Abstract
Quantitative estimates of past vegetation cover are needed both regionally so human-landscape interactions can be better understood, and globally to evaluate the effects of changing vegetation cover on the climate system. Models reducing the bias in the pollen representation of vegetation cover have been developed in the temperate regions of the northern hemisphere, while experience applying them in other parts of the world is limited. The Araucaria forest-grassland mosaic of southern Brazil is an area where open and forested plant communities exist naturally and have changed their cover over time due to changing climate and human activities. Therefore, this area is of particular interest for such studies. Modern pollen and vegetation comparisons were carried out in two protected areas: Vila Velha State Park in Paraná state and Aparados da Serra National Park in Rio Grande do Sul. Vegetation mapping and analysis focused on Araucaria angustifolia as the characteristic tree of this vegetation type. In the Araucaria forest-grassland mosaic open vegetation and woodland change at a scale of hundreds of metres. These changes are difficult to capture based on pollen proportions in surface samples. While the abundance of Poaceae pollen is not a good indicator of locally open conditions, several pollen taxa can be used as indicators of the local vegetation cover. Pollen vegetation ratios (R-values) compare well between the two study regions, indicating that pollen production of individual species within the large plant families of Poaceae and Asteraceae are similar within the overall region. Araucaria angustifolia pollen is underrepresented with regards to its vegetation cover, while Poaceae are among the highest pollen producers in the region. Diverse woodland species other than A. angustifolia were grouped as one forest taxon and as the species composition of woodlands differed between the two study areas, so did the estimated pollen productivity of this group. It would be rewarding in future investigations to estimate pollen productivity for groups of trees with the same pollen dispersal syndrome. The application of pollen dispersal models designed for closed canopy in the protected areas was challenging. Further model development is required to deal with pollen released at different levels in semi-open vegetation types.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the main goals of pollen analysis is to reconstruct the vegetation cover of past landscapes. Pollen provides a biased representation of the vegetation resulting in the need to study the relationship between pollen and modern vegetation to reduce that bias. A simple but effective approach is to collect modern samples from different landscapes as modern analogues to interpret the fossil record, which can be matched statistically (e.g. Zanon et al. 2018). While this method has been utilized since the development of pollen analysis (e.g. Auer 1927), it still provides valuable insights where a numerical description of environmental parameters associated to surface samples is available (e.g. Davis et al. 2013). In Europe and North America, pollen vegetation comparisons were developed with the final goal of correcting the values in pollen diagrams directly. Davis (1963) introduced the R-value as the ratio of pollen to vegetation in the same vegetation plot. However, pollen coming from beyond the vegetation plot requires more sophisticated analytical approaches to the problem (Parsons and Prentice 1981). Andersen (1970) used absolute pollen deposition data obtained from pollen traps and compared it to the basal area of the trees in the vicinity of the traps, producing correction factors for the production of common European trees. In this case of pollen traps situated under the canopy, differential pollen transport is not important. However, the physical properties of different pollen types result in some pollen types staying airborne for longer, which will influence the pollen signal in most situations.
Mechanistic models of pollen transport can describe this differential dispersal of pollen types. Applied within the Extended R-value Model (ERV), the differential pollen production relative to a reference taxon can be evaluated (Prentice 1985; Sugita 1993). Motivated by the desire to reconstruct the deforestation of European landscapes with the introduction and intensification of agriculture (Roberts et al. 2018), these models and approaches were refined mainly in Europe (Gaillard et al. 2008). Recognizing the importance of vegetation feedback for explaining past climate change (Gaillard et al. 2010; Strandberg et al. 2014) motivated quantitative estimates of global vegetation cover change (Harrison et al. 2020). However, while pollen diagrams are becoming more readily available globally (Williams et al. 2018), estimates of relative pollen production for major plant groups are hitherto not available for most regions.
In South America, a large number of local and regional pollen surface sample studies have been conducted generally to assist in the qualitative interpretation of pollen diagrams (Marchant et al. 2009; Flantua et al. 2015). Little of that data is available and/or linked to environmental parameters allowing quantitative reconstructions (Montade et al. 2019). This shortcoming is represented by the small number of studies available from South America providing any kind of quantitative reconstruction based on pollen, as visible in the distribution of global pollen-based climate reconstructions by Bartlein et al. (2011). Quantitative estimates of past vegetation cover in South America have so far only been attempted for south-western Amazonia, reducing the landscape to two vegetation types: forest and non-forest (Whitney et al. 2019). This lack of quantitative vegetation reconstructions is partly due to the shortage of detailed information on the pollen representation of South American vegetation types (Whitney et al. 2019). To date, no study has been published for South America estimating relative pollen productivity following approaches developed in Europe and North America based on individual field studies (Bunting et al. 2013) or using available data (Dawson et al. 2016).
Pollen vegetation relationship studies indicating overrepresented and underrepresented taxa are available for specific vegetation types such as saltmarsh (Medeanic et al. 2016), coastal vegetation (Fontana 2005; Masciadri et al. 2013) and Cerrado (Ledru 2002). Studies explicitly estimating the pollen vegetation ratio using pollen traps and forest inventory data have been published for Barro Colorado Island, Panama (Bush and Rivera 1998; Haselhorst et al. 2020) as well as Noel Kempff Mercado National Park and Beni Basin in Bolivia (Gosling et al. 2005, 2009; Jones et al. 2011). Montade et al. (2016) published averaged pollen vegetation ratios for three isolated mountains in Ceará, north-eastern Brazil, based on a collection of surface soil samples and botanical surveys. Analysing the pollen composition of moss cushions collected in a private forest reserve compared to vegetation survey and long-term monitoring data, Cárdenas et al. (2019) provide ratios of average pollen versus vegetation proportions. Overall, quantitative pollen vegetation studies are indeed rare and have so far not developed beyond the R-value approach. These are generally based on averaging pollen and vegetation information, rarely calculating the ratio of values, making them comparable between sites. While these values are informative, general problems of the R-values need to be considered (Parsons and Prentice 1981).
Reconstructing changes in vegetation cover in South America is of interest in understanding the impact of pre-Columbian societies (McMichael et al. 2012) and climatically driven forest cover changes (Rodrigues et al. 2016). This is of particular interest in the Araucaria forest-grassland mosaic of southern Brazil, where pollen diagrams document a stepwise shift from open grasslands to dense forests during the Holocene (e.g. Behling et al. 2004). The area is part of the Atlantic Forest biome, a global biodiversity hotspot (Colombo and Joly 2010) and understanding its natural dynamics will contribute to securing its future survival. Before European arrival, this landscape was home to the southern Jê, who had semi-sedentary lifestyles and might have contributed to the afforestation as their diet included the seeds of Araucaria (Iriarte and Behling 2007). Several fossil pollen diagrams exist from the region, and their interpretation has so far been qualitative, relying on expert knowledge.
This study aims to analyse the pollen vegetation relationship in the Araucaria region of southern Brazil to improve our ability to reconstruct local forest cover changes, both qualitatively and quantitatively. Going beyond the simple R-value approach, we aim to estimate relative pollen productivity for a few dominant pollen taxa with a special focus on A. angustifolia. We also investigate the general applicability of these values by selecting two study regions with contrasting climate.
Environmental setting and methods
Geography and climate
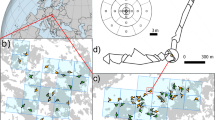
The two study regions are located in southern Brazil and differ in relation to their proximity to the Atlantic Ocean, resulting in differences in precipitation. Vila Velha State Park (25° 12ʹ S, 50° 02ʹ W) in Paraná state is situated more inland with a stronger seasonal climate. Aparados da Serra National Park (29° 10ʹ S, 50° 07ʹ W) in Rio Grande do Sul is located at the edge of the highland with a strong Atlantic influence (Fig. 1). The study areas are situated at an elevation of 850–960 m a.s.l. and ca. 900–1,000 m a.s.l. respectively. The climate in Vila Velha is characterized by a mean annual temperature of 17.4 °C and annual precipitation of 1,554 mm with more moisture availability during January and February. In Aparados da Serra the mean annual temperature is 14.8 °C with an annual precipitation of 1,807 mm, September and October being the wettest months (Alvares et al. 2013).
Vegetation
Vegetation in both parks is characterized by a mosaic of Araucaria forest and grassland. Species composition of forests and grasslands is different in the two parks. Vila Velha shows elements of the Cerrado vegetation biome. Open grassland areas are mainly related to shallow soils on the sandstone, while woodlands and semi-open areas dominated by large shrubs occupy the valleys and locations with deeper soils. Small groups of A. angustifolia trees occur in open areas, and the tree dominates some forest patches. However, the majority of the woodlands are dominated by taxa of the Myrtaceae, Fabaceae, Melastomataceae, Lauraceae and Asteraceae families; A. angustifolia is usually sub-dominant.
The grassland vegetation is also highly diverse, including Cyperaceae, Dicksonia sp., Poaceae and Asteraceae. The most abundant species are the grasses Aristida jubata, Andropogon bicornis and Trachypogon canescens. The most diverse plant family is the Asteraceae with 208 species, including 34 in the genus Baccharis (Cervi et al. 2007; Dalazoana et al. 2007; Mocochinski and Scheer 2008).
In contrast, in Aparados da Serra the forest patches are dominated by A. angustifolia in the emergent layer, usually accompanied by Mimosa scabrella and Pictocarpa angustifolia in the canopy layer and by arboreal elements such as Lamanonia speciosa, Ilex microdonta, Podocarpus lambertii, Sapium glandulatum and, Myrsine spp. in the understory. Small trees and shrubs from the families Melastomataceae (Tibouchina, Leandra, Miconia cinerascens and Miconia sellowiana) and Myrtaceae (Myrceugenia euosma and Siphoneugena reitzii) are common, the woody bamboo Merostachys multiramea is also present but less frequent (Rambo 1956; Baptista et al. 1979; MMA and IBAMA 2007).
A semi-open vegetation type is rare in Aparados da Serra. Here, the vast valleys are often filled with wetlands. Open vegetation patches are dominated by Poaceae, Baccharis spp. or Eryngium spp. Other important herbaceous taxa are Polygala spp., Euphorbia spp., Cyperus spp., Bulbostylis spp., Rhynchospora spp., Sisyrinchium spp., Dicksonia sellowiana, Cyathea spp., Acaena eupatoria, Tradescantia spp., Desmodium spp., Oxalis spp., and Chaptalia spp. In this park, the Asteraceae family is highly diverse, with 133 species, including 34 belonging to the genus Baccharis (Rambo 1956; Baptista et al. 1979; MMA and IBAMA 2007).
Pollen sampling and vegetation surveys
Vegetation surveys were carried out together with the collection of samples for pollen analysis in July 2018. Sites were selected to obtain a good representation of the different vegetation types and cover a gradient from fully open to fully forested situations. If possible, pollen samples were taken in small wet spots with mosses present. Here, the green parts of the mosses were sampled, assuming that these would contain at least 1 year of pollen deposition. Where no mosses were present, we sampled the soil litter. In all situations, several samples were collected from within about 1 m2 and mixed. The centre point for the collection of the pollen sample was taken as the centre for the vegetation sampling. The location was recorded with a GPS (ESM Table 1).
The vegetation was surveyed following the general strategy proposed by Bunting et al. (2013). Out to 10 m from the sampling points, the vegetation survey was conducted by walking in circles around the sampling point, increasing the radius by 2 m each time, estimating and mapping vegetation cover (Broström et al. 2004). The radius of the innermost circle was 1 m. The area out to 50 m from the sampling point was sketched and described based on transects in the four directions of the compass. Only mature, pollen-producing trees were included. A general vegetation map was constructed out to 2,000 m from the sampling points based on satellite images and field notes (Fig. 1). Mapping was done using QGIS (2019) 3.4.0 software.
Samples and data treatment
All samples were treated in the laboratory with 10% hot KOH, 10% hot HCL, 70% cold HF and 3 min of hot acetolysis. After the KOH treatment, the samples were sieved using a 150 µm mesh size to remove larger organic and inorganic remains. Mounted samples were analysed at × 400 and × 1,000 magnification; a minimum of 300 pollen grains per slide were counted. The identification of palynomorphs was carried out using the reference collections of the Department of Palynology and Climate Dynamics of the University of Göttingen as well as catalogues of pollen and palynological keys by Bauermann et al. (2013), Roubik and Moreno (1991) and Silva et al. (2016) and the online Neotropical Pollen Key (Bush and Weng 2007).
Sites were classified as open, semi-open or forested vegetation types based on the vegetation data within a 100 m distance from each pollen sample. The classification was carried out using unconstrained cluster analysis with the squared chord distance and Ward’s method for linking samples. The Dufrene-Legendre Indicator Species Analysis (IndVal) was performed on the pollen data using the vegetation classification to explore whether individual pollen taxa may identify the different vegetation types. PCA was performed to visualize the major differences in pollen composition and explore the similarities between pollen and vegetation data. Analyses were performed in R using the “vegan” and “labdsv” packages (Dufrêne and Legendre 1997; Oksanen et al. 2017).
We calculated the representation factor (R-value sensu Davis 1963) to compare to other studies from South America using this simple ratio and evaluate the results obtained with the more sophisticated Extended R-Value model (ERV). Representation factors were estimated for single sites as well as mean values over all sites in each study area, following Eq. (1):
Relative pollen productivity estimates (PPEs)
Estimates of the relative pollen productivity for A. angustifolia, Asteraceae, Baccharis, Cyperaceae, Poaceae and Forest were obtained with the Extended R-Value model, as implemented in ERV.Analysis.v2.5.3.exe software (Sugita 2019, unpublished) and ERV Analysis in R (Theuerkauf and Couwenberg 2021, unpublished). Analyses were initially run with all sub-models, however, ERV sub-model 1 gave the most consistent and logical results and analyses with sub-models 2 and 3 were not pursued further. We used taxon-specific distance-weighting assuming a Gaussian plume diffusion model (GPM) of small particles in the air (Prentice 1985) as well as the Lagrangian stochastic model (LSM) (Kuparinen 2006). Following previous ERV studies (Broström et al. 2004), we set basin radius to 1 m. The GPM predicts very high pollen deposition near the pollen source, resulting in an unrealistically high weight of the vegetation in the first metre. In the case of A. angustifolia, the GPM predicts that 98% of all pollen released would be deposited within 1 m from the tree.
Pollen productivity was initially computed relative to Poaceae as species of this family occur in all vegetation types. However, the lag of a strong gradient in the abundance of grasses led to spuriously high PPEs in some analysis. Therefore, we also performed the analyses with Cyperaceae as a reference taxon. The fall speed of pollen was estimated using Stoke’s law (Gregory 1973) based on measurements of the diameter of at least 30 pollen grains. The “forest group” is used as a single pollen taxon but represents a large mix of trees from different plant families with various pollen types. Different pollen types contained in that group were measured in a typical sample, and the average weighted by the proportional abundance of these pollen types was used to estimate the fall speed for this group.
Results
Modern pollen and vegetation data
The overall cover abundance of the vegetation within a buffer of 2,000 m around all surface samples yielded similar vegetation proportions in Vila Velha and Aparados da Serra (Table 1), with A. angustifolia (12 and 13%), Poaceae (23 and 24%), Cyperaceae (13 and 15%) and Baccharis (9 and 5%). The largest difference was found for “Forest taxa” with 26 and 19% in Vila Velha and Aparados da Serra respectively. The vegetation composition within 100 m around each surface sample was used in a cluster analysis and compared to the assignment samples in the field to one of three classes “open”, “semi-open” and “forest”. Results of this cluster analysis yielded two main groups in both regions (Fig. 2). The cluster “semi-open” vegetation is related to samples from the “open vegetation” in Vila Velha, and to “forest” in Aparados da Serra. In Vila Velha the cluster analysis is not fully separating sites from open and semi-open areas. One group collects all semi-open vegetation sites (3, 4, 8, 14), but includes sites 5 and 10 from open environments. Both sites 5 and 10 are situated near rock outcrops, which reduces the overall vegetation cover and might be the reason for the assignment of the samples to this group.
Pollen analysis of the 34 surface samples yielded a total of 75 different pollen and spore taxa (ESM Table 2). In Vila Velha (Fig. 2) the most abundant pollen type is Poaceae (average 40, min 18, max 55%), followed by Cyperaceae (19, min 2, max 40%). The proportion of A. angustifolia pollen varies between 0 and 18%, with an average of 5%. All other tree pollen taxa are combined in the forest group (average 16, min 4, max 42%), with Myrsine, Mimosa and Alchornea as the most abundant. Baccharis-type (average 9, min 1, max 34%) is the most abundant Asteraceae pollen, and as plants of the genus were mapped in the field, it is considered separately. Ambrosia-type and Senecio are most of the remaining Asteraceae pollen (average 11, min 1, max 27%), which are grouped under Asteraceae p.p. in further analyses.
Pollen percentages from both areas are compared to the results of the cluster analysis of the 100 m radius vegetation data (Fig. 2). For Vila Velha, pollen proportions of tree versus herbaceous pollen types do not show a clear difference between sites from the three environments. However, forest sites have higher pollen percentages of A. angustifolia and Myrtaceae and lower proportions of Baccharis-type. Pollen and vegetation data show better correspondence for the samples from Aparados da Serra. The open vegetation group contains a cluster of three sites (3, 13, 15), which are wetlands within large open areas, and their pollen content reflects this in the dominance of Poaceae and Cyperaceae.
We used the indicator species analysis (IndVal) to identify the pollen taxa that are characteristic for the vegetation groups identified by the cluster analysis (Table 2). In Villa Velha, higher pollen proportions of Baccharis-type are a strong indication (IndVal 0.94) that the sample is from open semi-open vegetation, while the occurrence of Schinus pollen indicates forest (IndVal 0.72). In Aparados da Serra the open vegetation is not well indicated by a particular pollen type, while tree pollen proportions of Myrtaceae (0.91), A. angustifolia (0.79), Ilex (0.78), Celtis (0.79), Myrsine (0.74) and Trema (0.78) are robust indicator taxa for forest plots.
Pollen vegetation relationships
Multivariate data analysis
Principal component analysis of the vegetation data (Fig. 3a, c) separated forested from open vegetation sites. Semi-open sites have different affinities in the two study areas and plot near open sites in Vila Velha and forest sites in Aparados da Serra. In Vila Velha, forest plots are characterized by A. angustifolia, which separates these plots from semi-open plots. As mentioned before, plot 5 and 10 differ from other open plots because they are situated near and within rock outcrops and exposed soil, changing the proportion of the mapped vegetation in comparison to the overall open character of these sites. Apart from these two samples, the PCA separates semi-open from open plots. In Aparados da Serra (Fig. 3c), the vectors for A. angustifolia and forest are not strongly separated, indicating similar tree composition in forest and semi-open plots.
The PCA of the pollen data (Fig. 3b, d) does not yield a clear separation of open, semi-open and forested sites, particularly for Vila Velha (Fig. 3b). Five of the open sites (5, 6, 7, 10, 13) with high pollen percentages of Baccharis-type and other Asteraceae are separated from semi-open and forested plots. Three open sites (1, 12, 15) instead appear inseparable from semi-open and forested plots because all are characterized by high values of Poaceae and Cyperaceae. In sample 1, for example, Poaceae and Cyperaceae sum up to ~ 75%, while pollen from the Asteraceae family is rare. Sample 15 comes from open vegetation; however, semi-open vegetation occurs only 100 m from the site. Pollen composition in sample 15 is similar to sample 14 from a site in this nearby semi-open vegetation.
Also, in Aparados da Serra (Fig. 3d), pollen composition does not separate open, semi-open and forested sites. The first axis represents the variance in the pollen proportions of Poaceae versus the forest elements, generally separating open versus forested sites. However, a sample from the forested site 17 and the semi-open site 8 are also high in Poaceae pollen and plot with the open grassland samples. Sample 17 has been taken in a small forest opening with grasses that may have contributed to its high Poaceae pollen proportion. Sample 8 is situated at the forest edge and was therefore classified as semi-open, while the local abundance of grasses may explain the position in the PCA. This comparison of the pollen and vegetation composition in the PCA analysis indicates that the pollen composition at each site is strongly influenced by the vegetation beyond the 100 m radius used here for comparison.
Representation factors (R-values)
A visual comparison of the average pollen proportions over all study sites with the average vegetation proportions in the 100 m radius of the sampling points (Fig. 4a–d) shows that Poaceae is the most overrepresented and A. angustifolia the most underrepresented taxon. Striking are the similarities in average R-values between the two regions, also when using the vegetation cover in an overall 2 km buffer around all sites (Fig. 5). The R-values for the six different taxa follow the same trend in both study regions except for an under-representation of Asteraceae p.p. in Aparados da Serra, while this taxon has a near one to one average pollen to average vegetation proportion in Vila Velha. In both regions, the pollen of A. angustifolia is strongly under-represented; Baccharis and the forest group are under-represented. Pollen from Cyperaceae and Poaceae are over-represented.
Vegetation and pollen proportions at individual sites show a large scatter. The site-based ratios follow the same pattern as the ratio based on averages (Table 3). The scatter of values around the mean is generally higher in Aparados da Serra, particularly for Asteraceae p.p. and A. angustifolia.
ERV analysis
Relative pollen productivity is expressed in relation to a reference taxon, which needs to occur in all pollen and vegetation samples. Poaceae fulfils this criterion in many study regions and is therefore commonly used as a reference taxon (Broström et al. 2008). However, as presented above, high proportions of Poaceae occur in samples of all vegetation types. The samples from Aparados da Serra do not contain a gradient of Poaceae pollen percentages long enough for successful ERV calculations. This situation led to spuriously high PPEs in some analyses. Thus, we have selected Cyperaceae as an alternative reference taxon, which is also present in all vegetation plots while showing a stronger gradient in the vegetation and pollen data.
In addition to two different reference taxa, PPEs were estimated using two different pollen dispersal models (GPM and LSM) implemented in two different programs (ERV.Analysis.v 2.5.3-D) and ERV Analysis in R-R). ERV results were evaluated considering the vegetation data in circles of different radii from the sampling point. Here we represent the results for the maximum distance of 100 m (Fig. 6). Ideally, the accuracy of ERV calculations should improve with more rings added in the analysis, i.e. a larger vegetation sampling area. However, in both study regions, the distance between modelled and empiric pollen values is lowest with the 50–100 m rings (Fig. 7), indicating that the ERV model performs best with only rings up to 100 m radius included in the analysis. This observation may indicate that vegetation composition has not been adequately described for rings beyond 100 m radius. Hence, the 100 m radius represents a good compromise of the higher data accuracy of the field-based vegetation survey and the representation of adjacent woodlands. Results are therefore discussed for this distance. Complete results, including spurious values, are presented in ESM Table 3.
The results of the ERV analysis differ consistently for the dispersal models regardless of the reference taxon. The difference is most pronounced for A. angustifolia, the taxon with the largest pollen grains and hence the highest fall speed (Table 4). For both study areas, pollen productivity of A. angustifolia is as high, or higher than pollen productivity of the reference taxon Poaceae or Cyperaceae when calculated with the GPM. In calculations with the LSM, pollen productivity of A. angustifolia is much lower. Still, LSM-PPEs of A. angustifolia are higher in Aparados da Serra than in Vila Velha. For the other taxa, differences in GPM- and LSM-PPEs are smaller. For the remaining taxa, both models result in different PPEs between regions, which, at least in the case of Baccharis-type and the forest taxon, can be explained by different species involved and agree with the regional differences in R-values.
Scatter plots (Fig. 8) of modelled against empiric pollen data indicate for Vila Velha good correspondence between pollen and vegetation abundances for Baccharis-type, and poor correspondence for the forest taxon and Poaceae. In Aparados da Serra A. angustifolia and the forest taxon show the best relationship between the variance in vegetation and pollen proportions.
Discussion
Representation of openness in the pollen spectra from the Araucaria forest-grassland mosaic
The Araucaria-grassland mosaic in southern Brazil is characterized by well-defined forest edges, clearly separating open and semi-open vegetation from woodlands. Despite that, it appears challenging to separate these two vegetation types in pollen data using ordination and classification techniques. These difficulties may be due to Poaceae and A. angustifolia pollen showing only a weak local signal, corresponding to the vegetation in a 100 m radius. In the case of Poaceae, this could be caused by species of the family frequently growing within the forest and dominating the surrounding grasslands. Both at Vila Velha and Aparados da Serra, the woodlands contained bamboo of the genus Merostachys and Chusquea.
The other effect obscuring the local separation of the two vegetation types is the different height at which the pollen is released, often described as the “edge effect” of woodlands leading to a spread of tree pollen over a larger area away from the forest (Bunting et al. 2013). Araucaria trees often raise their branches over the canopy of other trees so that perhaps a larger proportion of its pollen is transported above the canopy (Rempe 1937). In Aparados da Serra, the pollen of several trees growing below the canopy of Araucaria was found more frequently within or close to woodlands and was therefore identified as good indicators for forested sites. Schinus is the only tree taxon in Vila Velha consistently more abundant in samples from woodlands, while in Vila Velha Baccharis-type pollen was a reliable indicator of open vegetation. The different land use activities can explain the lack of abundant Baccharis-type pollen in Aparados da Serra, where Baccharis shrubs are suppressed by grazing and frequent fires (Rambo 1956).
The above mentioned weak local pollen signal of Poaceae and A. angustifolia has been observed in other studies from southern Brazil. For instance, a pollen vegetation comparison in the Araucaria-grassland mosaic in Santa Catarina State (Cárdenas et al. 2019) revealed no correspondence between pollen proportions and local site characteristics, including open and closed vegetation. Jeske-Pieruschka et al. (2010) only indicated small differences in pollen composition inside and outside the woodland in a study of 18 surface samples crossing an Araucaria dominated forest patch in Rio Grande do Sul State.
While grasses are the most abundant taxon in open vegetation types of South America and make up nearly one-third of the plant cover in the two study areas, they also act as high pollen producers, being the most abundant pollen type in the air. This high regional deposition of Poaceae pollen results in the pollen type also being abundant in surface samples from woodland patches with low grass cover. In the Neotropics, Bush (2002) observed that Poaceae pollen is dominant where trees with high pollen production are lacking. Nevertheless, Poaceae pollen provides an important measure to separate different ecosystems at the landscape scale (Rodrigues et al. 2016) and was an indicator taxon for Araucaria forest and high elevation grassland in an analysis of 196 surface samples from eastern Brazil (Montade et al. 2016). In a surface sample study from southern South America, Paez et al. (2001) observed that Poaceae pollen is abundant in samples from A. araucana populations, the second species in the genus with similar pollen morphology to A. angustifolia.
Representation of pollen taxa and pollen productivity
Campos vegetation is characterized by high abundances of Asteraceae, particularly Baccharis, Cyperaceae and the dominating Poaceae. Both Poaceae and Cyperaceae have R-values of above one, i.e. they are over-represented in pollen records. Plants with such high pollen representation are generally anemophilous, with numerous exposed anthers, small pollen grains (30–40 µm), and high pollen production (Davis 1984; Hjelle 1997). Also, conifers are usually over-represented in the pollen record as they are wind-pollinated, as is A. angustifolia. However, the tree is dioecious with rather large (60 µm), non-saccate pollen grains resulting in the highest estimated fall speed among the taxa investigated here.
Pollen trapping in A. angustifolia stands of north-eastern Argentina yielded pollen deposition values between 3,000 and 14,000 grains cm−1 year−1 in a situation directly below flowering trees (Simón et al. 2018). These values are similar to the average pollen influx measured in A. angustifolia dominated forest in the province Santa Catarina, Southern Brazil (Behling 1997) and compare to pollen deposition of trees in the genus Pinus in Europe (Abraham et al. 2020). A gene flow study of an isolated group of 11 Araucaria trees documents that the large pollen type is well dispersed and most trees in that group were fertilized by pollen travelling around 2 km from the nearest more extensive woodland (Bittencourt and Sebbenn 2007). In the study presented here, it was not feasible to identify all trees in the woodland. Although species of the genus Mimosa, Myrsine, Clethra and Myrtaceae family were also recorded, their abundance in the small areas mapped was often too low for reliable comparisons to their equally low pollen percentages. The combined forest taxa group yielded higher R-values compared to A. angustifolia. This result may be due to these trees producing more pollen than A. angustifolia.
The different R-values for the forest group between Vila Velha and Aparados da Serra are likely due to differences in forest composition, with higher abundances of pollen from Myrtaceae, Ilex and Myrsine in Aparados da Serra in the pollen samples. Cárdenas et al. (2019) indeed estimate higher R-values for several tree taxa, including Clethra, Myrsine and Podocarpus, compared to A. angustifolia. Also, taxa including the Myrtaceae family, identified as low pollen producers, revealed high R-values in other studies (Gosling et al. 2009; Cárdenas et al. 2019). These differences indicate that pollen production and dispersion differ substantially among species of this family. Moreover, many forest trees are palynologically silent (Gosling et al. 2009; Cárdenas et al. 2019), which hampers drawing any further conclusion from the forest group taxon.
Gosling et al. (2009) also report small R-values for Poaceae, which may be partially due to the available vegetation data coming mainly from tree inventories. As discussed above, pollen productivity of Poaceae may indeed be low compared to the trees present in a particular study region (Bush 2002). The calculation of R-values does not account for pollen dispersal. Hence, the difference in R-values between taxa may relate to differences in pollen productivity or pollen dispersal. PPE calculation does account for pollen dispersion by distance weighting plant abundances. As long as a suitable dispersal model is applied in the calculations, PPEs quantify differences in pollen productivity between taxa. Only A. angustifolia represents a single species in vegetation and pollen data, while even the genus Baccharis combines species with different life forms present in different abundances in the two study areas.
The choice and parameterization of the pollen dispersal model is the most important factor influencing the resulting pollen productivity estimates (Theuerkauf et al. 2013). The decrease in pollen contribution of a given plant with distance from the point of reference may be described by simple particle dispersal models such as GPMs (Bunting et al. 2013). For several regions, LSMs (Kuparinen et al. 2007) have shown to describe pollen deposition more realistically than GPMs (Theuerkauf et al. 2013; Mariani et al. 2016). In this study, dispersal model selection strongly affected A. angustifolia, with GPM-PPEs being much higher than the LSM-PPEs. This difference is due to the large difference in fall speed. The fall speed of pollen has a significant influence on the predicted dispersal pattern in the GPM but not as strong in the LSM (Fig. 9). The GPM model predicts that most A. angustifolia pollen would be deposited within metres of the parent tree, which is clearly not observed in the field. Because the GPM underestimated the dispersal distances of A. angustifolia pollen, the resulting PPEs are unrealistically high. Both models assume horizontally homogeneous canopies, which is violated in the current study where groups of A. angustifolia in the open grassland are more similar to smokestacks with respect to their pollen release. In this parkland situation with pollen being released at different heights and with edge effects occurring at forest margins, also the chosen LSM model may not be fully suited.
Nevertheless, the estimate of a pollen productivity for A. angustifolia of 0.8 relative to Poaceae for Aparados da Sierra based on the LSM model may be realistic considering that about half of the trees are female therefore non-pollen-producing. The A. angustifolia PPE of 0.1 in Villa Velha instead appears too low. For this study area, the scatterplots do not show the expected 1:1 relationship between modelled and empiric pollen values (Fig. 8). This pattern indicates that the relationship between abundances of A. angustifolia in the vegetation and pollen deposition is not that close. For example, pollen deposition is highest in site 16 (17.7%), although A. angustifolia is only intermediately abundant within 100 m distance from that site. The poor pollen vegetation relationship for A. angustifolia in Vila Velha may relate to the location of this area near the distribution limit of the tree. Near this limit, the growth and pollen production of trees may be more variable and influenced by local site conditions. Also, the age of a tree determines its pollen production (Matthias et al. 2012). Differences in age-structure may hence cause biases in the pollen vegetation relationship.
Similarly, pollen production will be related to the ratio of male versus female trees, which may differ between sites. Regardless, the inadequacy of the dispersal models considering the different release height of pollen is likely the major problem in this mosaic landscape. Developing dispersal models incorporating the effect of release height might improve pollen vegetation relationships in these landscapes.
Conclusions
The application of procedures designed in high latitude vegetation types of the northern hemisphere to estimate pollen productivity in the low latitudes of South America is challenging but possible. This study shows that it is possible for the Araucaria forest-grassland mosaic of southern Brazil, while documenting several problems for this particular vegetation type. Poaceae is the most overrepresented taxon in the Araucaria forest-grassland mosaic of southern Brazil. Grasses occur in all vegetation types with different cover abundance, while their pollen proportion in surface samples is not providing clear differences between forest and open areas. Locally open vegetation types can be detected by Baccharis-type pollen indicating open conditions and Araucaria and Celtis pollen indicating woodland.
Similarities in R-values and PPEs between regions document that pollen production of individual species within the large plant families of Poaceae and Asteraceae is similar, allowing the application of resulting PPEs on the broader region. Conversely the forest group consisting of trees from different families showed a different behaviour between the study areas. Here it seems necessary to develop PPEs for at least groups of trees with similar pollen dispersal syndromes. The dioecious nature of A. angustifolia hampers estimating pollen productivity of this iconic tree. The obtained productivity of 0.8 with respect to Poaceae is lower than the pollen productivity of the general forest, resulting in the tree being underrepresented by its pollen even though it is wind-pollinated.
The application of pollen dispersal models designed for closed canopies in the natural parkland situation with pollen released at different levels may explain some difficulties in estimating PPEs. Further model development needs to include the effect of pollen release at different heights.
The insights and obtained PPEs for 6 major pollen taxa are now available for exploring the increasing number of pollen diagrams from the region and perhaps refining previous interpretations.
Data availability
After publication pollen data will be submitted to the Latin American Pollen Database.
References
Abraham V, Hicks S, Svobodová-Svitavská H et al (2020) Patterns in recent and Holocene pollen influxes across Europe; the Pollen Monitoring Programme Database as a tool for vegetation reconstruction. Biogeosci Discuss. https://doi.org/10.5194/bg-2020-217
Alvares CA, Stape JL, Sentelhas PC et al (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Andersen ST (1970) The relative pollen productivity and pollen representation of North European trees, and correction factors for tree pollen spectra determined by surface pollen analyses from forests. C.A. Reitzel, Kopenhagen
Auer V (1927) Untersuchungen über die Waldgrenzen und Torfböden in Lappland. Comm ex inst quaest forest 12:1–46,47–52
Baptista LRM, Irgang BE, Vals JFM, Waechter JL (1979) Parque Nacional dos Aparados da Serra. Levantamento de vegetação. In: Plano de Manejor do Parque Nacional de Aparados da Serra. 1984. IBDF Porto Alegre, p 295
Bartlein PJ, Harrison SP, Brewer S et al (2011) Pollen-based continental climate reconstructions at 6 and 21 ka: a global synthesis. Clim Dyn 37:775–802. https://doi.org/10.1007/s00382-010-0904-1
Bauermann SG, Nunes Radaeski J, Cardoso Pacheco Evaldt A et al (2013) Pólen nas angiospermas diversidade e evolução. Editora da ULBRA, Universidade Luterana do Brasil, Canoas
Behling H (1997) Late Quaternary vegetation, climate and fire history of the Araucaria forest and Campos region from Serra Campos Gerais, Paraná State (South Brazil). Rev Palaeobot Palynol 97:109–121. https://doi.org/10.1016/S0034-6667(96)00065-6
Behling H, Pillar VD, Orlóci L, Bauermann SG (2004) Late Quaternary Araucaria forest, grassland (Campos), fire and climate dynamics, studied by high-resolution pollen, charcoal and multivariate analysis of the Cambará do Sul core in southern Brazil. Palaeogeogr Palaeoclimatol Palaeoecol 203:277–297. https://doi.org/10.1016/S0031-0182(03)00687-4
Bittencourt JVM, Sebbenn AM (2007) Patterns of pollen and seed dispersal in a small, fragmented population of the wind-pollinated tree Araucaria angustifolia in southern Brazil. Heredity 99:580–591. https://doi.org/10.1038/sj.hdy.6801019
Broström A, Sugita S, Gaillard M-J (2004) Pollen productivity estimates for the reconstruction of past vegetation cover in the cultural landscape of southern Sweden. Holocene 14:368–381. https://doi.org/10.1191/0959683604hl713rp
Broström A, Nielsen AB, Gaillard M-J et al (2008) Pollen productivity estimates of key European plant taxa for quantitative reconstruction of past vegetation: a review. Veget Hist Archaeobot 17:461–478. https://doi.org/10.1007/s00334-008-0148-8
Bunting MJ, Farrell M, Broström A et al (2013) Palynological perspectives on vegetation survey: a critical step for model-based reconstruction of Quaternary land cover. Quat Sci Rev 82:41–55. https://doi.org/10.1016/j.quascirev.2013.10.006
Bush MB (2002) On the interpretation of fossil Poaceae pollen in the lowland humid neotropics. Palaeogeogr Palaeoclimatol Palaeoecol 177:5–17. https://doi.org/10.1016/S0031-0182(01)00348-0
Bush MB, Rivera R (1998) Pollen dispersal and representation in a neotropical rain forest. Glob Ecol Biogeogr Lett 7:379–392. https://doi.org/10.2307/2997685
Bush MB, Weng C (2007) Introducing a new (freeware) tool for palynology. J Biogeogr 34:377–380. https://doi.org/10.1111/j.1365-2699.2006.01645.x
Cárdenas ML, Wilson OJ, Schorn LA et al (2019) A quantitative study of modern pollen–vegetation relationships in southern Brazil’s Araucaria forest. Rev Palaeobot Palynol 265:27–40. https://doi.org/10.1016/j.revpalbo.2019.03.003
Cervi AC, von Linsingen L, Hatschbach G, Ribas OS (2007) A vegetação do Parque Estadual de Vila Velha, município de Ponta Grossa, Paraná, Brasil. Bol Mus Bot Munic 69:01–52
Colombo AF, Joly CA (2010) Brazilian Atlantic Forest lato sensu: the most ancient Brazilian forest, and a biodiversity hotspot, is highly threatened by climate change. Braz J Biol 70:697–708. https://doi.org/10.1590/S1519-69842010000400002
Dalazoana K, da Silva MA, Moro RS (2007) Comparação de Três Fisionomias de Campo Natural no Parque Estadual de Vila Velha, Ponta Grossa. PR Braz J Biosci 5(Suppl 1):675–677
Davis MB (1963) On the theory of pollen analysis. Am J Sci 261:897–912
Davis OK (1984) Pollen frequencies reflect vegetation patterns in a great basin (U.S.A) mountain range. Rev Palaeobot Palynol 40:295–315. https://doi.org/10.1016/0034-6667(84)90013-7
Davis BAS, Zanon M, Collins P et al (2013) The European Modern Pollen Database (EMPD) project. Veget Hist Archaeobot 22:521–530. https://doi.org/10.1007/s00334-012-0388-5
Dawson A, Paciorek CJ, McLachlan JS et al (2016) Quantifying pollen-vegetation relationships to reconstruct ancient forests using 19th-century forest composition and pollen data. Quat Sci Rev 137:156–175. https://doi.org/10.1016/j.quascirev.2016.01.012
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366. https://doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
Flantua SGA, Hooghiemstra H, Grimm EC et al (2015) Updated site compilation of the Latin American Pollen Database. Rev Palaeobot Palynol 223:104–115. https://doi.org/10.1016/j.revpalbo.2015.09.008
Fontana SL (2005) Coastal dune vegetation and pollen representation in south Buenos Aires Province, Argentina. J Biogeogr 32:719–735. https://doi.org/10.1111/j.1365-2699.2004.01221.x
Gaillard M-J, Sugita S, Bunting MJ et al (2008) The use of modelling and simulation approach in reconstructing past landscapes from fossil pollen data: a review and results from the POLLANDCAL network. Veget Hist Archaeobot 17:419–443. https://doi.org/10.1007/s00334-008-0169-3
Gaillard M-J, Sugita S, Mazier F et al (2010) Holocene land-cover reconstructions for studies on land cover-climate feedbacks. Clim past 6:483–499. https://doi.org/10.5194/cp-6-483-2010
Gosling WD, Mayle FE, Tate NJ, Killeen TJ (2005) Modern pollen-rain characteristics of tall terra fume moist evergreen forest, southern Amazonia. Quat Res 64:284–297. https://doi.org/10.1016/j.yqres.2005.08.008
Gosling WD, Mayle FE, Tate NJ, Killeen TJ (2009) Differentiation between Neotropical rainforest, dry forest, and savannah ecosystems by their modern pollen spectra and implications for the fossil pollen record. Rev Palaeobot Palynol 153:70–85. https://doi.org/10.1016/j.revpalbo.2008.06.007
Gregory PH (1973) The microbiology of the atmosphere. Leonard Hill, Aylesbury
Harrison SP, Gaillard M-J, Stocker BD et al (2020) Development and testing scenarios for implementing land use and land cover changes during the Holocene in Earth system model experiments. Geosci Model Dev 13:805–824. https://doi.org/10.5194/gmd-13-805-2020
Haselhorst DS, Moreno JE, Punyasena SW (2020) Assessing the influence of vegetation structure and phenological variability on pollen-vegetation relationships using a 15-year Neotropical pollen rain record. J Veg Sci 31:606–615. https://doi.org/10.1111/jvs.12897
Hjelle KL (1997) Relationships between pollen and plants in human-influenced vegetation types using presence-absence data in western Norway. Rev Palaeobot Palynol 99:1–16. https://doi.org/10.1016/S0034-6667(97)00041-9
Iriarte J, Behling H (2007) The expansion of Araucaria forest in the southern Brazilian highlands during the last 4000 years and its implications for the development of the Taquara/Itararé Tradition. Environ Archaeol 12:115–127. https://doi.org/10.1179/174963107x226390
Jeske-Pieruschka V, Fidelis A, Bergamin RS et al (2010) Araucaria forest dynamics in relation to fire frequency in southern Brazil based on fossil and modern pollen data. Rev Palaeobot Palynol 160:53–65. https://doi.org/10.1016/j.revpalbo.2010.01.005
Jones HT, Mayle FE, Pennington RT, Killeen TJ (2011) Characterization of Bolivian savanna ecosystems by their modern pollen rain and implications for fossil pollen records. Rev Palaeobot Palynol 164:223–237. https://doi.org/10.1016/j.revpalbo.2011.01.001
Kuparinen A (2006) Mechanistic models for wind dispersal. Trends Plant Sci 11:296–301. https://doi.org/10.1016/j.tplants.2006.04.006
Kuparinen A, Markkanen T, Riikonen H, Vesala T (2007) Modeling air-mediated dispersal of spores, pollen and seeds in forested areas. Ecol Model 208:177–188. https://doi.org/10.1016/j.ecolmodel.2007.05.023
Ledru M-P (2002) Late quaternary history and evolution of the cerrados as revealed by palynological records. In: Oliveira P, Marquis R (eds) The Cerrados of Brazil: ecology and natural history of neotropical savanna. Columbia University Press, New York, pp 33–50
Marchant R, Cleef A, Harrison SP et al (2009) Pollen-based biome reconstructions for Latin America at 0, 6000 and 18 000 radiocarbon years ago. Clim past 5:725–767. https://doi.org/10.5194/cp-5-725-2009
Mariani M, Connor SE, Theuerkauf M et al (2016) Testing quantitative pollen dispersal models in animal-pollinated vegetation mosaics: an example from temperate Tasmania, Australia. Quat Sci Rev 154:214–225. https://doi.org/10.1016/j.quascirev.2016.10.020
Masciadri S, Stutz S, García-Rodríguez F (2013) Modern pollen–vegetation relationship of plant communities in the Uruguayan Atlantic coast. Braz J Bot 36:31–44. https://doi.org/10.1007/s40415-013-0006-5
Matthias I, Nielsen AB, Giesecke T (2012) Evaluating the effect of flowering age and forest structure on pollen productivity estimates. Veget Hist Archaeobot 21:471–484. https://doi.org/10.1007/s00334-012-0373-z
McMichael CH, Piperno DR, Bush MB et al (2012) Sparse Pre-Columbian human habitation in Western Amazonia. Science 336:1,429-1,431. https://doi.org/10.1126/science.1219982
Medeanic S, Costa CSB, Diniz D (2016) Modern pollen–vegetation relationships in saltmarsh habitats along a salinity gradient of a fluvial estuary. Rev Palaeobot Palynol 233:67–76. https://doi.org/10.1016/j.revpalbo.2016.07.010
MMA, IBAMA (2007) Plano de Manejo Parque Nacional de Aparados da Serra e Serra Geral. Brasil, p 211
Mocochinski AY, Scheer MB (2008) Campos de altitude na serra do mar paranaense: aspectos florísticos. Floresta 38:625–640. https://doi.org/10.5380/rf.v38i4.13158
Montade V, Diogo IJS, Bremond L et al (2016) Pollen-based characterization of montane forest types in north-eastern Brazil. Rev Palaeobot Palynol 234:147–158. https://doi.org/10.1016/j.revpalbo.2016.07.003
Montade V, Ledru M-P, Giesecke T et al (2019) A new modern pollen dataset describing the Brazilian Atlantic Forest. Holocene 29:1,253-1,262. https://doi.org/10.1177/0959683619846981
Oksanen J, Blanchet G, Friendly M et al (2017) Vegan: community ecology package. R package version 2.4–3. https://cran.r-project.org. Retrieved 1 Dec 2019
Paez MM, Schäbitz F, Stutz S (2001) Modern pollen-vegetation and isopoll maps in southern Argentina: modern pollen in Argentina. J Biogeogr 28(997–1):021. https://doi.org/10.1046/j.1365-2699.2001.00616.x
Parsons RW, Prentice IC (1981) Statistical approaches to R-values and the pollen— vegetation relationship. Rev Palaeobot Palynol 32:127–152. https://doi.org/10.1016/0034-6667(81)90001-4
Prentice IC (1985) Pollen representation, source area, and basin size: toward a unified theory of pollen analysis. Quat Res 23:76–86. https://doi.org/10.1016/0033-5894(85)90073-0
QGIS (2019) QGIS 3.4 Geographic information system user guide. Open Source Geospatial Foundation Project. http://www.qgis.org. Retrieved 1 Mar 2019
Rambo B (1956) A flora fanerogâmica dos Aparados riograndenses. Sellowia 7:235–298
Rempe H (1937) Untersuchungen über die Verbreitung des Blütenstaubes durch die Luftströmungen. Planta 27:93–147. https://doi.org/10.1007/BF01939376
Roberts N, Fyfe RM, Woodbridge J et al (2018) Europe’s lost forests: a pollen-based synthesis for the last 11,000 years. Sci Rep 8:716. https://doi.org/10.1038/s41598-017-18646-7
Rodrigues JM, Behling H, Giesecke T (2016) Differentiating vegetation types from eastern South American ecosystems based on modern and subfossil pollen samples: evaluating modern analogues. Veget Hist Archaeobot 25:387–403. https://doi.org/10.1007/s00334-016-0558-y
Roubik D, Moreno J (1991) Pollen and spores of Barro Colorado Island. Missouri Botanical Garden, St. Louis, p 268
Silva FHM, Santos FAR, Lima LCL (2016) Flora polínica das Caatingas: Estação biológica de canudos (Canudos, Bahia, Brasil), 1st edn. Micron, Feira de Santana
Simón BE, Latorre F, Rotundo C (2018) Study of the reproductive phenology of Araucaria angustifolia in two environments of Argentina: its application to the management of a species at risk. Glob Ecol Conserv 16:e00483. https://doi.org/10.1016/j.gecco.2018.e00483
Strandberg G, Bärring L, Hansson U et al (2014) CORDEX scenarios for Europe from the Rossby Centre regional climate model RCA4. SMHI, Norrköping
Sugita S (1993) A model of pollen source area for an entire lake surface. Quat Res 39:239–244. https://doi.org/10.1006/qres.1993.1027
Sugita S (2019) ERV. Analysis.v.2.5.x.exe. Retrieved 21 Sept 2019
Theuerkauf M, Kuparinen A, Joosten H (2013) Pollen productivity estimates strongly depend on assumed pollen dispersal. Holocene 23:14–24. https://doi.org/10.1177/0959683612450194
Theuerkauf M, Couwenberg J (2021) Pollen productivity estimates strongly depend on assumed pollen dispersal II: extending the ERV model. The Holocene: under review. First decision: Major revision
Whitney BS, Smallman TL, Mitchard ET et al (2019) Constraining pollen-based estimates of forest cover in the Amazon: a simulation approach. Holocene 29:262–270. https://doi.org/10.1177/0959683618810394
Williams JW, Grimm EC, Blois JL et al (2018) The neotoma paleoecology database, a multiproxy, international, community-curated data resource. Quat Res 89:156–177. https://doi.org/10.1017/qua.2017.105
Zanon M, Davis BAS, Marquer L et al (2018) European Forest Cover During the Past 12,000 Years: a palynological reconstruction based on modern analogs and remote sensing. Front Plant Sci 9:253. https://doi.org/10.3389/fpls.2018.00253
Acknowledgements
We are grateful to Vivian L. Jeske-Pieruschka for the assistance in the field and for organizing the permits to work in the parks: Instituto Ambiental do Paraná, Number: 55.16 (Vila Velha) SISBIO, Number 63378-1 (Aparados da Serra). We also thank Shinya Sugita for his suggestions and valuable advice while processing part of our data. The research was funded by the Deutsche Forschungsgemeinschaft, DFG grant GI 732/8-1. This study is a contribution to LandCover6k, a working group of the Past Global Changes (PAGES) project, which in turn received support from the Swiss National Science Foundation, the US National Science Foundation, the Swiss Academy of Sciences and the Chinese Academy of Sciences
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Gosling.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Piraquive Bermúdez, D., Theuerkauf, M. & Giesecke, T. Towards quantifying changes in forest cover in the Araucaria forest-grassland mosaic in southern Brazil. Veget Hist Archaeobot 31, 107–122 (2022). https://doi.org/10.1007/s00334-021-00841-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-021-00841-2