae, sponges and crustose coralline algae at four reef sites of the Caribbean islands of Curaçao and Bonaire over a time span of 40 yr. Permanent 9 m2 quadrats at 10, 20, 30 and 40 m depth were photographed at 3- to 6-yr intervals from 1973 to 2013. The temporal and spatial dynamics in the six dominant benthic groups were assessed based on image point-analysis. Our results show consistent patterns of benthic community change with a decrease in the cover of calcifying organisms across all sites and depths from 32.6 (1973) to 9.2% (2013) for corals and from 6.4 to 1% for crustose coralline algae. Initially, coral cover was replaced by algal turfs increasing from 24.5 (1973) to 38% around the early 1990s. Fleshy macroalgae, still absent in 1973, also proliferated covering 12% of the substratum approximately 20 yr later. However, these new dominants largely declined in abundance from 2002 to 2013 (11 and 2%, respectively), marking the rise of benthic cyanobacterial mats. Cyanobacterial mats became the most dominant benthic component increasing from a mere 7.1 (2002) to 22.2% (2013). The observed increase was paralleled by a small but significant increase in sponge cover (0.5 to 2.3%). Strikingly, this pattern of degradation and phase change occurred over the reef slope down to mesophotic depths of 40 m. These findings suggest that reefs dominated by algae may be less stable than previously thought and that the next phase may be the dominance of slimy cyanobacterial mats with some sponges.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past decades benthic sessile communities of tropical coral reefs have undergone significant changes worldwide as a consequence of continuous human population expansion and industrial development (Hughes 1994; Gardner et al. 2003; Hughes et al. 2003). The synergistic effects of anthropogenic disturbances such as overfishing, coastal development, pollution and climate change have triggered shifts in benthic species composition, competition, productivity and biodiversity (Hughes 1994; Jackson et al. 2014; De Bakker et al. 2016a). The Caribbean region and Gulf of Mexico have been particularly affected with an estimated decrease in scleractinian coral cover up to 80% on many reefs. This is from an average of 35% cover in the 1970s to a mean of only 14–16% between 0 and 20 m depth in more recent years (Gardner et al. 2003; Wilkinson and Souter 2008; Jackson et al. 2014). Over the same time span, benthic fleshy macroalgae have become a dominant component on many reef slopes (Hughes 1994; McCook et al. 2001; Nugues and Bak 2008), with an average increase in cover from 7 to 23% (Jackson et al. 2014). This phenomenon is commonly referred to as a ‘coral–macroalgal phase shift’. The dynamics of both corals and macroalgae, as well as their competitive interactions, have received significant attention (e.g. Hughes 1994; McCook et al. 2001; Nugues and Bak 2008; Bruno et al. 2009). However, long-term studies following changes in benthic reef components other than stony corals and macroalgae are scarce.
The relative absence of studies including sessile organisms such as sponges, crustose coralline algae (CCA), gorgonians and benthic cyanobacterial mats (BCM) highlights a lack of information on a large proportion of the reef benthos. A more holistic view of the coral reef benthos could provide useful insights into the structure and functioning of reef communities. Additionally, the vast majority of coral community studies cover only the shallow zone of the fore-reef. Communities below 20 m depth have largely been neglected in temporal studies, even though these reefs are hypothesised to play a key role in reef resilience functioning as a possible refuge or buffer for coral reef organisms (Glynn 1996). The few existing temporal studies that have included reefs below 20 m show that reef degradation also occurs on upper mesophotic reefs (down to 40 m) (e.g. Bak et al. 2005; Nagelkerken et al. 2005; Nugues and Bak 2008; De Bakker et al. 2016a).
The exact mechanisms driving coral reef degradation are likely impelled by a complex mixture of confounding anthropogenic and natural drivers. Increased eutrophication, sedimentation and decreased herbivory are among the most commonly designated factors reducing the ability of corals and CCA to cope with the various stressors impacting coral reefs (Littler and Littler 1998; Kuffner et al. 2008). Chronic nutrient enrichment increases the severity and prevalence of diseases and bleaching in corals (Bruno et al. 2003; Vega Thurber et al. 2014) and sedimentation due to land clearing and other disturbance of soil smothers corals (Rogers 1990; Nugues and Roberts 2003a, b). In addition, the massive die-off of the grazing sea urchin Diadema antillarum (Bak et al. 1984) around 1983 and continuous overharvesting of herbivorous fishes (Jackson et al. 2001) has significantly reduced grazing pressure on fleshy macroalgae in many Caribbean islands (Mumby et al. 2006). It is recognised that these processes and events have altered reef condition in favour of more adaptive, fast-growing organisms such as macroalgae (Hughes 1994; Bruno et al. 2009).
Besides macroalgae, algal turfs appear to benefit from a changing reef environment, in particular from eutrophication (Gorgula and Connell 2004; Vermeij et al. 2010). Due to their opportunistic life-history characteristics, turfs are able to rapidly occupy newly available substratum and algal turfs have become the dominant cover on many coral reefs worldwide (e.g. Gorgula and Connell 2004; Sandin et al. 2008; Haas et al. 2010). Algal turfs are multispecies assemblages of algae and cyanobacteria that can undergo successional patterns (Connell et al. 2014). They can shift to macroalgal stands when herbivore pressure is relatively low, but also to BCM (Bender et al. 2014).
BCM also appear to benefit from increased levels of eutrophication (Brocke et al. 2015a) and possibly from selective herbivore browsing because the production of nitrogenous secondary metabolites makes them largely unpalatable (Thacker et al. 1997; Nagle and Paul 1998). Several studies describe the occurrence of harmful blooms, particularly of thermo-tolerant species (e.g. Hallock 2005; Paul et al. 2005; Paul 2008; Smith and Schindler 2009; Charpy et al. 2012). Dense cyanobacterial mats can have a variety of negative effects on reef communities: they inhibit recruitment (Kuffner et al. 2006), act as pathogens (Carlton and Richardson 1995), overgrow and smother reef benthos (Ritson-Williams et al. 2005; De Bakker et al. 2016b), create an anoxic environment (Brocke et al. 2015b) and produce chemicals that cause coral and fish mortality (Nagle and Paul 1998). Thus, an increase in the frequency of BCM blooms could significantly affect benthic reef communities.
Sponges are another benthic group abundantly present on coral reefs, but often overlooked in time-series studies. They can exceed all other benthic components in cover and biomass and are in many aspects essential reef components (e.g. Diaz and Rützler 2001; De Goeij et al. 2013). Sponge cover appears to have remained relatively steady for a long time period (Nagelkerken et al. 2005). However, new studies describe a recent increase in sponge densities (e.g. Loh et al. 2015; McMurray et al. 2015), in particular coral-excavating sponges (Ward-Paige et al. 2005; Carballo et al. 2013). Although sponges are generally not able to rapidly colonise newly available substratum, they are strong competitors for space. In many cases they actively overgrow and kill other benthic organisms (Pawlik et al. 2007). It is hypothesised that sponges might profit from increased macroalgal densities, feeding on the dissolved organic carbon they release (Mueller et al. 2014; Pawlik et al. 2016; Rix et al. 2016). This, in combination with a reduction of spongivores due to overfishing, may give a competitive advantage over reef building corals (Loh et al. 2015).
We studied the temporal development of the most significant benthic components including hard coral, fleshy macroalgae, crustose coralline algae, algal turfs, benthic cyanobacterial mats and sponges using the longest running (>40 yr) coral reef time series (Bak et al. 2005; De Bakker et al. 2016a). Our objectives were to (1) elaborate on consistencies and variations in benthic coral reef community dynamic at four Caribbean reef sites over the fore-reef slope from 10 to 40 m depth and (2) follow temporal trajectories of major individual benthic sessile groups.
Materials and methods
Site description
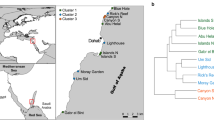
Three sites on the leeward reef of Curaçao (Carmabi Buoy One [I and II] and Carmabi Buoy Two [III]) and one site on the leeward reef of Bonaire (Karpata [IV]) (Fig. 1) have been visited since 1973 (1974 for Karpata) at intervals of 3–6 yr. At each site four permanent square quadrats (9 m2) were arranged along a transect at 10, 20, 30 and 40 m depth. During a survey, overview and detail (~1 m2) photographs were collected to allow for detailed benthic community analysis and identification. The time period of 40 yr includes the D. antillarum mass die-off in 1983, three mass bleaching events in the southern Caribbean (1998, 2005 and 2010) and several tropical storms (Joan: 1988, Lenny: 1999, Ivan: 2004, Omar: 2008). Detection of sponges, CCA and cyanobacteria required photographs of the highest quality. This limited the photographs available for analysis to 10 surveyed years between 1973 and 2013 (see Fig. 2 for exact years). Most pictures were collected using a Nikonos-V underwater camera followed by a Nikon D300 and a Nikon D800, all with high quality lenses and power flashes.
Non-metric multidimensional scaling ordinations for each individual quadrat (site-depth combination). Plots are based on the six most dominant benthic groups: hard coral (HC), algal turf (TF), benthic cyanobacterial mats (BCM), macroalgae (MA), crustose coralline algae (CCA) and sponges (SP). Each survey is labelled with the last two numbers of the year of the survey. Lines represent the degree and direction of community change between two surveys. Vector plots are given in inset. Note MA was absent in I-10 m throughout the entire time series and is therefore not shown
Benthic community analysis
Overview images were de-skewed using the free transform option in Photoshop CS6 (Adobe Photoshop CS6 v13.0.1) to compensate for any picture angle distortion and conform to the original 9 m2 quadrats. Each overview picture was overlain with a 100-point (10 × 10) regular grid (e.g. Nugues and Bak 2008), and benthic cover—biotic and abiotic—underneath each individual point was determined in the program Coral Point Count with Excel extension (CPCe) v4.1 (Kohler and Gill 2006). Detail pictures were used to resolve ambiguities. The following categories were assigned: live hard (scleractinian) coral (HC); algal turfs—filamentous assemblages <1 cm in height (TF); benthic cyanobacterial mats (BCM); fleshy macroalgae (MA)—almost exclusively Lobophora spp. and Dictyota spp.; sponges (SP), further separated into massive and encrusting growth forms; crustose coralline algae (CCA); bare substratum (BS) including recently dead coral, coral rubble and pavement; and sand (SD). Gorgonians, hydrocorals (milleporids), tunicates (with the exception of Trididemnum solidum in 1989 at IV-20 m) and other invertebrates were extremely rare in the photographs and grouped under the category other (OT). When it was impossible to identify a point, e.g. due to shading, it was classified as unidentifiable (25 of approximately 16,000 analysed points). When a group was overgrowing another, only the overgrowing group was scored. The category BCM was only assigned when clear mats were visible. Distinguishing between turf and early cyanobacterial mats was challenging, especially in black and white images. This might have led in some cases to a minor underestimation of the cyanobacterial fraction and as a consequence our estimation of cover by BCM should be viewed as conservative. To test for the adequacy of the 100 points count method, we compared our results for coral cover with results obtained by digitising the contour of each coral colony and adding all 2D colony surfaces for each quadrat. Both methods showed obvious similarity in cover and temporal patterns (Electronic supplementary material, ESM Fig. S1).
Statistical analyses
Statistical analyses were conducted in the R programming environment v3.2.3 (R Core Team 2014) and Primer-E v7.0.9. Within R the following packages were used: vegan (Oksanen et al. 2016) and mgcv (Wood 2012). Statistical assumptions of univariate analyses were assessed through graphical exploration of residual errors.
Multivariate analyses
We applied multivariate analysis techniques (Gauch 1982) to visualise changes in community composition, based on a matrix of the original counts for the six most dominant biotic groups (HC, TF, BCM, MA, SP and CCA) with site (I, II, III, IV), depth (10, 20, 30 and 40 m) and year (n = 10) as factors. By omitting the category ‘other’ (due to low counts) and the non-biotic categories bare substratum (BS) and sand (SD) any collinearity among the remaining variables was removed. The point data were fourth-root-transformed to reduce the effect of high-cover groups. A Bray–Curtis similarity matrix (Bray and Curtis 1957) was computed between years, sites and depths. We explored temporal changes in composition of our benthic groups by non-metric multidimensional scaling (nMDS). nMDS is a widely used method in ecological studies and provides good discrimination of multivariate patterns (Kruskal 1964; Clarke et al. 1993). In order to visually compare temporal linearity in multivariate space, years were displayed as points and joined in single nMDS plots for all respective quadrats (Fig. 2). A seriation test was applied to test for linearity in the development of the community (Clarke et al. 1993). The RELATE routine in Primer-E tests the similarity between patterns of any two matrices of rank dissimilarities based on Spearman rank correlation coefficient (Kendall 1948), defined as the index of multivariate seriation (IMS) (Clarke et al. 1993). Linearity of the observed temporal patterns was assessed using this RELATE procedure (M = 9999 simulations) in which the correlation between the community distance (sub)matrix and a model matrix representing the distances in time between samples was calculated. A two-way RELATE test was used to assess correlation between patterns at different depths and sites by comparing submatrices of the individual quadrats (M = 9999 simulations).
In addition to the seriation tests, multivariate control chart analysis (Anderson and Thompson 2004) was applied to the Bray–Curtis matrices to determine when a community had become significantly different from the start of the time series (ESM Fig. S2). The distance-based control chart method is a robust method that can be applied to sequential multivariate observations. In contrast to seriation, this method compares the community of each point in time to a set reference point, in our case the first survey in 1973 (Curaçao) or 1974 (Bonaire). Bootstrapping (1000 bootstraps) was used to provide percentile limits (50, 75, 90 and 95%). Points positioned above the 90% limit were considered to have a significantly different community compared to the start of each time series (Anderson and Thompson 2004). R code for the multivariate control chart analysis was developed by EM.
Univariate analyses
Understanding the observed changes in the sessile community as a whole is only possible when looking at the temporal changes in the individual benthic components. We applied regression modelling to describe the historical trajectories of the six dominant benthic groups. Because ecological data, including our data, are often non-linear, generalised additive models (GAMs) were used to fit the data. GAMs are a non-parametric extension of generalised linear models (GLMs) and model the relationship between the response variable and the explanatory variables by means of smoothing curves (Chambers and Hastie 1992). Each benthic group was individually modelled with percentage cover as response variable for the explanatory variable ‘year’. In total, four nested models were designed and tested for goodness of fit. The first model contained a smoothing term combining both depth and site [f(DS)], leading to a total of 16 smoothers. Assuming analogy in patterns with depth or site, two less complicated nested models were designed having four smoothers for either depth or site. The simplest model had one smoother for time and the factors depth and site. Inference tests for selection of the best model were carried out by an F-ratio test (ESM Table S1).
Various error distributions were applied to the GAMs and, although the resulting fits were comparable, models using transformed cover data with a normal (Gaussian) error distribution, identity link and a gamma value of 1.4 (Kim and Gu 2004) gave the most reliable and robust outcomes. Several other error distributions, for instance a beta distribution, caused an estimate bias when a series included many sequential zero values (BCM, MA, SP and CCA). Exploration of the model residuals with variogram plots indicated there was no need to account for temporal or spatial auto-correlation.
Investigating general patterns
From the GAMs a new matrix was extracted with fitted values for each year for all 96 time series (i.e. a 40 × 96 matrix). A square-root transformation was applied. To remove the effect caused by large differences in percentage cover, fitted values were standardised on the basis of the maximum cover in each time series. Using Euclidean distance a nMDS plot was computed to display patterns in temporal development within and among the six main benthic groups. To reveal dominant clusters of time series with similar patterns a cluster analysis was run using the unweighted pair group method (ESM Fig. S3).
Results
Multivariate benthic community analyses
The sessile biotic community on the studied reefs of Curaçao and Bonaire developed strongly unidirectionally over the past four decades (Fig. 2). The observed dynamics showed significant linearity in temporal development at all four sites and within sites at all four depths (ESM Tables S2, S3). The nMDS plots displayed a regular distribution of points from left to right along the horizontal axis, very much following time. Patterns were homogenous among sites, but some variation among time series was found as well (e.g. III-40 and IV-40 m). Vector plots indicated the direction of the strongest correlation with the six main groups. The overall dynamics showed a pattern directed away from hard coral (HC) and crustose coralline algae (CCA) and moving towards more benthic cyanobacterial mats (BCM), macroalgae (MA) and, below 10 m, sponges (SP). Linearity in community change was most pronounced at 20 m for all sites (P > 0.76; ESM Table S2) and at 30 m particularly at sites I (P = 0.82, p = 0.001) and III (P = 0.83, p = 0.001). Control charts confirmed a gradual change in community composition through time (ESM Fig. S2). Both methods—seriation and control charts—indicated that significant reef community change was less pronounced at greater depths. Accordingly, moderate to weak but significant IMS scores were found at 40 m. This is the case, for instance, at sites II (P = 0.39, p = 0.01) and III (P = 0.36, p < 0.01) which also clearly had a more narrow dispersion of points in the nMDS plots. Rank correlation of multivariate time series indicated very similar patterns among depths in Bonaire (P > 0.64, p < 0.05; Table S3) and medium to high correlation between depths at the other sites. Values for within-site correlation were generally smaller than between sites at the same depth (ESM Tables S2, S3), indicating that community responses were more structured by depth than by site. Significantly deviating correlations were found at 10 m depth between site II and both sites III (P 2 = 0.24, p = 0.10) and IV (P 2 = 0.19, p = 0.15) and at 40 m between Karpata (IV) and all other sites (P 2 < 0.39, p > 0.087; ESM Table S2). The latter indicated that there has been more variability in patterns of development at 10 and 40 m depth, compared to 20 and 30 m. Here, community development was very consistent. Overall, the impact of local incidental disturbances (e.g. hurricanes, bleaching events, nutrient spikes), or seasonal variation (e.g. algal blooms during summer), was less pronounced. Because these effects may be temporary and the community might have the opportunity to recover, a response may fall outside the temporal and spatial detail of our time series.
Individual benthic groups
Scleractinian corals
Between 1973 and 2013 coral cover in Curaçao and Bonaire declined by 71.8% over all sites and depths (Fig. 3), from a mean of 32.6 to 9.2 (Table 1). Only at II-30 m did coral cover not decline significantly (Fig. 3), but here cover was already relatively low in 1973. Although coral cover declined universally, the trajectories varied between time series. At the shallow fore-reef (10, 20 m) cover decreased by 79% over all quadrats between 1973 and 2013, while at deeper reefs, a slightly lower decline (61%) was observed. In general, the decreasing trend in coral cover occurred gradually through time in a relatively linear pattern with some exceptions (Fig. 3). The continuous decrease was only briefly interrupted by a period of apparent recovery between approximately 1992 and 2002 across the entire depth gradient of site I. At sites II and III at 40 m depth, coral cover initially increased until 1998, but subsequently decreased exponentially towards 2013. At 20 m in IV, coral cover declined exponentially between 1983 and 1992, likely due to the expansion of Trididemnum solidum (Bak et al. 1981; Van Duyl et al. 1981). Around the late 1980s T. solidum became very abundant on these reefs, particularly around 20 m, covering up 26% of the quadrat (ESM Fig. S4), and actively overgrowing and killing corals and other benthic components (Bak et al. 1996). The GAM with 16 smoothers explained 97.4% of the deviance (adjusted R 2 = 0.95) and was always significantly better than all three alternative nested models (ESM Table S1), which suggests considerable influence of local variables.
Temporal change in cover of the six dominant benthic groups between 1973 and 2013. Lines represent the estimated models (with 95% confidence bands). Columns represent sites; I, II, III are in Curaçao, IV is in Bonaire. Rows represent depths. Black dots are original data points. Note that Y-axes (percentage cover) vary for the different benthic groups. Significance level of smoothers: *<0.05, **<0.01
Algal turfs
Turf assemblages increased in cover from 1973, becoming the dominant biotic benthic component with a mean cover close to 40% over all quadrats around the late 1980s to early 1990s. Subsequently, however, an often exponential decline in cover was observed starting around the late 1990s towards a mean cover of 10.7% in 2013. This is less than half of the initial cover of 24.7% (Table 1). Similar to corals, the GAM with 16 separate lines was the best model. With the second best model (same trends at depth), however, the difference was less convincing (p = 0.02; ESM Table S1). In both shallow quadrats (10, 20 m) of site I, cover of algal turfs did not show a significant change (ESM Table S4). With the exception of these latter two quadrats and the almost linear decline at IV-40 m, the dynamic of algal turfs developed in a highly comparable fashion over all sites and especially within the same depth.
Benthic cyanobacterial mats
In recent years benthic cyanobacterial mats (BCM) have shown a striking increase at all sites and depths (Fig. 3). At the start of the time series BCM were virtually absent covering a mere 0.1% (Table 1). Other than a small peak around 1989 on the shallow reef (<20 m) of Curaçao, particularly at 10 m, the period preceding the rise in BCM is marked by a cover of less than 10% and an absence of any notable dynamics. After 2002, however, cover increased strongly, sometimes even exponentially, and by 2009 BCM had become the dominant biotic benthic group covering 22.2% of the substratum (ESM Fig. S4). GAM results showed that all but two (sites I-40 and IV-40 m) curves were significant (ESM Fig. S4), suggesting a lack of change at these two deeper sites. The most complex model with 16 smoothers was the best model indicating differences in patterns of development between sites and depths.
Fleshy macroalgae
There were obvious differences among depths in temporal patterns of fleshy macroalgae (Fig. 3). At 10 m macroalgae were almost absent while at 20 and 30 m the expansion on Curaçao started around 1998. On Bonaire (IV) macroalgae started increasing from 1983 onward. In addition, at 40 m depth, macroalgae on Bonaire reached high densities while they remained almost absent on Curaçao at this depth. Most significant trends showed a gradual decrease in cover after approximately 2002. With so many zero values the approximate p values of the smoothers should be interpreted with caution. Values above 0.01 should probably be considered as not significant. The model with 16 different trends was the best model, confirming the highly variable trajectories between time series in macroalgal cover. A notable interaction between Dictyota spp. and benthic cyanobacterial mats was frequently observed, where one group was overgrowing the other. BCM often appeared to facilitate growth of Dictyota spp. on sand, possibly by stabilizing the sandy substratum. In various cases, however, BCM also actively overgrew and potentially smothered Dictyota spp. This interaction could contribute to the observed recent decline in macroalgal cover since we only incorporated the overgrowing component in our analyses.
Sponges
Although sponges were generally present at low cover, they increased in the last 10–15 yr, particularly below 10 m depth (Fig. 3). Sponge cover remained low between 1973 (0.5%) and 1998 (0.4%). After 1998 it increased almost sixfold to 2.3% in 2013 (Table 1). At 10 m depth, sponge cover was generally very low and patterns of change were highly variable among sites, slightly increasing at sites I and IV and decreasing at sites II and III. The model with 16 smoothers was the best model. Differences with models 3 and 4, however, were relatively small (p = 0.03), indicating some similarity among sites. Massive and encrusting sponges both increased in cover between 1973 and 2013 by 74% and 62, respectively.
Crustose coralline algae
Similar to corals, CCA cover decreased during the first 20 yr and remained very low to 2013 (Fig. 3). Mean CCA cover over all quadrats decreased from 6.4 (1973) to only 1.0% (2013) (Table 1). Model 1 was significantly better than the three simpler models.
Similarity and variation between time series
Despite the large number of comparisons (40 × 96), time series formed distinct clusters per benthic group, in particular for HC, TF and BCM, which confirms similarity in the temporal trajectories of cover within each group (Fig. 4). There was a clear clustering of groups that increased over time (BCM, MA, SP), as well as considerable overlap of groups that ultimately declined (HC, TF, CCA). The time series of SP and CCA were more scattered, but still relatively closely grouped (Fig. 4). This suggests that patterns of change in these groups were largely consistent, but that local variation also existed. MA showed strongest diversity in patterns, with the deeper (>10 m) quadrats at site IV (Bonaire) clearly separated from the sites on Curaçao.
Non-metric multidimensional scaling plot displaying the degree of similarity between time series within and among the six main benthic groups: hard coral (HC, blue triangles), algal turfs (TF, yellow inverted triangles), benthic cyanobacterial mats (BCM, brown squares), macroalgae (MA, green diamonds), sponges (SP, pink circles) and crustose coralline algae (CCA, black crosses). Each point represents the pattern of changes in cover of a specific benthic category at one site and depth (i.e. an individual time series). Closer points indicate more similar patterns through time. The label of each point consists of the abbreviation of the benthic group, depth (10, 20, 30, 40 m) and site (I, II, III, IV)
Discussion
Phase shifts on the reefs of Curaçao and Bonaire
The composition of the benthic coral reef community in Curaçao and Bonaire between 10 and 40 m depth has changed significantly between 1973 and 2013. In general, the direction of the shift was similar across sites and depths (Fig. 2), suggesting spatial homogeneity in the forcing factor(s) driving the trajectories of change in reef community composition. We have integrated temporal trends pooling sites and depths in Fig. 5. Most striking is the unprecedented increase in cover of BCM from the late 1990s onwards. BCM have become a dominant component on these reefs at least down to 40 m. The reefs on Curaçao and Bonaire are commonly classified among the least degraded in the Caribbean region (Jackson et al. 2014). Unlike Jackson et al. (2014), our sites only included reefs downstream or close to urban areas. In the 1970s the community was dominated by calcifying organisms (corals and CCA). Over a 40-yr period, it has developed towards a community dominated by algae and subsequently cyanobacterial mats. The obvious decline in coral cover and the increase in algal abundance from 1973 to 2000 match those observed on coral reefs throughout the wider Caribbean region (Gardner et al. 2003; Bak et al. 2005; Jackson et al. 2014) and worldwide (Hughes 1994; McCook et al. 2001). CCA also declined. Although CCA cover was generally low and fluctuated between time series, our results highlight a conspicuous loss in benthic calcifying organisms. Reduced calcification and cementation on a reef means a decline in reef accretion. Ultimately, this will lead to a further loss of reef structural complexity (Alvarez-Filip et al. 2011).
Trajectories of change for six benthic groups: hard coral (HC, blue), algal turfs (TF, yellow), benthic cyanobacterial mats (BCM, brown), macroalgae (MA, green), sponges (SP, pink) and crustose coralline algae (CCA, black). Lines represent estimated models (with 95% confidence bands) of the change in mean percentage cover over all sites (I, II, III, IV) and depths (10, 20, 30, 40 m)
Over the past 40 yr coral cover gradually declined (Fig. 3). This decline in coral cover is not clearly linked to a sudden increase in cover of any of the benthic competing organisms (TF, MA or BCM) and does not support competitive loss to a given benthic group as a primary cause of coral mortality. The observed shift towards fast-growing organisms may, however, constrain the ability of corals to reclaim invaded substratum and prevent coral recovery after disturbance (McCook et al. 2001). The absence of obvious fluctuations in cover of sand and bare substratum indicates rapid colonisation of free substratum after coral mortality and suggests that competition for newly vacant space on hard substratum is intense. The increase in BCM coupled with a decline in algal turfs and, to a lesser extent, fleshy macroalgae, and an increase in sponge cover may indicate a new successional phase of coral reefs.
The rise of BCM and sponges
The trend of algal turf assemblages becoming the dominant benthic cover on the reefs of Curaçao and Bonaire has been observed on reefs worldwide, e.g. Curaçao (Vermeij et al. 2010), Australia (Gorgula and Connell 2004), the Gulf of Arabia (Haas et al. 2010) and the remote Line Islands in the Pacific (Sandin et al. 2008). The general consensus is that algal turfs benefit from elevated nutrient loads (Gorgula and Connell 2004; Vermeij et al. 2010). It is often suggested that a phase shift has occurred on many coral reefs where algae have mostly replaced the calcifying organisms (e.g. Hughes 1994). From the late 1990s onwards, however, BCM increased and became dominant at the expense of algal turfs and macroalgae while sponges showed a more limited but steady and significant increase (Fig. 3). The frequently observed interaction in which BCM overgrow fleshy macroalgae may have led to an underestimated recent algal cover. Despite being overgrown, these macroalgae (almost exclusively Dictyota spp. and Lobophora spp.) may still be functionally important in the benthic assembly. Nevertheless, macroalgae have declined in cover and the observed shifts suggest that former reefs recently dominated by algae may be less stable than previously thought and that a next successional phase may be dominated by BCM and sponges with some fleshy macroalgae.
Algal turfs consist of a large consortium of species, including cyanobacteria (Connell et al. 2014). They may develop to later successional taxa such as standing crops of macroalgae or BCM depending on local and global conditions. For instance, reduced water quality, high grazing pressure and elevated water temperature stimulate BCM growth over macroalgal growth (Kuffner and Paul 2001; Bender et al. 2014). Many cyanobacterial species can fix nitrogen (Larkum et al. 1988), but are limited in growth by phosphorous or iron (Kuffner and Paul 2001), indicating they may benefit from elevated nutrient levels in the water. They also respond positively to organic matter enrichment in sediments (Brocke et al. 2015a).
High BCM cover has been observed along the leeward side of Curaçao, Bonaire, and on multiple other reefs throughout the Caribbean including St. Eustatius, Martinique, and on Saba Bank (de Bakker et al. pers. obs.). In addition, high BCM cover has been observed throughout the entire year, suggesting high cover is not necessarily only seasonal. Increased occurrence of reef dwelling cyanobacterial blooms has been found on coral reefs around the world (e.g. Charpy et al. 2012). It is plausible that a decline in water quality and elevated water temperature initiated the recent shift to dominance of BCM on these reefs (Brocke et al. 2015a; Den Haan 2015). The reefs of both islands receive untreated sewage water via discharge and/or groundwater (Buth and Ras 1992; Lapointe and Mallin 2011). This probably applies to the entire course of the time series, because local sewage treatment plants are overall not working properly (Curaçao Ministry of Health, Environment and Nature 2014). A considerable rise in the number of inhabitants (50% increase since 2001 in Bonaire) and tourists on both islands (CBS Curacao, www.cbs.cw; CBS Dutch Caribbean, www.rijksdienstcn.com) has further increased the pressure of sewage pollution as well as land-based run-off and sedimentation on these reefs. Obtaining supporting evidence for such eutrophication is challenging because the largest pool of nutrients reaching the reef will be rapidly converted into biomass or trapped in the sediment (Koop et al. 2001). Slijkerman et al. (2014) and Govers et al. (2014) found indications of eutrophication on the reefs of Curaçao and Bonaire; however, long-term measurements of nutrient concentrations are virtually absent. Apparently, the entire Caribbean basin suffers from elevated nutrient load as a consequence of human impact (Siung-Chang 1997; Pawlik et al. 2016). Accordingly, increased anthropogenic induced stress both local (e.g. eutrophication) and regional (e.g. elevated temperature) likely initiated the observed community shift from algal dominance towards dominance of BCM and sponges. The descriptive nature of the data, however, allows only speculation with regard to the exact underlying causes behind the observed trends.
Below 10 m depth, sponge cover started increasing at approximately the same time that BCM appeared on a large scale (Fig. 5). Explanations for this increase in sponge cover could be the removal of spongivorous fishes (Loh et al. 2015) and/or a positive feedback loop between sponges, macroalgae and microbes on Caribbean reefs (Pawlik et al. 2016). Sponges feed on dissolved organic matter (DOM) produced by macroalgae (Mueller et al. 2014; Rix et al. 2016). In turn sponges release nutrients (De Goeij et al. 2013) that can fuel macroalgal expansion. A similar feedback loop may exist involving BCM which also release DOM (Brocke et al. 2015b). However, whether BCM-derived DOM is a suitable source for sponges remains to be investigated. Nevertheless, algae, BCM and sponges all compete for space with corals and can have negative consequences for the coral microbiome, reducing coral resilience to disturbances and reinforcing reef degradation (Vega Thurber et al. 2009; Pawlik et al. 2016).
Community dynamics across a depth transect
Deeper reefs (>20 m) have rarely been considered in long-term ecological studies (but see Bak and Luckhurst 1980; Bak et al. 2005; Nagelkerken et al. 2005; Nugues and Bak 2008; De Bakker et al. 2016a). Our study shows that the observed main trends in community dynamic occurred down to 40 m depth. Although changes at 40 m depth were less pronounced (Fig. 2), trends on the upper mesophotic reefs (30 and 40 m) were also marked by a loss of calcifying organisms, mainly driven by the mortality of Agaricia species (Bak et al. 2005), and a shift towards BCM dominance (Fig. 3). It is commonly assumed that deeper reefs are less subjected to anthropogenic impacts and events such as hurricanes, bleaching or harmful blooms and serve as potential refugia or buffer for coral reef species (Glynn 1996; Hughes and Tanner 2000). However, our study shows that mesophotic reefs are not free of stress (see also Bongaerts et al. 2010). For example, bleaching and increased sedimentation have also impacted the deeper reefs of Curaçao and Bonaire (Bak et al. 2005; Nugues and Bak 2008). The degradation of communities on deep reefs suggests both reef zones (shallow and deep) are now subject to anthropogenic induces disturbances.
In conclusion, the composition of benthic communities on the fore-reef of Curaçao and Bonaire has changed dramatically over the past 40 yr. Calcifying organisms have been reduced to mere figurants, and cyanobacterial mats have become a dominant benthic component. Our results suggest that the shift from coral and CCA towards turf and macroalgae may be a transitional phase that can further develop towards a new successional phase of BCM and sponge dominance with a less prominent role for fleshy macroalgae. This new community state has not previously been thoroughly described. Considering the ongoing increase of human impact (both local and global) on the coral reef environment, it may represent the next alarming step in the degradation of coral reef health.
References
Alvarez-Filip L, Gill JA, Dulvy NK, Perry AL, Watkinson AR, Côté IM (2011) Drivers of region-wide declines in architectural complexity on Caribbean reefs. Coral Reefs 30:1051–1060
Anderson MJ, Thompson AA (2004) Multivariate control charts for ecological and environmental monitoring. Ecol Appl 14:1921–1935
Bak RPM, Luckhurst BE (1980) Constancy and change in coral reef habitats along depth gradients at Curaçao. Oecologia 47:145–155
Bak RPM, Sybesma J, Van Duyl FC (1981) The ecology of the tropical compound ascidian Trididemnum solidum. II. Abundance, growth and survival. Mar Ecol Prog Ser 6:43–52
Bak RPM, Carpay MJE, De Ruyter Van Steveninck ED (1984) Densities of the sea urchin Diadema antillarum before and after mass mortalities on the coral reefs on Curaçao. Mar Ecol Prog Ser 17:105–108
Bak RPM, Nieuwland G, Meesters EHWG (2005) Coral reef crisis in deep and shallow reefs: 30 years of constancy and change in reefs of Curaçao and Bonaire. Coral reefs 24:475–479
Bak RPM, Lambrechts DYM, Joenje M, Nieuwland G, Van Veghel MLJ (1996) Long-term changes on coral reefs in booming populations of a competitive colonial ascidian. Mar Ecol Prog Ser 133:303–306
Bender D, Diaz-Pulido G, Dove S (2014) Warming and acidification promote cyanobacterial dominance in turf algal assemblages. Mar Ecol Prog Ser 517:271–284
Bongaerts P, Ridgway T, Sampayo EM, Hoegh-Guldberg O (2010) Assessing the ‘deep reef refugia’ hypothesis: focus on Caribbean reefs. Coral Reefs 29:309–327
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of southern Wisconsin. Ecol Monogr 27:325–349
Brocke HJ, Polerecky L, De Beer D, Weber M, Claudet J, Nugues MM (2015a) Organic matter degradation drives benthic cyanobacterial mat abundance on Caribbean coral reefs. PLoS One 10:e0125445
Brocke HJ, Wenzhoefer F, De Beer D, Mueller B, Van Duyl FC, Nugues MM (2015b) High dissolved organic carbon release by benthic cyanobacterial mats in a Caribbean reef ecosystem. Sci Rep 5:8852
Bruno JF, Petes LE, Harvell CD, Hettinger A (2003) Nutrient enrichment can increase the severity of coral diseases. Ecol Lett 6:1056–1061
Bruno JF, Sweatman H, Precht WF, Selig ER, Schutte VGW (2009) Assessing evidence of phase shifts from coral to macroalgal dominance on coral reefs. Ecology 90:1478–1484
Buth L, Ras J (1992) Inventory of the land-based sources of marine pollution Netherlands Antilles. Department of Public Works and Environmental Service, Curaçao, p 29
Carballo JL, Bautista E, Nava H, Cruz-Barraza JA, Chávez JA (2013) Boring sponges, an increasing threat for coral reefs affected by bleaching events. Ecol Evol 3:872–886
Carlton RG, Richardson LL (1995) Oxygen and sulfide dynamics in a horizontally migrating cyanobacterial mat: black band disease of corals. FEMS Microbiol Ecol 18:155–162
Chambers JM, Hastie TJ (1992) Linear models. In: Chambers JM, Hastie TJ (eds) Statistical models in S. Wadsworth & Brooks/Cole Pub, Pacific Grove, California, pp 95–144
Charpy L, Casareto BE, Langlade M, Suzuki Y (2012) Cyanobacteria in coral reef ecosystems: a review. J Mar Biol 2012:1–9
Clarke K, Warwick R, Brown B (1993) An index showing breakdown of seriation, related to disturbance, in a coral-reef assemblage. Mar Ecol Prog Ser 102:153–160
Connell SD, Foster MS, Airoldi L (2014) What are algal turfs? Towards a better description of turfs. Mar Ecol Prog Ser 495:299–307
Curaçao Ministy of Health, Environment and Nature (2014) National report sustainable development Curaçao. Third international SIDS conference, Apia, Samoa
De Bakker DM, Meesters EH, Bak RP, Nieuwland G, van Duyl FC (2016a) Long-term shifts in coral communities on shallow to deep reef slopes of Curaçao and Bonaire: are there any winners? Front Mar Sci 3:247
De Bakker DM, Meesters EHWG, Van Bleijswijk JDL, Luttikhuizen PC, Breeuwer HJAJ, Becking LE (2016b) Population genetic structure, abundance, and health status of two dominant benthic species in the Saba Bank National Park, Caribbean Netherlands: Montastraea cavernosa and Xestospongia muta. PLoS One 11:e0155969
De Goeij JM, Van Oevelen D, Vermeij MJA, Osinga R, Middelburg JJ, De Goeij AFPM, Admiraal W (2013) Surviving in a marine desert: the sponge loop retains resources within coral reefs. Science 342:108–110
Den Haan J (2015) Effects of nutrient enrichment on the primary producers of a degraded coral reef. Ph.D. thesis, University of Amsterdam, p 38–58
Diaz MC, Rützler K (2001) Sponges: an essential component of Caribbean coral reefs. Bull Mar Sci 69:535–546
Gardner TA, Côté IM, Gill JA, Grant A, Watkinson AR (2003) Long-term region-wide declines in Caribbean corals. Science 301:958–960
Gauch HG (1982) Multivariate analysis in community ecology. Cambridge University Press, Cambridge
Glynn PW (1996) Coral reef bleaching: facts, hypotheses and implications. Glob Chang Biol 2:495–509
Gorgula SK, Connell SD (2004) Expansive covers of turf-forming algae on human-dominated coast: the relative effects of increasing nutrient and sediment loads. Mar Biol 145:613–619
Govers LL, Lamers LPM, Bouma TJ, De Brouwer JHF, Van Katwijk MM (2014) Eutrophication threatens Caribbean seagrasses—an example from Curaçao and Bonaire. Mar Pollut Bull 89:481–486
Haas A, El-Zibdah M, Wild C (2010) Seasonal monitoring of coral–algae interactions in fringing reefs of the Gulf of Aqaba, Northern Red Sea. Coral Reefs 29:93–103
Hallock P (2005) Global change and modern coral reefs: new opportunities to understand shallow-water carbonate depositional processes. Sediment Geol 175:19–33
Hughes TP (1994) Catastrophes, phase shifts, and large-scale degradation of a Caribbean coral reef. Science 265:1547–1551
Hughes TP, Tanner JE (2000) Recruitment failure, life histories, and long-term decline of Caribbean corals. Ecology 81:2250–2263
Hughes TP, Baird AH, Bellwood DR, Card M, Connolly SR, Folke C, Grosberg R, Hoegh-Guldberg O, Jackson JBC, Kleypas J (2003) Climate change, human impacts, and the resilience of coral reefs. Science 301:929–933
Jackson JBC, Donovan M, Cramer K, Lam V (2014) Status and trends of Caribbean coral reefs: 1970–2012. Global Coral Reef Monitoring Network, IUCN, Gland, Switzerland
Jackson JBC, Kirby MX, Berger WH, Bjorndal KA, Botsford LW, Bourque BJ, Bradbury RH, Cooke R, Erlandson J, Estes JA (2001) Historical overfishing and the recent collapse of coastal ecosystems. Science 293:629–637
Kendall ME (1948) Rank correlation methods. Charles Griffin and Company, London, UK, p 212
Kim YJ, Gu C (2004) Smoothing spline Gaussian regression: more scalable computation via efficient approximation. J R Stat Soc Series B Stat Methodol 66:337–356
Kohler KE, Gill SM (2006) Coral point count with Excel extensions (CPCe): a Visual Basic program for the determination of coral and substrate coverage using random point count methodology. Comput Geosci 32:1259–1269
Koop K, Booth D, Broadbent A, Brodie J, Bucher D, Capone D, Coll J, Dennison W, Erdmann M, Harrison P (2001) ENCORE: the effect of nutrient enrichment on coral reefs. Synthesis of results and conclusions. Mar Pollut Bull 42:91–120
Kruskal JB (1964) Multidimensional scaling by optimizing goodness of fit to a non-metric hypothesis. Psychometrika 29:1–27
Kuffner IB, Paul VJ (2001) Effects of nitrate, phosphate and iron on the growth of macroalgae and benthic cyanobacteria from Cocos Lagoon, Guam. Mar Ecol Prog Ser 222:63–72
Kuffner IB, Andersson AJ, Jokiel PL, Rodgers KS, Mackenzie FT (2008) Decreased abundance of crustose coralline algae due to ocean acidification. Nat Geosci 1:114–117
Kuffner IB, Walters LJ, Becerro MA, Paul VJ, Ritson-Williams R, Beach KS (2006) Inhibition of coral recruitment by macroalgae and cyanobacteria. Mar Ecol Prog Ser 323:107–117
Lapointe BE, Mallin MA (2011) Nutrient enrichment and eutrophication on fringing coral reefs of Bonaire and Curaçao, Netherlands Antilles. Report to the United Nations Environment Programme for the NACRI Coral Reef Monitoring Program, Harbor Branch Oceanographic Institute, Ft Pierce Fl, 42 pp
Larkum AWD, Kennedy IR, Muller WJ (1988) Nitrogen fixation on a coral reef. Mar Biol 98:143–155
Littler MM, Littler DS (1998) An undescribed fungal pathogen of reef-forming crustose coralline algae discovered in American Samoa. Coral Reefs 17:144
Loh TL, McMurray SE, Henkel TP, Vicente J, Pawlik JR (2015) Indirect effects of overfishing on Caribbean reefs: sponges overgrow reef-building corals. PeerJ 3:e901
McCook L, Jompa J, Diaz-Pulido G (2001) Competition between corals and algae on coral reefs: a review of evidence and mechanisms. Coral Reefs 19:400–417
McMurray SE, Finelli CM, Pawlik JR (2015) Population dynamics of giant barrel sponges on Florida coral reefs. J Exp Mar Bio Ecol 473:73–80
Mueller B, de Goeij JM, Vermeij MJ, Mulders Y, van der Ent E, Ribes M, van Duyl FC (2014) Natural diet of coral-excavating sponges consists mainly of dissolved organic carbon (DOC). PLoS One 9:e90152
Mumby PJ, Dahlgren CP, Harborne AR, Kappel CV, Micheli F, Brumbaugh DR, Holmes KE, Mendes JM, Broad K, Sanchirico JN (2006) Fishing, trophic cascades, and the process of grazing on coral reefs. Science 311:98–101
Nagelkerken I, Vermonden K, Moraes OCC, Debrot AO, Nagelkerken WP (2005) Changes in coral reef communities and an associated reef fish species, Cephalopholis cruentata (Lacepede), after 30 years on Curaçao (Netherlands Antilles). Hydrobiologia 549:145–154
Nagle DG, Paul VJ (1998) Chemical defense of a marine cyanobacterial bloom. J Exp Mar Bio Ecol 225:29–38
Nugues MM, Roberts CM (2003a) Partial mortality in massive reef corals as an indicator of sediment stress on coral reefs. Mar Pollut Bull 46:314–323
Nugues MM, Roberts CM (2003b) Coral mortality and competition with algae in relation to sedimentation. Coral Reefs 22:507–516
Nugues MM, Bak RPM (2008) Long-term dynamics of the brown macroalga Lobophora variegata on deep reefs in Curaçao. Coral Reefs 27:389–393
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2016) vegan: community ecology package. R package version 2.3-5
Paul VJ (2008) Global warming and cyanobacterial harmful algal blooms. In: Hudnell HK (ed) Cyanobacterial harmful algal blooms: state of the science and research needs. Advances in experimental medicine and biology, Springer, Netherlands, pp 239–257
Paul VJ, Thacker RW, Banks K, Golubic S (2005) Benthic cyanobacterial bloom impacts the reefs of South Florida (Broward County, USA). Coral Reefs 24:693–697
Pawlik JR, Burkepile DE, Vega-Thurber RL (2016) A vicious circle? Altered carbon and nutrient cycling may explain the low resilience of Caribbean coral reefs. BioScience [doi:10.1093/biosci/biw047]
Pawlik JR, Steindler L, Henkel TP, Beer S, Ilan M (2007) Chemical warfare on coral reefs: sponge metabolites differentially affect coral symbiosis in situ. Limnol Oceanogr 52:907–911
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ritson-Williams R, Paul VJ, Bonito V (2005) Marine benthic cyanobacteria overgrow coral reef organisms. Coral Reefs 24:629
Rix L, De Goeij JM, Mueller CE, Struck U, Middelburg JJ, Van Duyl FC, Al-Horani FA, Wild C, Naumann MS, Van Oevelen D (2016) Coral mucus fuels the sponge loop in warm-and cold-water coral reef ecosystems. Sci Rep 6:18715
Rogers CS (1990) Responses of coral reefs and reef organisms to sedimentation. Mar Ecol Prog Ser 62:185–202
Sandin SA, Smith JE, DeMartini EE, Dinsdale EA, Donner SD, Friedlander AM, Konotchick T, Malay M, Maragos JE, Obura D (2008) Baselines and degradation of coral reefs in the northern Line Islands. PLoS One 3:e1548
Siung-Chang A (1997) A review of marine pollution issues in the Caribbean. Environ Geochem Health 19:45–55
Slijkerman DME, De León R, De Vries P (2014) A baseline water quality assessment of the coastal reefs of Bonaire, Southern Caribbean. Mar Pollut Bull 86:523–529
Smith VH, Schindler DW (2009) Eutrophication science: where do we go from here? Trends Ecol Evol 24:201–207
Thacker RW, Nagle DG, Paul VJ (1997) Effects of repeated exposures to marine cyanobacterial secondary metabolites on feeding by juvenile rabbitfish and parrotfish. Mar Ecol Prog Ser 147:21–29
Van Duyl F, Bak R, Sybesma J (1981) The ecology of the tropical compound ascidian Trididemnum solidum. I. Reproductive strategy and larval behavior. Mar Ecol Prog Ser 6:35–42
Vega Thurber RL, Burkepile DE, Fuchs C, Shantz AA, McMinds R, Zaneveld JR (2014) Chronic nutrient enrichment increases prevalence and severity of coral disease and bleaching. Glob Chang Biol 20:544–554
Vega Thurber RL, Willner-Hall D, Rodriguez-Mueller B, Desnues C, Edwards RA, Angly F, Dinsdale E, Kelly L, Rohwer F (2009) Metagenomic analysis of stressed coral holobionts. Environ Microbiol 11:2148–2163
Vermeij MJA, Van Moorselaar I, Engelhard S, Hörnlein C, Vonk SM, Visser PM (2010) The effects of nutrient enrichment and herbivore abundance on the ability of turf algae to overgrow coral in the Caribbean. PLoS One 5:e14312
Ward-Paige CS, Risk MJ, Sherwood OA, Jaap WC (2005) Clionid sponge surveys on the Florida Reef Tract suggest land-based nutrient inputs. Mar Pollut Bull 51:570–579
Wilkinson CR, Souter DN (2008) Status of Caribbean coral reefs after bleaching and hurricanes in 2005. Global Coral Reef Monitoring Network, IUCN, Gland, Switzerland
Wood SN (2012) Mixed GAM computation vehicle with GCV/AIC/REML smoothness estimation. R package version 1.8-12
Acknowledgements
We are deeply grateful to the CARMABI research institute on Curaçao and STINAPA on Bonaire for their assistance over the years. The present research project (Ph.D. DdeB) was funded by Wageningen Marine Research, formerly known as IMARES, at the request of and with funding from the Ministry of Economic Affairs for the purposes of the Policy Supporting Research Theme ‘Analyse fotomateriaal koraalrif/fase 1’ (Project No. BO-11-019.02-038). The Royal Netherlands Institute for Sea Research provided funding of expenses and in-kind support. We would like to thank H. Groen and both reviewers for a thorough review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ecology Editor Dr. Alastair Harborne
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Method comparison. a Modelled patterns of development in hard coral cover over time based on the 100-point grid estimate b Modelled patterns of development in hard coral cover over time based on the actual total coral cover in the quadrats. Patterns are given for all 16 quadrats (EPS 30860 kb)
Fig. S2
Distance-based multivariate control charts for all 16 quadrats. Each point segment represents the multivariate distance between the beginning of the time series and that point in time. The four horizontal lines are percentile limits at 50%, 75%, 90% and 95% of the bootstrapped distributions (n = 10.000). Time points (dots) positioned above the 90% limit are considered to have a significantly different community. The reference point is set to zero and not shown in these figures (EPS 1444 kb)
Fig. S3
Dendrogram of cluster analysis. Each sample represents the time series of a benthic group: hard coral (HC), algal turfs (TF), benthic cyanobacterial mats (BCM), macroalgae (MA), sponges (SP) and crustose coralline algae (CCA) at each site (I-IV) and depth (10, 20, 30, 40 m) (EPS 1338 kb)
Fig. S4
a Close up of site IV (20 m) in 1989 showing Trididemnum solidum covering a large portion of the reef; b Benthic cyanobacterial mats covering most of the substratum in site I (20 m) in 2013 (EPS 1618 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
de Bakker, D.M., van Duyl, F.C., Bak, R.P.M. et al. 40 Years of benthic community change on the Caribbean reefs of Curaçao and Bonaire: the rise of slimy cyanobacterial mats. Coral Reefs 36, 355–367 (2017). https://doi.org/10.1007/s00338-016-1534-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-016-1534-9