Abstract
We present the postglacial history of vegetation, human activities and changes in lake level in the context of climate change in northeast Poland from ~14,000 cal. b.p. to the present day. The palaeoecological reconstruction is based on the results of high-resolution plant macrofossil analyses as well as records from pollen, Cladocera and radiocarbon dating. Climate fluctuations and human activity have caused many changes in vegetation development in Jezioro Linówek and in the vicinity of this lake. The Early Holocene warming that occurred at ~9500 b.c. caused an increase in Betula and the colonisation of Linówek by Potamogeton lucens, Nymphaea alba and Chara sp. At ~2300 b.c., climate cooling was accompanied by the spread of Picea abies and the appearance of Potamogeton alpinus and Nuphar pumila in the lake. The first traces of farming in the form of Cerealia pollen have been dated back to ~2100 b.c. The cultivation of Triticum began at ~250 b.c., Secale at ~a.d. 550, and Fagopyrum at ~a.d. 1720. The rapid increase in human activity at ~a.d. 1700 and the simultaneous loss of woodland is associated with the establishment of villages in the area and is expressed by the decline of tree curves. In Linówek, which was formed ~14,000 cal. b.p., three periods of high water level occurred (12000–9400, 7000–4000 and 1450 b.c.–a.d. 650), and two periods of low water level (9400–7100 and 3700–1700 b.c.). The changes of water level correspond well with other sites in central and northern Europe.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Because of its location, the area of northeastern Poland where our research site is located is favourable for palaeoecological studies. This region is under the influence of a transitional climate, so it shows both oceanic and continental features. The occurrences of different air masses have a wide range of influences upon the natural distributions of plants typical of both the oceanic (Cladium mariscus and Juncus subnodulosus) and the boreal climate (Picea abies, Polemonium caeruleum, Nymphaea candida and Nuphar pumila). It is very important that the studied region serves as a migration gateway through which plants have moved in north–south–north and east–west–east directions during the last several thousand years of the Holocene. The postglacial changes in vegetation became apparent in the study area during earlier research, as illustrated by the phenomenon of glacial–interglacial cycles (Iversen 1964; Tobolski 1976; Birks 1986; Lang 1994) and the migration of plants with different climatic requirements. At the beginning of the 20th century, it was shown that Juncus subnodulosus (Gałka 2009), a plant typical of the oceanic zone (Meusel et al. 1965), was still growing in this area, but its current distribution is limited to northwest Poland (Markowski and Stasiak 1988). However, new species have entered the area, such as Sphagnum wulfianum (Gałka 2010), which has its optimal habitat in the boreal zone (Daniels and Eddy 1990). Good examples of the withdrawal of oceanic species from this part of Europe include the withdrawal of Cladium mariscus, which is disappearing from the studied area (Gałka and Tobolski 2012) and Najas flexilis, whose occurrence was limited to the climatic optimum (Gałka et al. 2012). Considering these examples, it should be noted that palaeoecological research in this area is a key to understanding the mechanisms of plant migration, including the time of their appearance, the length of their existence, and the causes of their withdrawal from central Europe.
The Suwałki region of northeast Poland also has a unique history of human settlement. Specialists in human history have demonstrated the specific and unique character of economic activity in northeast Poland in relation to central Europe (Nowakowski 1986; Kozłowski and Kaczanowski 1998; Karczewski 2011). In spite of intensive archaeological research conducted in the area, mainly focused on the history of Jatvingian settlement, related archaeobotanical knowledge is poor. So far, macrofossils of cultivated plants have been analysed from two archaeological sites—Osinki, 15 km southeast of Linówek, and Osowa, 10 km south of Linówek. At one site—Góra Zamkowa, ~3 km east of Linówek, a find of various cereal grains was reported (Karczewski 2011). At two sites, Korklin (Stasiak 1970) and Wigry (Kupryjanowicz 2007), pollen analysis identified cultivated cereals.
The results presented in this article are based on high-resolution research of plant macroremains, pollen and Cladocera records, as well as radiocarbon dates on Jezioro Linówek sediments and the peat bog that has developed around it. The aim of the study was to reconstruct: (1) the postglacial history of vegetation in northeast Poland at regional and local scales, (2) the history of the development of Linówek and fluctuations in its water level, and (3) the history of human economic activity and its impact on vegetation.
So far, a postglacial vegetation history of this area covering the period from the withdrawal of the Scandinavian ice sheet to the present day has not yet been published. Multidirectional research was carried out on another lake, Jezioro Hańcza, located ~4 km from Linówek, but the time span of the palaeoenvironmental and palaeoclimatic reconstructions in this research was ~13,000–4,000 cal. b.p. (Lauterbach et al. 2011). Multi-proxy palaeoecological studies involving Late-glacial and Holocene environmental changes were carried out on another lake, Jezioro Wigry, located ~30 km from the study site (Kupryjanowicz 2007; Zawisza and Szeroczyńska 2007). The first pollen studies were conducted in the Suwałki region by Ołtuszewski (1937) and in the Puszcza Romincka (25 km to the north) by Gross (1935). The nearest palaeoecological and palaeobotanical reconstructions that include the Late-glacial and Holocene periods were carried out in Lithuania (Stančikaitė et al. 2008, 2009; Gaidamavičius et al. 2011) and western Belarus (Stančikaitė et al. 2011).
Study site
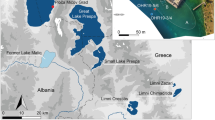
The study area is located in northeast Poland, in the Suwalski Park Krajobrazowy (Suwałki Landscape Park, SLP), ~60 km north of the extent of the last glaciation (Krzywicki 2002; Marks 2002). The current landforms were formed by the receding ice sheet as well as fluvioglacial water activity and glaciotectonics (Ber 1974, 2000; Krzywicki 2002). Typical Late-glacial landforms in the vicinity of Linówek include kame hills, eskers and depressions with lakes or peat bogs, and with a height range >100 m. The warmest month is July with an average 17 °C and the coldest is January with the average −5 °C (Woś 1999). The study site displays features of a temperate transitional climate, with clear characteristics of a continental climate. The mean annual air temperature is 2 °C lower than that of western Poland (Lorenc 2005), and the total mean annual precipitation is >650 mm, with the maximum occurring in July and the minimum in February. The current vegetation shows distinct differences compared with the other parts of Poland, associated with the strong impacts of the continental climate. The specificity of the vegetation allows it to be placed in a separate Polish geobotanical region.
The Suwalski Park Krajobrazowy is located in the most northeasterly part of Poland in the area close to the boreomeridional zone (Moen 1999) and in the range of northern spruce woodland (the northern boreal range—Bortynski 2007). The majority of the seven woodland complexes in the SLP are fresh mixed Corylo-Piceetum woods and pine-spruce mixed coniferous Calamagrostio arundinaceae-Piceetum woods (Sokołowski 1973). In addition, Picea abies is a component of the oak-hornbeam woods (Tilio-Carpinetum, sub-boreal type), which occurs in the damp mixed woodland (Querco-Piceetum) and forms a stand in the boreal vegetation complex of Sphagno girgensohnii-Piceetum (Matuszkiewicz 2001).
With an area of 2.74 ha, Linówek is located among terminal moraine hills. The water level in the lake stands at 199.7 m a.s.l., and the maximum depth is 4.2 m. The central part of the lake bottom is devoid of vegetation. The lakeshore zone is characterised by Stratiotes aloides, Nymphaea alba, Potamogeton natans and Chara sp., among others, and the shallow margins with rushes are populated by the helophytes Typha latifolia, Equisetum fluviatile and Sparganium sp. A transition mire has developed at the lakeshore, with widespread Sphagnum teres, Oxycoccus palustris and Thelypteris palustris. The occurrence of Sphagnum obtusum, S. fuscum and S. magellanicum as well as Drosera rotundifolia and Scheuchzeria palustris is quite rare. The edge area of the peat bog is dry and overgrown with Betula pubescens and Salix cinerea.
Materials and methods
Coring for laboratory analyses was performed manually using a Russian corer with a tube diameter of 7 cm and length of 100 cm. Two cores were collected from Jezioro Linówek on the eastern shore of the lake (Fig. 1). One of the cores, LL I (Site I, 54°13′24.30″N and 22°50′29.99″E), had a length of 590 cm (200–800 cm) and included lake sediments as well as the organic and mineral sediments found beneath (Table 1). The second core, LL II (Site II), with a length of 200 cm, was collected at a distance of 2.5 m from the location of the first core and included the upper portion of the lake sediments and the peat that developed above them (Table 1). The necessity to collect the upper layer of the sediment from elsewhere was due to the unsuitable nature of the peat deposits in Site I. These deposits were waterlogged and included inserts of water lenses, making the deposits unfit for analysis. The lacustrine-peat sediment was placed in PVC tubes. The sediments were extracted and purified in the laboratory and subsequently divided into layers 1 cm thick with a surgical scalpel.
In order to establish the chronology of the results, macrofossils of terrestrial plants from eight samples were selected for AMS radiocarbon dating (Table 2). The material was collected from 1 cm thick layers of the deposits. In six cases these were tree macrofossils, in one it was trees and peat plants, while in the last one it was a seed of a plant growing in a terrestrial habitat.
The 14C dating was done in the Poznań Radiocarbon Laboratory. The samples were prepared with the standard AAA method, and 14C was measured using the AMS technique (Goslar et al. 2004). The resulting conventional radiocarbon dates were calibrated against the INTCAL09 calibration curve (Reimer et al. 2009). An age-depth model is presented in Fig. 2.
The pollen analyses, on 122 samples, were performed at a variable depth interval resulting from the arrangement of the sediment layers, but mostly a 5 cm resolution. The time resolution of the counted samples fluctuated around 11500 b.c.–a.d. 2010. The results of the pollen analyses are presented in diagrams (Figs. 3, 4), which were calculated and drawn using Tilia software (Grimm 1991). For the pollen studies, 1 cm3 of gyttja or 2 cm3 of peat, due to the low concentration of pollen in the latter, were used. Each sample was acetolysed following the modified Erdtman method with an addition of hydrofluoric acid (Fægri and Iversen 1989). Pollen taxa were identified and counted using a Zeiss AMPLIVAL light microscope with 400× and 1,000× magnifications. Pollen was identified with the use of specialist keys and atlases, particularly Beug (2004). About 500 tree and herb pollen grain taxa per sample were counted. Percentage values of sporomorphs in individual spectra were calculated on the basis of particular taxa values in relation to the total pollen number (AP + NAP), excluding local taxa (cryptogams, limnophytes, telmatophytes and Cyperaceae). The percentage values of local pollen taxa and cryptogams were also calculated in relation to the total pollen sum. The estimation of human economic activity in northeastern Poland was based on plant indicators. Summary curves were made for human activity (anthropogenic) indicators distinguished according to Behre (1981), as well as Van der Linden and Van Geel (2006). The human indicators include Artemisia, Centaurea cyanus, Chenopodiaceae, Plantago lanceolata, Rumex acetosa/acetosella and Cerealia spp. Zonal names, such as Allerød or Younger Dryas, refer to the data defined by Litt et al. (2001).
Percentage pollen diagram from Site II (for lithology, see Table 1)
In the study of plant macroremains, both cores were analysed at a resolution of 1 cm, with a total of 790 samples. The time resolution of the counted samples fluctuated around 12500 b.c.–a.d. 2010. The volume of the samples taken from the core of Site I was around 25 cm3, and from the second core it was about 50 cm3. The samples were rinsed on sieves with 0.25 mm mesh size in warm water. Macrofossils of the selected plants were studied with the use of a Nikon SMZ800 stereoscopic microscope at a magnification of 10–200×, and a transmitted light microscope. Species determination of individual plant macrofossils was done with the help of the following keys: Berggren (1968, 1981), Grosse-Brauckmann (1974), Grosse-Brauckmann and Streitz (1992), Tobolski (2000), Velichkevich and Zastawniak (2006, 2009) and Hölzer (2010). The results are shown in the diagrams of plant macroremains (Figs. 5, 6), which were prepared with the use of the graphics program C2 (Juggins 2003). The results are summarised in the diagrams in absolute numbers, while the vegetative parts of vascular plants as well as brown mosses and Sphagnum are given in percentages of the total volume of the sediment samples. Plant nomenclature follows Mirek et al. (2002).
Plant macrofossil diagram showing local vegetation changes at Site I, description of plant macroremains. f fruits, s seeds, e endocarp, o oospores, fs fruit scales, bs bud scales, n needle, l leaves, ms megaspores (for lithology, see Table 1)
Cladocera: methodology
The analysis of subfossil Cladocera was performed at 10 cm intervals for the limnic part of the sequence (LL I). The sample size was 1 cm3 of fresh sediment. In order to obtain the remains of the Cladocera fauna deposited in the sediment, the samples were prepared according to standard laboratory procedures (Szeroczyńska 1985; Frey 1986; Korhola and Rautio 2001). In the laboratory each sample was placed in a 250 ml glass beaker, to which 100 ml of 10 % KOH was added and boiled on an electric cooker. The sample was then transferred to a magnetic stirrer with heating option up to 80 °C for 25 min, after which the beaker was filled up with distilled water and allowed to stand for at least 12 h. The next day the obtained residue was washed with distilled water on a sieve with a mesh size of 40 μm. If the sample contained Calcium carbonate it was removed on the sieve using 10 % HCl and then washed again with distilled water. The prepared samples were placed in 10 ml tubes and stored in a refrigerator. A few drops of Glycerol-safranin were added to the samples to colour the chitin remains of Cladocera, each time before the microscopic study. The obtained preparations were volatile. Using an automatic pipette, 0.1 ml of the obtained solution was taken and transferred to a glass slide where it was covered with a cover slip measuring 24 × 60 mm. The analysis was performed using an Olympus biological microscope with 10× eyepiece magnification, zoom lens 10, 20, 40×. All the remains of Cladocera which were found were counted: headshields, shells, postabdomens, postabdomenal claws and ephippia. For each sample, the microscopic analysis was performed on 2–4 preparations depending on the amount of remains. The species identification followed Szeroczyńska and Sarmaja-Korjonen (2007) as well as Flössner (2000). The results of the species composition and frequency of individuals are presented in an absolute frequency diagram (Fig. 7), which was drawn up using the graphics program C2 (Juggins 2003).
Results
Lithology and chronology
The age-depth model was constructed with a free-shape algorithm (Goslar et al. 2009), which has generated (using the Monte-Carlo approach) large sets of age-depth curves which are monotonic, as smooth as possible, and at the same time keeping average deviations of 14C dates from the radiocarbon calibration curve close to the 1 σ uncertainties of the dates. For the full set of dates, the conditions above were impossible to fulfill, unless the date from 289 to 290 cm (deviating from the calibration curve by >3 σ, and most probably representing a sample of reworked material) was rejected. Therefore, this date was not included in the diagrams showing the palaeoecological results.
One must mention that the absolute dating was made neither at the bottom of the LL I core nor in the LL II core. In the case of core LL I, the age-depth model starts from 678 cm, where radiocarbon dating gave the result of 11,690 ± 60 b.p. (Fig. 2). The lower layers were not dated due to lack of suitable material for dating and rare presence of pollen in the sediments below 700 cm. The absence of the regular presence of pollen may indicate that the bottom deposits, which are made up of alternate layers of sand and mud, were formed in an environment of turbulent accumulation. The bottom deposit contained few plant macrofossils, and the only observed examples were fragments of leaves of Dryas octopetala as well as an endocarp of Potamogeton filiformis and a fruit of Carex sp. The presence of 14 species of Cladocera is noteworthy and undoubtedly indicates the presence of a lake in the area.
Taking into account the presence of open water taxa in the area (Bosminidae, Daphnidae) as well as the numerous taxa that live in association with water plants (mainly from the family Alonidae, Acroperus sp.), it can be concluded that conditions were unfavourable for the development of the Cladocera fauna at that time. Based on the presence of Cladocera, it can be assumed that this layer was deposited during the Bølling period. However, in the absence of radiocarbon dating and the occasional presence of pollen, no attempt was made to define the age of these sediment layers.
Lake and peat sediment was collected in the second core, which included the top layer of the deposit (200 cm). Two samples from depths of 150 and 80 cm were radiocarbon dated, and the results were used to plot the time curve. The age curve was not continued below a depth of 150 cm, which was dated at ~1280 a.d.
Pollen
The history of vegetation development based on the pollen data is shown in Figs. 3 and 4, and the description of the 11 pollen zones which have been defined is in Table 3. The Late-glacial history of vegetation consists of two phases. The first, Phase LLp-I1, indicates the Bølling-Allerød period (Fig. 3). The older part, up to a depth of 670 cm, is dominated by Betula (up to 57 %) and Hippophaë rhamnoides is also present. This part of the sediments probably accumulated during the Older Dryas. Betula nana, Juniperus, Artemisia and Cyperaceae records reach several percent. In the second part, down to 660 cm, the amount of Pinus rapidly increases (up to 60 %), and the amount of Betula nana, Juniperus, Artemsia and Cyperaceae is reduced. The second phase, LLp-I2, is attributed to the Younger Dryas and is characterised by a high proportion of Juniperus (up to 18 %), Betula nana (up to 10 %), Artemisia (up to 17 %) and Chenopodiaceae (up to 4 %). During this period, the presence of Selaginella, Ephedra fragilis and E. distachya is recorded. Moreover, the share of Pediastrum boryanum and P. kawraiskyi increases. The early Holocene succession with dominant Betula (up to 74 %) and the first record of Ulmus pollen falls within the Pre-boreal period. Potamogeton, Nuphar and Typha latifolia appear in the lake, and the beginning of the explicit expansion of more thermophilous deciduous trees, Corylus, Alnus and Ulmus, occurs in Phase LLp-I4. The 3 % record of Populus is also noteworthy. The development of woods typical of the climatic optimum occurs in Phase LLp-I5, which was dominated by Quercus, Tilia, Ulmus and Corylus, and in wetter places by Fraxinus and Alnus. The second half of this period is characterised by a sharp increase in the amount of Botryoccocus.
The rapid expansion of Picea at 382 cm and the beginning of the constant presence of Carpinus is characteristic of Phase LLp-I6. The end of this phase and beginning of the next phase is also determined by declines of Corylus, Tilia, Ulmus and Fraxinus. The development trend of vegetation is noted during the upper part of Phase LLp-I7 (Fig. 3) and in the bottom part of Phase LLp-II1 (Fig. 4). In both studied cores at this time we noticed the relatively constant presence of Picea (10 %), Quercus (~5 %) and Carpinus (~4 %) and a steady decline in Tilia, Ulmus, Corylus and Acer. During Phase LLp-II1 there are signs of increased human activity which are indicated by increasing Cerealia indet. and the appearance of Triticum type at 175 cm and Secale at 170 cm. At the time of Phase LLp-II1 this part of the lake changed into a peat bog. Development of peat bog sedimentation is evident as a rise of the curves of Cyperaceae and Dryopteris thelypteris. Human activity in the first half of Phase LLp-II2 decreases and then slightly increases the share of trees, Picea, Carpinus and Corylus. In the second half of Phase LLp-II2 Picea and Carpinus increase, and human indicators increase continuously. The constant presence of Secale and Rumex acetosa-acetosella is recorded starting from a depth of 130 cm. At the depth of 100 cm Tricticum-type and Plantago lanceolata pollen appear. The peaks of Picea (34 %) and Carpinus (15 %) are reached at a depth of 85 cm. At 80 cm a significant change in the pollen record is marked by a rapid decline in deciduous trees and Picea, while the amount of NAP increases (up to 36 %). In the first half of this period, the share of Pinus also increases (50 %).
Loss of woodland from the area is shown by the pollen analysis as an increase of cereals and Artemisia. The amount of Secale reaches 17 %, and the changes in the local vegetation are distinctly pronounced, so that Vaccinium sp. appears, and the amount of Sphagnum increases. The development of vegetation most like that of modern times was found at a depth of 47.5 cm and shows a small (Carpinus, Quercus) or occasional (Tilia, Ulmus, Fraxinus) amount of deciduous trees, with Pinus dominating (~60 % at the beginning and end) in addition to Picea (ca. 5 %). In the peat bog, the presence of cf. Andromeda (20 %) increases.
Macrofossils
The development of local vegetation has been divided into 12 phases (Table 4; Figs. 5, 6). Eight phases cover the period when the lake held water, while the remaining four cover the history of the peat bog which then developed there. Potamogeton filiformis, Chara sp. and Batrachium sp. were present in the first phase of lake development (Fig. 5, LLmI-1) during the Late-glacial period. Leaf fragments of Dryas octopetala at 782 cm and a seed of Arctostaphylos uva-ursi at 678 cm were found in Late-glacial deposits in addition to a number of fruits and fruit scales of Betula nana. The Younger Dryas is well marked between 650 and 625 cm. During this period B. nana increases and Potamogeton filiformis and Batrachium sp. appear. In the early Holocene (Pre-boreal period), which includes Phase LLmI-2, there is a significant increase in aquatic plants. Chara sp. increases, numerous pondweeds such as Potamogeton lucens, P. natans and P. crispus appear, and there are signs that Typha sp. grew in the coastal zone near the shore. For the first time there is evidence of trees such as Pinus sylvestris and Betula pubescens.
The Middle Holocene stage includes Phases LLmI-3 and LLmI-4. This period is characterised by significant changes in aquatic vegetation, and the dichotomy of the period is clearly visible. During Phase LLmI-3, new aquatic plants appear, including Ceratophyllum demersum, Najas flexilis, Nuphar lutea, Potamogeton obtusifolius and P. pusillus. However, aquatic vegetation is reduced in Phase LLmI-4 when such rushy plants as Schoenoplectus lacustris appear in addition to vegetation on the edge of the peat bog, including Lycopus europaeus and Menyanthes trifoliata.
The Late Holocene period is defined by lacustrine and peat bog phases. The beginning of Phase LLmI-5 is defined by a significant increase in Chara sp. and Potamogeton obtusifolius. Other Potamogeton species appear later, including P. pusillus and P. alpinus, and Nuphar pumila somewhat earlier. A characteristic of the beginning of this period is the rapid spread of Picea abies around Linówek, as shown by the presence of needles, bud scales and seeds. Macrofossils of Alnus glutinosa and Acer sp. are present for the first time. During the shallowing process of the lake and the development of the peat bog, the proportion of Carex increases, including C. nigra and C. pseudocyperus in addition to Menyanthes trifoliata, and Cladium mariscus appears at a depth of 262 cm.
Nymphaea alba and Nuphar lutea are present in the last phase of the lake existence along with Sparganium minimum and Eleocharis sp. In the first stage of peat bog development (Phase LLmII-2, Fig. 6), the largest share is taken up by such sedges as Carex pseudocyperus, C. paniculata and C. nigra as well as brown mosses such as Drepanocladus sp. and Calliergon sp. A significant change took place in the peat bog along with the appearance of Scheuchzeria palustris in Phase LLmII-2, and the proportion of its vegetative organisms is as high as 80 %.
A key moment in the history of the peat bog is the appearance of Ericaceae, which coincides with an increase in microfossils of Picea abies in Phase LLmII-4. Further changes in the vegetation composition are expressed by the presence of Andromeda polifolia, followed by Sphagnum, among which Sphagnum teres dominates at the beginning. In addition to S. teres, Meesia triquetra and Bryum pseudotriquetrum (LLmII-4) were also found. The top part of the profile shows a characteristic succession system of Sphagnum teres–S. angustifolium–S. teres–S. angustifolium (LLmII-4, LLmII-5), which is accompanied by a significant increase in the presence of Oxycoccus palustris. Currently, Sphagnum teres and Oxycoccus palustris are common at the sampling site.
Subfossil Cladocera
26 species of Cladocera were identified, belonging to four families, Bosminidae, Daphniidae, Chydoridae and Sididae. Throughout the studied sedimentary sequence, the Cladoceran community is dominated by littoral taxa that constitute more than 70 % of the total Cladocera identified in the sediment. Eight Cladoceran phases are identified that reflect the lake development stages (Table 5). The changes in Cladoceran assemblages allow us to trace the ecological history of the lake from the Late-glacial period to ~a.d. 1000. The results of the subfossil Cladocera analysis are shown in an absolute frequency diagram (Fig. 7).
The initial phase of the lake in Zone LLcI-1 is correlated with the Late-glacial period. During this time, only three Cladocera species are present, Bosmina (Eubosmina) longispina, Acroperus harpae and Alona rectangula, and their frequencies are quite low. Over time (Zone LLcI-2), the ecological condition of the lake changed. Numerous open-water taxa appear here, Bomina longirostris, Eubosmina and Daphnia longispina group and they reach their highest frequency in the core (Fig. 7). Taxa living in association with plants are also abundant, represented primarily by Alonidae and Acroperus harpae. Additionally, sediment-associated taxa such as Leydigia sp. and Rynhotalona falcata were found only in the sediments of one zone, LLcI-2 (Fig. 7).
The Early Holocene sediment sequence is represented by the Cladocera Zone LLcI-3. During this time, littoral taxa are dominant and constitute 96 % (Figs. 7, 8), particularly taxa from the family Alonidae and Acroperus harpae, Chydorus sphaericus and Sida crystallina. Cladocera living in open water are represented by only one species, Bosmina longirostris, whose frequency is notably low, with a maximum 4 %.
The Middle Holocene period is represented by two Cladocera zones, LLcI-4 and LLcI-5. Littoral Cladocera are dominant, particularly taxa living in association with plants such as the Alonidae family and Chydorus sp., Eurycercus lamellatus and Graptoleberis testudinaria. During this period, open water taxa re-appear, including Bosmina longirostris in Zone LLcI-4 and the Daphnia longispina group in Zone LLcl-5. However, their frequency is significantly lower than in Zone LLcI-2 and does not exceed 8 %.
The uppermost sediments of the core were deposited during the Late Holocene period, and only littoral taxa are noted in the older sediments of this period (Zone LLcI-6, Fig. 7). The dominant species are Alona rectangula, Alonella excisa and Chydorus sphaericus.
At the beginning of Zone LLcI-7, the presence of planktonic taxa is noted again. Bosmina longirostris re-appears and reaches a maximum of 16 %. Taxa living in association with plants are also quite numerous and are dominated mostly by the Alonidae family, Sida crystallina, Graptoleberis testudinaria and Kurtzia latisima.
In the upper part of the studied sediment core, the remains of planktonic taxa completely disappear in Zone LLcI-8. Littoral taxa are dominant, including Alona rectangula, Alonella excisa and Alonella exigua. Additionally, the frequency of all Cladocera remains decrease and fall to the lowest level in the Holocene sediment sequence.
Discussion
Postglacial history of vegetation
Late-glacial period: ca. 12500–9600 b.c. (Phases LLpl 1 and 2, LLmI 1, LLcI 1)
The start of the development of lakes in the whole of northeastern Poland was connected with the melting of blocks of dead ice after climate warming (Ber 2006). The beginning of biogenic accumulation in the lakes began in the Bølling period, but it was at various times due to the different sizes of dead ice depressions. The bottom sediments in Linówek date back to ~12000 b.c. (Fig. 2). The age of the bottom lake sediments in Jezioro Kojle, ~5 km from Linówek, is ~11600 b.c. (Gałka and Sznel 2013) and ~10900 b.c. for Jezioro Hańcza (Lauterbach et al. 2011). The sediments in Jezioro Wigry started to accumulate ~13,700 cal. b.p. (Zawisza and Szeroczyńska 2007). After the dead ice had melted, plants typical of open and cold habitats appeared in the area. The pollen picture shows a clear dichotomy in the development of vegetation in the vicinity of Linówek (Fig. 8, Zone A). In the first phase dominant taxa include Betula (max. 57 %) as well as Hippophaë rhamnoides, Betula nana and Artemisia (Fig. 3). Moreover, there are significant records of Juniperus and Chenopodiaceae, and Equisetum grew in the wetlands.
The presence of Betula nana is recorded from ~12000 b.c., which proves that it was one of the first plants to appear at the site. B. nana owes its ability to grow even in the vicinity of the ice sheet snout to its rapid colonisation of newly uncovered sites (Binney et al. 2009). Taking into account the presence of Hippophaë rhamnoides in the bottom layer of the analysed sediment, it can be concluded that the minimum July temperature in northeast Poland was ~12 °C (Kolstrup 1979, 1980). The development of vegetation, in which one of the main elements was the light-demanding Hippophaë rhamnoides, may be linked to the Oldest Dryas, Bølling or Older Dryas. H. rhamnoides occurred during these periods in other lakes in Poland, at Witów (Wasylikowa 1964); Mikołajskie (Ralska-Jasiewiczowa 1966), Łukcze (Bałaga 1990), Miłkowskie (Wacnik 2009) and Kojle (Gałka, unpublished data). H. rhamnoides as a pioneer in heliophilous plant communities could grow at the edges of woodland on rather poor carboniferous soils (Kolstrup 1980, Wacnik 2009).
The first aquatic plant in the lake, which was characterised by a high water level, was Potamogeton filiformis. The amount of Pinus rises sharply (up to 61 %) in the second phase starting from ~11000 b.c., but the amounts of Betula, Juniperus, Betula nana and Artemisia decrease. A similar model of vegetation development was found in Miłkowskie (Wacnik 2009) and in other parts of Europe (Litt and Stebich 1999; Mortensen et al. 2011).
The rapid cooling of the climate at ~10700 b.c., which indicates the beginning of the Younger Dryas, is indicated by the decrease of Betula and Pinus pollen and an increase in Betula nana, Juniperus, Artemisia and Chenopodiaceae. The abundance of Betula nana in this area is reflected in the presence of its many fruits and fruit scales in the deposits (Fig. 8, Zone A). During this period, the amount of Helianthemum, Rosaceae and Ranunculaceae increases significantly. High levels of Juniperus (15 %) were found in Hańcza at ca. 10900 b.c. (Lauterbach et al. 2011). Caused by climate cooling, the rapid increase in Betula nana was also observed 100 years earlier at 10800 b.c. in Denmark (Mortensen et al. 2011) and in Estonia (Saarse et al. 2009). However, the period of rapid cooling at these sites cannot be directly correlated, because there are no radiocarbon dates from this part of the Linówek sediment. During the cold period of the Younger Dryas, Potamogeton filiformis and Batrachium sp. grew in the lake, which belong to the pioneer plants, and are cold period indicators (Bennike 2000; Birks 2000; Mortensen et al. 2011). Both taxa are often found in Late-glacial sediments in west-central Europe (Guiter et al. 2005; Stachowicz-Rybka et al. 2009; Fajer et al. 2012; Maj and Gałka 2012; Gałka and Sznel 2013).
Taking into account the presence of Myriophyllum spicatum pollen and the presence of Batrachium sp. fruit, it can be concluded that the minimum mean July temperature in this period oscillated around 10 °C (Kolstrup 1979, 1980; Brinkkemper et al. 1987). The cold period is also shown by the presence of macro- and microspores of Selaginella selaginoides, which is a typical species of tundra vegetation (Tobolski 2006; Stančikaitė et al. 2009; Gaidamavičius et al. 2011; Mortensen et al. 2011). The occurrence of S. selaginoides may even suggest lower temperatures of ~7 °C (Kolstrup 1979, 1980). A much lower water temperature can be deduced from the presence of Pediastrum kawrayskyi, whose share rose significantly at this time. P. kawrayskyi is a species typical of cold and clean waters and is often found in the sediments of the Younger Dryas (Janowská and Komárek 2000; Wacnik 2009).
Early Holocene period: 9600–6900 b.c. (LLpI 3 and 4, LLmI 2, LLcI 3)
Early Holocene climate warming occurred in the study area at ~9600 b.c. (Lauterbach et al. 2011), resulting in significant changes in the vegetation. From the initial phase of the Pre-boreal period around 8100 b.c., Betula is dominant, reaching 74 % (Fig. 8, Zone B). Such a large percentage of Betula is typical of the Pre-boreal period and this has also been reported from other parts of Europe, including Poland and Germany (Litt et al. 2001), the Netherlands (Bos et al. 2007), Lithuania (Stančikaitė et al. 2008) and Denmark (Mortensen et al. 2011). At ~8100 b.c., there is a significant increase in the proportion of deciduous trees with higher thermal requirements, including Ulmus and Corylus followed by Alnus at ca. 7400 b.c. and Quercus and Tilia at ca. 6800 b.c.
The beginning of the spread of Alnus in the area at ca. 7400 b.c. represents a significant increase of wetness in this part of Europe (Lauterbach et al. 2011), which caused a rise in the water level in the lakes of the area (Gałka and Sznel 2013; Gałka et al. unpublished data). Results from lakes show similar vegetation changes and a period of spreading of deciduous trees at Hańcza (Lauterbach et al. 2011), Wigry (Kupryjanowicz 2007) and Miłkowskie (Wacnik 2009).
A notable issue is the presence of Picea abies in this part of Europe in the Early Holocene. The occurrence of spruce at ~7400 b.c. is confirmed macroscopically from needles, bud scales, seeds in peat sediments and the overlying gyttja between Jezioro Kojle and Perty in the northern part of the Suwałki Landscape Park (Gałka and Tobolski 2013). The presence of P. abies at this time in the pollen results from Linówek is lower than 2 % (Fig. 3, LLp-I4). However, Picea pollen at ~0.6–1.0 % proves its presence in the area of the site (Środoń 1967; Giesecke and Bennett 2004).
With climatic warming and an increase in the water temperature in the early Holocene and the increase in lake level in Linówek, Chara spread and Potamogeton lucens, P. natans and Nymphaea alba appeared. Taking into account the presence of N. alba seeds, it can be concluded that the average July temperature was ~12 °C at ~9200 b.c. (Kolstrup 1979, 1980). At that time, Typha sp. was also present, confirmed by the presence of seeds and pollen of T. latifolia at ~8600 b.c., which suggests that the July temperature could have been higher, ~13 °C (Isarin and Bohncke 1999), and this temperature is also indicated by the presence of the fruits of Ceratophyllum sp. (Isarin and Bohncke 1999). The appearance of Potamogeton lucens in Linówek at ~9300 b.c., a species that grew in the lake for ~600 years, is not only associated with an increase in water temperature but also with an increase in calcium because it is characteristic of lakes rich in calcium carbonate (Zalewska-Gałosz 2008). The fossil presence of P. lucens has also been found in other parts of southwest Poland, where it was one of the first plants to colonise the ponds and lakes created during the Allerød (Maj and Gałka 2012). Climate warming allowed other thermophilous taxa to migrate to the area. The arrival of Corylus at ~8100 b.c. was accompanied by a further increase in temperature of ~2 °C. According to Hoffmann et al. (1998), the presence of Corylus shows that the average July temperature at that time was 15 °C. Cladium mariscus seeds were found at Kojle-Perty at ~7500 b.c. (Gałka and Tobolski 2012), which suggests that the mean July minimum temperature was not below 16 °C, and the mean minimum January temperature was ~−4 °C (Wasylikowa 1964).
The dynamic spread of Cladium mariscus in Poland in the Early Holocene is confirmed by its fossil presence in a lake on the southern Baltic coast, Łebsko, where the oldest evidence of its presence in the area have been radiocarbon dated to 9,850 b.p. (9767–8930 b.c.) (Tobolski 1987). For the Pre-boreal period, the presence of C. mariscus has been reported for southern Finland by Valovirta (1962).
Middle Holocene period: 6900–2300 b.c. (LLpI 5, LLmI 2-4, LLcI 4-6)
The dichotomy in the history of Middle Holocene vegetation in northeast Poland is well known (Fig. 8, Zone C). From 6900 to ~3650 b.c., the woods were dominated by Tilia with an almost constant 10 % and Corylus up to 30 %. Other taxa present include Ulmus up to 17 %, Quercus at ~5 % and Alnus at ~15 %.
From 6500 to 6100 b.c. we record the visible change in the proportion of trees. Corylus, Alnus, Quercus and Fraxinus decrease, and there is a rapid increase of Pinus and a slight one of Picea. This can be correlated with the cooling known as the “8.2 ka Event” which is widely recorded in the northern Atlantic and northern Europe (Tinner and Lotter 2001; Mayewski et al. 2004; Seppä and Poska 2004; Szeroczyńska and Zawisza 2011). The same pattern of plant succession between 8,350 and 8,100 cal. b.p. in Scandinavia has been observed (Sarmaja-Korjonen and Seppä 2007). The cooling at the time is also proved by the appearance in the lake of Alonopsis elongata, which is typical of lakes in northern areas (Hessen and Walseng 2008). It appeared in other Polish lakes during the “8.2 ka Event” (Szeroczyńska and Zawisza 2011). It is worth emphasizing that at this time Potamogeton pusillus disappeared from the lake, and it appeared there again in the warmer period afterwards. During the “8.2 ka Event” the average temperature of the warmest month could have been ~2 °C lower than the current temperature, which is shown by palaeoclimatic data (Heiri et al. 2004; Weninger et al. 2006; Magny et al. 2007).
Viscum pollen was found for the first time at ~5950 b.c., which proves that the temperature of the warmest month was by then at least 16 °C (Granoszewski 2003). At this time in addition to Potamogeton obtusifolius, P. pusillus and Ceratophyllum demersum, Najas flexilis, a plant typical of a climatic optimum (Godwin 1975; Lang 1994) was also found in the lake, which maintained a high water level at this time. The occurrence of N. flexilis at 5900 b.c. corresponds with a further increase in temperature and the beginning of the Holocene thermal maximum, which lasted from 6000 to 2500 b.c. in northeastern Europe (Heikkilä and Seppä 2003; Seppä and Poska 2004). The presence of Ceratophyllum demersum shows that Linówek was influenced by lime-rich water (Kłosowski et al. 2011). At ~4500 b.c., submerged plants almost completely disappeared from the lake, an event that occurred at the time of an increased share of Botryococcus and Pediastrum boryanum (Fig. 8). Such a large reduction in the presence of this group of aquatic plants is most probably related to the reduction in light intensity brought about by the excessive presence of algae (Dodds 2002).
At the transition to the second part of this period, which lasted from 3650 to 2300 b.c., a significant increase in Ulmus and Quercus took place, with a subsequent decline in Tilia and Corylus. The decrease in Tilia at ~3600 b.c. was also noted at Hańcza (Lauterbach et al. 2011) and Wigry (Kupryjanowicz and Jurochnik 2009). It can be assumed that this decrease was caused by a decrease in precipitation, because the water level in Linówek was low in that period. There was also a significant reduction in submerged plants in the lake. The taxa present include Chara sp. and Najas marina, which has an episodic occurrence. The presence of N. marina for a period of several thousand years was noted in the lakes at Kojle, Perty and Purwin (Gałka, unpublished data). During this time, the rush zone developed on the shores of Linówek, with Schoenoplectus lacustris, Carex paniculata and Lycopus europaeus. The presence of L. europaeus at ~3250 b.c. shows that the average July temperature at this time was no lower than 16 °C (Bell 1970). It is worth mentioning that in this period Ulmus is represented with high amounts, up to 20 % at ~2800 b.c. This is much higher than the values of up to 10 % reported in the specialist literature from neighbouring regions (Ralska-Jasiewiczowa et al. 2003; Zachowicz et al. 2004).
Late Holocene period: 2300 b.c.–a.d. 2010 (LLpI 6 and 7, LLpII 1–4, LLmI 5–7, LLmII 1-5, LLcI 6–8)
This period is characterised by increasing amounts of Picea and Carpinus (Fig. 8, Zone D). Picea abies spread widely at ~2300 b.c., as observed in the pollen results, and as finds of needles, seeds and bud scales show. Our date for the sudden increase of P. abies at ~2300 b.c. corresponds to the pattern of Picea distribution made by Latałowa and Van der Knaap (2006).
A clear cooling trend started at 4,500 cal. b.p. in Europe (Heikkilä and Seppä 2003; Seppä and Poska 2004) and in northeast Poland. The climate cooling and increase of precipitation at ~2300 b.c. favoured the expansion of Picea around Linówek. Increased precipitation encourages spruce growth (Koprowski 2013). Picea usually grows on damp soils (Giesecke and Bennett 2004) and it needs cold, snowy winters with average temperatures less than −6 °C for its reproduction (Dahl 1998). It is known for occurring in areas with a more continental climate. Changes to the structure of the woods in the vicinity of the lake, with a high contribution of Picea, caused an inflow to the lake of acidic substances from decomposing Picea needles, so that the lake sediments contained numerous spruce macrofossils. Changes in water quality resulting from acidification favoured the appearance of Nuphar pumila and Potamogeton alpinus. The presence of N. pumila indicates soft water (poor in Na+ and SO4 2−) but rich in total Fe and acidic substrates containing low amounts of Ca2+ (Kłosowski et al. 2011). An additional factor influencing the appearance those two aquatic plants is climate cooling. P. alpinus and Nuphar pumila are typical of the Boreal zone characteristic of the beginning or the end of interglacial periods. They indicate a cooling of the climate as well as changes in the moisture supply (Velichkevich and Zastawniak 2006, 2009). A decrease in temperature would have provided good conditions for their existence. The correlation between the occurrence of spruce in the vicinity of the lake and the appearance of those two plants in the lake was determined at Kojle-Perty, where P. alpinus and N. pumila appeared simultaneously at ~2500 b.c. (Gałka, unpublished data). A positive correlation between changes in aquatic vegetation and the appearance of Picea in the vicinity was also observed in other parts of Europe, as in eastern Lithuania at Bevardis (Gaidamavičius et al. 2011) and in northeast Poland at Puszcza Romincka (Gałka et al. 2012).
Changes in regional vegetation, as recorded in the Linówek lake sediments after 2300 b.c. are connected with regional changes of climate and soils representing the protocratic stage of an interglacial cycle (Birks 1986).
At about 950 b.c., the significant decrease from 14 to 4 % of Ulmus pollen corresponds to data from other sites in central Europe which can be connected with changes in soils from the meso- to oligocratic stages of an interglacial (Ralska-Jasiewiczowa et al. 2003).
An increase of Picea up to 12 % and of Carpinus up to ~4 % occur about 800 b.c., at the same time as a significant decline of Ulmus, Tilia and Corylus. Among the declining deciduous trees, it should be noted that the smallest decline is of Quercus. The same trend for reductions of the deciduous taxa Ulmus, Corylus and Tilia and no significant decline of Quercus has been found in southern Finland (Heikkilä and Seppä 2003). This correlates with the reduction of Chara sp. and the loss of Potamogeton pusillus and Schoenoplectus lacustris from Linówek, which can be associated with increased precipitation and an increase in the lake level.
The reason for the expansion of Picea and Carpinus in the woods of northeast Poland is climate cooling and deterioration of the quality of soils, which favoured both species, which grow better in more continental conditions. Considering the succession of vegetation in the Eemian interglacial, the two species appear in the protocratic stage (Granoszewski et al. 2012; Kołaczek et al. 2012). An increase in the contribution of Carpinus in woods in northern Poland has also been recorded at other sites (Ralska-Jasiewiczowa et al. 2003).
The cooling of climate and increase in precipitation between 800 and 600 b.c. was a general trend in the whole of Europe. It was recorded in the Netherlands (Van Geel et al. 1996), Switzerland (Holzhauser et al. 2005), Northern Ireland (Swindles et al. 2007), Finland (Tuittila et al. 2007), Germany (Voigt et al. 2008) and Sweden (Andersson and Schoning 2010). It corresponds to a widespread climate reversal identified at the Sub-boreal to Sub-atlantic transition (Van Geel et al. 1996). During this time the glaciers in the Alps were growing (Holzhauser et al. 2005) and accumulation on the peat bogs increased (Gałka et al. 2013). Climatic oscillations at this time can be associated with low solar activity (Holzhauser et al. 2005; Mauquoy et al. 2008).
Around a.d. 100 when an increase in temperature was noted in Europe (Seppä and Poska 2004; Moschen et al. 2011), Cladium mariscus which responds to more oceanic climate and higher temperature conditions appeared in the Linówek sediments. The modern distribution of C. mariscus is mainly on the Baltic coast of Latvia (Salmina 2004). At about a.d. 750 Ulmus disappears almost completely from the study area, while the amounts of Corylus and Tilia decline significantly, most probably due to increased human pressure here. Around a.d. 1000–1100 the lake disappeared. The final phase (LLmI-7) is characterised by Nymphaea alba and Nuphar lutea, which are still present in the area today. Cladium mariscus grew in the rush zone, but it is not found any more in the Linówek area. It disappeared in modern times, as its seeds were found in the top layer of the sediments (Gałka and Tobolski 2012). Due to the increasing shallowness of the lake, the area turned into a fen with the sedges Carex paniculata and C. nigra in addition to Menyanthes trifoliata.
At ~a.d. 1470, the amounts of Carpinus betulus and Picea abies increased in the vicinity of the lake, as seen in an increase in the pollen curve. Nowadays P. abies grows in the vicinity of the lake and its needles were found in the studied peat sediments. The increase in both these trees corresponds to the appearance of Scheuchzeria palustris on the peat bog (Fig. 9, Zone E). The correlation between the changes in vegetation in the catchment of the peatland and those on the peatland was observed on Baltic raised bogs in northern Poland as well, at Stążki (Gałka et al. 2013), Mechacz Wielki (Gałka, unpublished data) and Gązwa (Gałka, unpublished data).
At about a.d. 1650, the amount of Picea abies again increases significantly to a maximum of 35 %, which corresponds to the beginning of the development of the transitional bog. Larger numbers of Oxyccocus palustris and Andromeda polifolia macrofossils were found as well as Sphagnum teres and Meesia triquetra, followed by Sphagnum angustifolium and S. sect. Acutifolia (Fig. 9, Zone F). The appearance of vegetation representing a more acid environment can be associated with the influence of Picea abies, which grew on the bog, where its presence caused acidification of soils. The increased share of Picea abies in these two periods can be associated with climatic cooling during the “Spörer Minimum” (a.d. 1420–1550) and the “Maunder Minimum” (a.d. 1645–1715).
In summarising the discussion of the history of the woodlands in the surroundings of Linówek, special attention should be paid to the dynamics of spruce and its proportion in various woodland communities. The presented material suggests the need for in-depth studies of the role of spruce stands in their plant community, in the border zone of the temperate and boreo-nemoral climate zone, particularly in the context of human impact factors.
Human activity
Based on the presence of indicators of human activity at the site of Linówek, three main periods have been defined and are explained in sequence.
1. 12000–2100 b.c.
According to the archaeological data and based on the finds of single flint tools, humans appeared in northeastern Poland during the Bölling period, and this was related to the hunting of reindeer (Brzozowski and Siemaszko 2005). The first traces of human activity in the study area can be associated with the presence of a Mesolithic hunter-gatherer group from burning of the woodland. The spores of Pteridium aquilinum are found in the sediment from ~6650 b.c. (Fig. 3), which may indicate fires that may have been caused by the Mesolithic people. During this period, the Kunda Culture developed in northeastern Poland (Brzozowski and Siemaszko 2005). A well-equipped camp of this culture dating back to 8300 b.c. was found near Ełk, ~60 km west of Linówek, and numerous sites of this culture have been discovered in the Pojezierze Suwalskie (Suwałki Lake District) and also in the Baltic states (Brzozowski and Siemaszko 2005). Between 5650 and 4150 b.c., the Pteridium aquilinum curve shows growth that can be interpreted as an increasing intensity in burning of woodland (Göransson 1986; Madeja et al. 2004), but on the other hand it could also have been influenced by increased grazing pressure from wild herbivores. The first traces of human activity are recorded around the same time in the lake pollen results from Wigry, as shown by the growing curve of Pteridium aquilinum (Kupryjanowicz 2007). Rumex acetosa/acetosella pollen, whose presence is considered an indicator of a pastoral economy (Behre 1981), was found about 4150, 3300 and 2400 b.c. (Fig. 3).
2. 2100 b.c.–a.d. 1400
The first traces of Cerealia pollen were found about 2050 b.c. (Fig. 3). At the same time, Cerealia pollen and Plantago lanceolata pollen which was ~400 years older (2461 b.c.), was found at Wigry (Kupryjanowicz and Jurochnik 2009).
The presence of Cerealia and Plantago lanceolata pollen shows that the first open spaces used as fields or pastures were created in the vicinity of Linówek. This corresponds with a decline in Ulmus and an increase in Picea abies and Carpinus betulus. A similar correlation was found in Wigry (Kupryjanowicz and Jurochnik 2009). In the Baltic states, the increase in human activity demonstrated by the presence of cereal pollen was found several centuries earlier (Stančikaitė et al. 2002, 2006; Zernitskaya and Mikhailov 2009; Ozola et al. 2010). Cerealia pollen was reported in the Pojezierze Mazurskie (Great Masurian Lake District) ~100 years earlier (Wacnik et al. 2012).
Another distinct phase of human settlement development started at ~950 b.c., which is confirmed by the presence of Cerealia and Plantago lanceolata pollen as well as Rumex acetosa/acetosella and an increase in Artemisia. The increase in human activity was regional, and was also noted in the area of Jezioro Wigry (Kupryjanowicz and Jurochnik 2009). There is a decline of Ulmus and Corylus as well as an increase of Carpinus, Picea and Betula in this period.
Around 500 b.c. an increase of human activity in northeastern Poland is associated with the arrival of the Baltic tribes in the area. They were the first people with strong agricultural traits who settled in the northeast of Poland (Brzozowski and Siemaszko 2005). The growing importance of agriculture is shown by the curves of Cerealia and Plantago lanceolata. The first Triticum type pollen found ca. 250 b.c. corresponds to a further decline in Ulmus and Acer (Fig. 3, Zone LLpI-7), and this can be linked to occupation of the Suwałki region by the people of the Bogaczewo Culture, which was based on farming and animal husbandry (Brzozowski and Siemaszko 2005). Since then, the pollen of these taxa is present only sporadically. A decline in Ulmus and Acer as well as Corylus and Tilia indicates woodland clearance and use of these areas as fields.
According to the current state of knowledge, the oldest known evidence of Triticum cultivation in the area was found at the archaeological site of Osinki, 15 km southeast of our site. Several grains of Triticum aestivum and T. diococcum were found in sediments dated to the Bogaczewo Culture (Karczewski 2011). At another site, Góra Zamkowa in Szurpiły, 3 km east of Linówek, grains of Triticum sp., Hordeum vulgare and Pisum sp. were found in remains of hearths (Karczewski 2011). They were initially dated to the time of the early Roman period, but this age cannot be unequivocally adopted (Okulicz-Kozaryn 1993). It cannot be excluded that earlier remains of cereal cultivation in the area will be discovered in the future.
During late Roman and Migration period, the local people cultivated wheat and rye, as shown by pollen data from our lake site Linówek and at Wigry (Kupryjanowicz 2007).
The first pollen of Secale at Linówek was found in a level dated around a.d. 550, which corresponds to the settling of tribal groups around the villages of Szurpiły and Jeleniewo, within 5 km of Linówek (Szkiruć 1986). According to the archaeological sites in the village of Szwajcaria, ca. 20 km from Linówek, there was a tribal centre there until the 5th century and then, for unknown reasons, it slowly ceased to exist (Szkiruć 1986; Kowalski 2000).
Earlier, from ~a.d. 400, a pollen grain of Secale was recorded at the site of Mechacz Wielki, ~25 km north of Linówek (Gałka, unpublished data). The oldest grains of Secale cereale from there are dated to the 3rd–4th century a.d. and were found at the archaeological site of Osowa (Karczewski 2011). As well as Secale cereale, grains of Triticum aestivum, T. diococcum, T. spelta, T. compactum, Hordeum vulgare and Pisum sativum were also found. At another archaeological site, Osinki, dated to the 5th-6th century, Secale cereale was found among numerous grains of Triticum aestivum, T. spelta, T. compactum, T. diococcum and Hordeum vulgare (Czeczuga and Kossacka 1973).
In the 9th century settlement activity in the tribal centre of Szurpiły, ~3 km from Linówek began, where a stronghold or fortified settlement was established by the Jatvingians, the most populous and richest group among the Prussian tribes. The location of the settlement on a hill surrounded by four lakes, Szurpiły, Jeglówek, Kluczysko, Jeglóweczek and wetlands, shows the settlement strategy of the Balts, with the use of natural terrain for building defensive forts (Wiliński 1984; Łowmiański 1986). The heyday of this settlement complex was reached in the 12th–13th century, when the expansion of the gord (a medieval Slavic settlement) and defensive fortifications (Engel and Sobczak 2012) as well as an increase in the area of farmland led to the loss of woodland shown by the decrease of Ulmus, Tilia and Corylus (Fig. 4). The significance of the economic centre and the well-developed farming are proven by finds of cereal grains (Triticum sp., Sereale cereale) and the bones of cattle, pigs, goats and sheep on archaeological sites (Szkiruć 1986; Wróblewski and Sawicka 2012).
In the second half of the 13th century the Jatvingian settlements ended in the area, a change which is clearly visible in the pollen diagram (Fig. 9). It was due to the war campaigns of the Teutonic Knights, which led to depopulation by gradual raids into these areas. The gord eventually collapsed, and the local population was either displaced or fled beyond the rivers Niemen or Biebrza (Szkiruć 1986). The accepted date of the fall of Szurpiły is a.d. 1283 (Engel and Sobczak 2012).
As demonstrated by the disappearance of cereals and Rumex acetosa/acetosella (Fig. 9, Zone E), economic activity ceased at that time. The depopulation of these territories in the 1280s was described by Peter of Dusburg, the Teutonic chronicler (Engel and Sobczak 2012). After that event, the woods in the area regenerated, as shown by increased values of Picea, Carpinus and Corylus (Fig. 9, Zone F) as well as the slight increase of the Quercus curve. A similar development can be observed at Wigry (Kupryjanowicz et al. 2009). All economic activity in the area was halted before the beginning of the 15th century, and for decades, the woods remained as a hunting area for Polish kings and Lithuanian princes.
3. a.d. 1400–2010
At about a.d. 1400 the area was repopulated, shown by pollen of Secale and Rumex acetosa/acetosella, with decreases in the tree curves of Corylus, Tilia and Carpinus. Around a.d. 1450 the first Centaurea cyanus pollen is recorded, corresponding to the occurrence of Cerealia pollen (Fig. 9, Zone F). C. cyanus pollen is argued to strongly reflect the presence of permanent field systems (Vuorela 1986). Despite the presence of farm fields in the area, the values of Picea, Carpinus and Tilia rise again as well as gradual rises in the curves of Alnus and Picea. The increased human activity was not only connected to farming but also to forestry. Settlements were located in the wooded area, and the inhabitants worked at felling timber, producing tar and beekeeping (Szkiruć 1986). From the 15th century onwards the woodlands in eastern Poland were an important source of timber for ship building, paintings and sculptures (Ważny 2005; Haneca et al. 2005).
A significant increase in human indicators takes place around the 1620. An increase in Secale and Rumex acetosa/acetosella is also recorded, accompanied by a decrease in Carpinus and Picea (Fig. 9, Zone F). This is connected with the founding of the village of Blaskowizna and the construction of two mills ~1 km from Linówek (Szkiruć 1986). In 1668, a Camaldolese (Benedictine) monastery was located near Jezioro Wigry, and at the end of the 17th century many new villages were established in the vicinity of modern town of Suwałki (Matusiewicz et al. 2005).
Since 1720, there has been a significant increase in human indicators, which show the spread of cereal cultivation, the most intensive in the history of this region. The increase in human activity caused an increase in pollen of Secale, Triticum-type, R. acetosa/acetosella and Plantago lanceolata (Fig. 9, Zone G). 1720 is a landmark date for the decline of deciduous woods in the area which were almost completely cut down, as a result of the arrival of settlers from Russia in the area (Szkiruć 1986). Since then, the amounts of Tilia, Carpinus and Ulmus dropped sharply and that of Quercus even more significantly (Fig. 9, Zone G). Fagopyrum (buckwheat) pollen was found for the first time in the sediments of Linówek dated to about 1720. Even single Fagopyrum pollen grains prove that it was grown in the direct vincity of the lake, because it produces only small amounts of pollen which is not very well dispersed (Behre 1981). Fagopyrum pollen was found dating from ~150 years earlier in Wigry (Kupryjanowicz and Jurochnik 2009) and in the lakes of the eastern part of the Pojezierze Mazurskie (Great Masurian Lake District), as well as in the lake sediments from Staświńskie (a.d. 820) and in the Miłkowskie and Wojnowo lakes (about a.d. 1200, Wacnik et al. 2012). The decline in economic indicators in the vicinity of Linówek around 1830 is related to war campaigns and the partial depopulation of the villages. The next decrease of human indicators around the year 1915 is associated with the First World War, which led to regeneration of woodlands and an increasing amount of Picea.
Lake level changes: 11600 b.c.–a.d. 1000
Using the sedimentary sequence, three periods of high water level were determined (12000–9400, 7000–4000 and 1450 b.c.–a.d. 650), based on the results of subfossil Cladocera and analysis of plant macroremains. Between these, two periods (9400–7100 and 3700–1600 b.c.) with low water levels were identified (Fig. 8).
High level
1. 12000–9400 b.c.
The first phase of high water level at Linówek was noted during the Late-glacial and early Holocene and is shown by a high proportion (75 %) of planktonic Cladocera taxa of the genus Bosminidae and the Daphnia longispina group (Fig. 7). An increasing water level during the Late-glacial was also recorded in nearby Wigry, where planktonic taxa from the Eubosmina family were present (Zawisza and Szeroczyńska 2007). The same was also noted in other lakes in the Polish lowlands (Milecka and Szeroczyńska 2005; Milecka et al. 2011; Szeroczyńska and Zawisza 2007). The increase in the water level of Linówek also corresponds to the data from Belarus, where the cutoff between the Allerød and Younger Dryas was marked by a significant increase in the water levels of the lakes there (Novik et al. 2010). This rise in water level was most probably the result of the final melting of dead ice blocks.
2. 7100–4000 b.c.
The second period with a higher water level for Linówek occurred during the middle Holocene period. The beginning of this period was significantly wetter, indicated by the presence of Alnus (Fig. 8). The increase in water level has also been observed in the Cladocera assemblages since 7100 b.c. Planktonic Bosmina longirostris appeared at the beginning of this period, followed by Daphnia longispina group, typical open-water taxa (Fryer 1985; Flössner 2000). The presence of planktonic Cladocera, especially Bosmina longirostris, implies an increase in water level as well as the amount of nutrients in the lake. The trophic increase is also shown by the increase in aquatic plants characteristic of eutrophic water such as Potamogeton obtusifolius, P. pusillus and Najas flexilis (5800–4550 b.c.) (Ellenberg et al. 1991; Zarzycki et al. 2002). As observed by Backman (1948) and Wingfield et al. (2004), the optimal water depth for Najas flexilis is ~2 m. However, this species is able to grow in a water depth of up to 12–14 m, as at Lake Shoal, Manitoba, Ontario (Pip and Simmons 1986).
There is a high probability that the period around 7100 b.c. was characterised by higher rainfall, which was caused by the increased influence of warm and moist air brought by the westerly winds from the Atlantic Ocean after the final decay of the ice sheet between 10,000 and 9,500 cal. b.p. (Yu and Harrison 1995; Lauterbach et al. 2011). Higher precipitation might have been responsible for the increasing water level and the nutrient supply to the lake from the catchment.
In Kojle-Perty, a sudden increase in water level was recorded at ~7400 b.c. and was shown by the accumulation of gyttja on top of peat and a change in the type of vegetation; aquatic taxa also appeared (Gałka et al., unpublished data). The same tendency to an increasing water level since 7100 b.c. was noted by Zawisza and Szeroczyńska (2007) and Kupryjanowicz et al. (2009) in Wigry.
Our dates correlate well with the palaeoecological results from other sites in Europe. Magny (2004) distinguished three periods of high water levels in central European lakes at 6300–4400 b.c. (6350–6000, 5600–5300 and 4400–4950 b.c.) which correlate well with our data. Based on the results of Cladocera analysis, high water levels were determined in southern Finland in Iso Lehmälampi Jaarvi during 7000–4000 b.c. (Sarmaja-Korjonen 2001).
3. 1450 b.c.–a.d. 650
The last period with higher water levels in Linówek is correlated with the Sub-boreal period. This period of increasing water level is mostly distinguished by the re-appearance of planktonic Cladocera represented by Bosmina longirostris, the presence of which indicates the existence of the lake pelagic zone; however, this species can also periodically occur in the littoral zone (Burks et al. 2002). The absence of other planktonic taxa and the increasing frequency of taxa associated with plants (Alonidae, Graptoleberis testudinaria) indicate that a significant part of the lake was overgrown by aquatic vegetation. As suggested by our data, the last lake level rise recorded in Linówek was significantly lower than the previous ones.
The period around ~750 b.c. was crucial for the development of aquatic vegetation. Since then, the share of Chara sp. and Nuphar lutea decrease. The hypothesis of an increase in water level is also confirmed by the disappearance of Schoenoplectus lacustris and Typha sp., which are typically found in shallow water (Hannon and Gaillard 1997).
The results obtained from Linówek correspond well to the rise in water level in other lakes in central Europe. Magny (2004) distinguishes between 1550 b.c. and a.d. 250 three periods with higher water level (1550–1150, 800–400 b.c., a.d. 150–250).
Around the year 1550 b.c. the climate cooled and precipitation increased, as shown by high water levels recorded by Heikkilä and Seppä (2003) in the Finnish lakes and by Terasmaa (2011) and Punning et al. (2003) in Estonian lakes. Increasing water levels around 1500 b.c. were also noted in peat bogs located on the Polish Baltic coast (Stążki peat bog, Gałka et al. 2013) and in south Finland (Väliranta et al. 2007). Since 1500 b.c., increased water levels have been reported in the mires of the north of Ireland (Swindles et al. 2010). Starkel (2011) reconstructed the high water level in general in Polish lakes between 400 b.c. and a.d. 50, using several palaeoecological methods.
Low level
1. 9400–7100 b.c.
The beginning of the Holocene was marked by significant climate warming. Climatic changes, particularly an increase in the mean annual temperature, had an important influence on the Linówek lake ecosystem as shown by the decreasing water level there. An early drop in the water level in the Holocene is well known from Cladocera analysis and plant macrofossil results. In Cladocera species composition the frequency of pelagic taxa decreases and plant-associated taxa predominate (Fig. 8).
The common occurrence of Chara spp., many of which grow in shallow water, was the first sign of a lower lake level. This is also shown by the presence of Potamogeton crispus and P. filiformis, which are also shallow water species (Zalewska-Gałosz 2008). According to Siitonen et al. (2011), P. filiformis grows in shallow depths (0.1–0.5 m), the maximum being around 3 m. P. lucens also has similar ecological needs (max. depth 3 m), so its presence indicates quite a low water level (Podbielkowski and Tomaszewicz 1996). The reduction of the water level in Linówek is well correlated with the data from other lakes in Suwalki Landscape Park. At that time, reduction in water level was noted in Kojle and Perty (Gałka, unpublished data). In the area of Hańcza, small lakes disappeared and peat bogs developed in their place (Gałka and Sznel 2013). The same process occurred with a small water body (now a peat bog) located ~300 m from Linówek (Zdunek and Gałka, unpublished data). At ~8250 b.c., a decrease in water level was also noted in Wigry, the largest lake in the area (Zawisza and Szeroczyńska 2007).
Early Holocene changes in water level were also recorded in other parts of Poland, in the lakes Łukcze (Bałaga 1990), Mikołajki (Ralska-Jasiewiczowa and Latałowa 1996), Gościąż (Starkel et al. 1998) and Sierzywk (Milecka et al. 2011). A decrease in water level at this time has been noted in other parts of Europe, including Scandinavia (Digerfeldt 1971, 1986; Gaillard 1985), central Europe (Magny 2004), the Netherlands (Bos et al. 2007), Estonia (Hang et al. 2008; Terasmaa 2011) and Belorussia (Novik et al. 2010), showing the regional nature of these changes.
2. 3700–1700 b.c.
The second period with a notably lower water level is correlated with the end of the Atlantic period and the beginning of the Sub-boreal period. During this time, only littoral Cladocera taxa were present at the coring site. The Cladocera community was dominated by species associated with plants, namely, by Alona rectangula, Alonella excisa and Chydorus sphaericus. The community of Cladocera taxa indicate a reduction in water level and suggests the beginning of the overgrowing process in the lake. The dominant Cladocera taxa are known as littoral and also as resistant to unfavourable environmental conditions and low pH values. It is significant that in this phase, Botryococcus is most abundant (Fig. 8). The lower water level is also indicated by the considerable decrease in aquatic plants (Fig. 8). However, it should be mentioned that the decline of aquatic plants could be also combined with an abundance of Botryococcus. Blooming Botryococcus may have reduced the amount of light, which may have led to the disappearance of submerged plants. The occurrence of Schoenoplecus lacustris (since 2722 b.c.) and Typha sp. (since 2350 b.c.) indicate shallow water and the development of a littoral zone (Hannon and Gaillard 1997).
The decreasing water levels noted in Linówek correspond well with data from northwest Poland (Ralska-Jasiewiczowa and Latałowa 1996) and northern Estonia (Punning et al. 2003), where periods of low water level were noted at 2850–2200 and 2000–1650 b.c. The lowering of water level in the lakes appears to have occurred on a regional scale, and was noted in the lakes located in the Swiss Plateau (Haas et al. 1998), in southern Sweden (Digerfeldt 1998) and on the central European lowlands (Magny 2004). Lower lake water levels are associated with a dry climate phase, which correlates with a more continental climate, as is well known from the low rate of accumulation of peat bogs in northeastern Europe (Lamentowicz et al. 2008; Starkel 2011). The dry climate phase since 3700 b.c. in northeast Poland can also be related to climate cooling in the Northern Hemisphere between 4000 and 3000 b.c., which was studied by Mayewski et al. (2004).
Conclusions
Based on the results of the palaeoecological analyses with high-resolution plant macroremains and pollen, Cladocera fossils and radiocarbon dates, the following conclusions can be stated:
-
1.
The formation of a lake at Jezioro Linówek is connected with melting of dead ice blocks ~14,000 years ago.
-
2.
Climate, soil change and human activity have influenced the character of the vegetation at both the regional level regarding the woods around the lake, and local levels on the lacustrine and peat bog vegetation.
-
Climate warming at ~6800 b.c. and improved soil quality allowed the expansion of Alnus, Tilia and Quercus, and Potamogeton obtusifolius, P. pusillus and Nuphar lutea in the lake.
-
Climate cooling at ~6200 b.c. caused a visible change in proportions of trees, with a decrease in Corylus, Alnus, Quercus and Fraxinus, and an increase in Pinus and Picea, as well as in the vegetation in the lake, where the Cladocera Alonopsis elongata appeared and Potamogeton pusillus disappeared. This can be correlated with cooling during the “8.2 ka Event” which is widely recorded in the Northern Hemisphere.
-
Climate cooling, deterioration of the quality of soils and water acidification at ~4300 b.c. favoured the migration and distribution of taxa in areas with a more continental climate, and a reduction in deciduous taxa such as Ulmus, Corylus, and Tilia. At the same time there was a sharply increased share of Picea abies and Carpinus betulus in the woods and Potamogeton alpinus and Nuphar pumila appeared in the lake.
-
At about a.d. 1650, the share of Picea abies again increased significantly to a maximum of 35 %, which corresponds to the beginning of the development of the transitional bog. The presence of Picea abies on the bog and acidification caused the development of vegetation typical of a more acid environment, with Oxyccocus palustris, Andromeda polifolia, and Sphagnum spp.
-
-
3.
Based on the pollen data, three settlement phases were defined in the study area, 6800–2100, 2100 b.c.–a.d. 1400 and a.d. 1400–2010. The first traces of agricultural activity noted from Cerealia pollen were dated to ~2100 b.c. The first Triticum pollen appeared at ~250 b.c., that of Secale began at ~a.d. 550 and that of Fagopyrum began around a.d. 1720. The most intense human activity and major changes in vegetation, consisting of cutting down or burning the trees, took place at the time of the Jatvingian settlement in the area (ca. a.d. 800–1250), and also after the establishment of several villages around the year 1700. In between there was a decline in human activity, expressed as a decrease in the records of economic indicators and an increase in trees, which was recorded after a.d. 1250. This was due to the displacement of the local Jatvingian population by the Teutonic Knights, and the resulting regeneration of woodlands.
-
4.
Based on the studies of subossil Cladocera and analysis of the presence of local lake vegetation in Linówek, three periods of high water level were delimited, 12000–9400, 7000–4000 and 1450 b.c.–a.d. 650, and two periods of low water level, 9400–7100 and 3700–1700 b.c. Pronounced fluctuations in the water level in Linówek are correlated with data from other research sites in Europe.
-
5.
The palaeoecological analyses of sediments collected from small lakes provides a good potential for reconstructing the impact of climate changes on the lacustrine vegetation in the catchment.
References
Andersson S, Schoning K (2010) Surface wetness and mire development during the late Holocene in central Sweden. Boreas 39:749–776
Backman AL (1948) Najas flexilis in Europa während der Quartärzeit. Acta Bot Fenn 43:1–44
Bałaga K (1990) The development of Lake Łukcze and changes in the plant cover of the south-western part of the Łęczna-Włodawa Lake district in the last 13,000 years. Acta Palaeobot 30:77–146
Behre K-E (1981) The interpretation of anthropogenic indicators in pollen diagrams. Pollen Spores 23:225–245
Bell FG (1970) Late Pleistocene floras from Earth, Huntingdonshire. Phil Trans R Soc B 258:347–378
Bennike O (2000) Palaeoecological studies of Holocene lake sediments from west Greenland. Palaeogeogr Palaeoclimatol Palaeoecol 155:285–304
Ber A (1974) The quaternary of the Suwałki Lake District. Biul Inst Geol PAN 269:23–106
Ber A (2000) Pleistocene of north-eastern Poland and neighbouring areas against crystalline and sedimentary basement. Pr Państ Inst Geol 170
Ber A (2006) Pleistocene interglacials and glaciations of northeastern Poland compared to neighbouring areas. Quat Int 149:12–23
Berggren G (1968) Atlas of seeds and small fruits of Northwest-European plant species (Sweden, Norway, Denmark, East Fennoscandia and Iceland) with morphological descriptions. Part 2. Cyperaceae. The Swedish Museum of Natural History, Stockholm
Berggren G (1981) Atlas of seeds an small fruits of Northwest-European plant species (Sweden, Norway, Denmark, East Fennoscandia and Iceland) with morphological descriptions. Part 3. Salicaceae—Cruciferae. The Swedish Museum of Natural History, Stockholm
Beug HJ (2004) Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete. Pfeil, München
Binney HA, Willis KJ, Edwards ME, Bhagwat SA, Anderson PM, Andreev AA, Blaau M, Damblon F, Haesaerts P, Kienast F, Kremenetski KV, Krivonogov SK, Lozhkin AN, MacDonald GM, Novenko EY, Oksanenm P, Sapelko TV, Väliranta M, Vazhenina L (2009) The distribution of late-Quaternary woody taxa in northern Eurasia: evidence from a new macrofossil database. Quat Sci Rev 28:2,445–2,464
Birks HJB (1986) Late-Quaternary biotic changes in terrestrial and lacustrine environments, with particular reference to north-west Europe. In: Berglund BE (ed) Handbook of Holocene palaeoecology and palaeohydrology. Wiley, Chichester, pp 3–65
Birks HH (2000) Aquatic macrophyte vegetation development in Kråkenes Lake, western Norway, during the lateglacial and early-Holocene. J Paleolimnol 23:7–19
Bortyński A (2007) The Central European disjunctions in the range of Norway spruce. In: Tjoelker MG, Boratyński A, Bugała W (eds) Biology and ecology of Norway spruce (Forestry Sciences 78). Springer, Dordrecht
Bos JAA, Van Geel B, Van der Plicht J, Bohncke SJP (2007) Preboreal climate oscillations in Europe: wiggle-match dating and synthesis of Dutch high-resolution multi-proxy records. Quat Sci Rev 26:1,927–1,950
Brinkkemper O, Van Geel B, Wiegers J (1987) Palaeoecological study of a Middle-Pleniglacial deposit from Tilligte, The Netherlands. Rev Palaeobot Palynol 51:235–269
Brzozowski J, Siemaszko J (2005) Najstarsze dzieje okolic Suwałk (The earliest history of the Suwalki area, in Polish). In: Kopciała J (ed) Suwałki miasto nad Czarną Hańczą. Wydawnictwo Hańcza, Suwałki, pp 67–87
Burks RL, Lodge DM, Jeppesen E, Lauridsen TL (2002) Diel horizontal migration of zooplankton: costs and benefits of inhabiting the littoral. Freshw Biol 47:343–365
Czeczuga B, Kossacka W (1973) Nasiona roślin uprawowych i chwastów z osady jaćwiskiej z Osowej, powiat Suwałki (III–IV w. n.e.) Acta Baltico-Slawica 8:151–164
Dahl E (1998) The phytogeography of northern Europe. Cambridge University Press, Cambridge
Daniels E, Eddy A (1990) Handbook of European Sphagna. Institute of Terrestrial Ecology, Natural Environment Research Council, London
Digerfeldt G (1971) The post-glacial development of the ancient lake Torreberga, Scania, South Sweden. GFF 93:601–624
Digerfeldt G (1986) Studies on past lake-level fluctuations. In: Berglund BE (ed) Handbook of Holocene palaeoecology and palaeohydrology. Wiley, Chichester, pp 127–144
Digerfeldt G (1998) Reconstruction of Holocene lake-level changes in southern Sweden: technique and results. Paleoclim Res 25:87–98
Dodds WK (2002) Fresh water ecology, concept and environmental applications. Academic Press, New York
Ellenberg H, Weber HE, Düll R, Wirth V, Werner W, Paulißen D (1991) Zeigerwerte von Pflanzen in Mitteleuropa. Scr Geobot 18:1–248
Engel M, Sobczak C (2012) Gród jećwieskiego wodza Szjurpy (Sjurpy) i jego system obronny. In: Rant-Tanajewska M, Świerubska T, Dziubiak M, Buczyński P (eds) XXXV lat Suwalskiego Parku Krajobrazowego. Suwalski Park Krajobrazowy, Jeleniewo, pp 27–36
Fægri K, Iversen J (1989) In: Fægri K, Kaland PE, Krzywinski K (eds) Textbook of pollen analysis, 4th edn. Wiley, Chichester
Fajer M, Waga JM, Rzetala M, Szymczyk A, Nita M, Machowski R, Rzetala MA, Ruman M (2012) The Late Vistulian and Holocene evolution of Jezioro Lake: a record of environmental change in southern Poland found in deposits and landforms. J Paleolimnol 48:651–667
Flössner D (2000) Die Haplopoda und Cladocera (ohne Bosminidae) Mitteleuropas. Backhuys, Leiden
Frey DG (1986) Cladocera analysis. In: Berglund BE (ed) Handbook of Holocene palaeoecology and palaeohydrology. Wiley, New York, pp 667–692
Fryer G (1985) The ecology and distribution of the genus Daphnia (Crustacea: cladocera) in restricted areas: the pattern in Yorkshire. J Nat Hist 19:97–128
Gaidamavičius A, Stančikaite M, Kisieliene D, Mažeika J, Gryguc G (2011) Post-glacial vegetation and environment of the Labanoras Region, East Lithuania: implications for regional history. Geol Quart 55:269–284
Gaillard MJ (1985) Postglacial palaeoclimatic changes in Scandinavia and central Europe—a tentative correlation based on studies of lake level fluctuations. Ecol Medit 9:159–175
Gałka M (2009) A Juncus subnodulosus SCHRANK fossil site in Holocene biogenic sediments of Lake Kojle. Stud Lim et Tel 3:55–59
Gałka M (2010) Sphagnum wulfianum Girgens in the Suwałki Landscape Park (NE Poland). Stud Lim et Tel 4:51–56
Gałka M, Sznel M (2013) Late glacial and Early Holocene development of lakes in northeastern Poland in view of plant macrofossil analyses. Quat Int 292:124–135
Gałka M, Tobolski K (2012) Palaeoecological studies on the decline of Cladium mariscus L. (Pohl.) in NE Poland. Annal Bot Fenn 49:305–318
Gałka M, Tobolski K (2013) Macrofossil evidence of the early Holocene presence of Picea abies (Norway spruce) in NE Poland. Ann Bot Fenn 50:129–141
Gałka M, Tobolski K, Kołaczek P (2012) The Holocene decline of slender naiad (Najas flexilis (Willd.) Rostk. & W.L.E. Schmidt) in NE Poland in the light of new palaeobotanical data. Acta Palaeobot 52:127–138
Gałka M, Miotk-Szpiganowicz G, Goslar T, Jęśko M, Van der Knaap WO, Lamentowicz M (2013) Palaeohydrology, fires and vegetation succession in the southern Baltic during the last 7500 years reconstructed from a raised bog based on multi-proxy data. Palaeogeogr Palaeoclimatol Palaeoecol 370:209–221
Giesecke T, Bennett KD (2004) The Holocene spread of Picea abies (L.) Karst. in Fennoscandia and adjacent areas. J Biogeogr 31:1523–1548
Godwin H (1975) The history of the British flora: a factual basis for phytogeography, 2nd edn. Cambridge University Press, Cambridge
Göransson H (1986) Man and the forests of nemoral broad-leafed trees during the Stone Age. Striae 24:143–152
Goslar T, Czernik J, Goslar E (2004) Low-energy 14C AMS in Poznan radiocarbon Laboratory, Poland. Nucl Instrum Methods Phys Res B 223–224:5–11
Goslar T, Van der Knaap WO, Kamenik C, Van Leeuwen JFN (2009) Free-shape 14C age-depth modelling of an intensively dated modern peat profile. J Quat Sci 24:481–499
Granoszewski W (2003) Late Pleistocene vegetation history and climatic changes at Horoszki Duże, eastern Poland: a palaeobotanical study. Acta Palaeobot Suppl 4:3–95
Granoszewski W, Winter H, Rylova TB, Savčenko IE (2012) The course and correlation of the Late Pleistocene pollen sequences from Poland and Belarus. Przegląd Geologiczny 60:605–614
Grimm EC (1991) Tilia and Tilia Graph. Illinois State Museum, Springfield
Gross H (1935) Zur Entwicklungsgeschichte des Fichtenanteils der Rominter Heide. Forstliche Wochenschrift Silva 23
Grosse-Brauckmann G (1974) Über pflanzliche Makrofossilien mitteleuropäischer Torfe, II. Weitere Reste (Früchte und Samen, Moose u.a.) und ihre Bestimmungsmöglichkeiten. Telma 4:51–117
Grosse-Brauckmann G, Streitz B (1992) Über pflanzliche Makrofossilien mitteleuropäischer Torfe, III. Früchte und Samen, Moose und einige Gewebe (Fotos von fossilen Pflanzenresten). Telma 22:53–102
Guiter F, Andrieu-Ponel V, Digerfeldt G, Reille M, De Beaulieu JL, Ponel P (2005) Vegetation history and lake-level changes from the Younger Dryas to the present in Eastern Pyrenees (France): pollen, plant macrofossils and lithostratigraphy from Lake Racou (2000 m a.s.l.). Veget Hist Archaeobot 14:99–118
Haas JN, Richoz I, Tinner W, Wick L (1998) Synchronous Holocene climatic oscillations recorded on the Swiss Plateau and at timberline in the Alps. Holocene 83:301–309
Haneca K, Ważny T, Van Acker J, Beekman H (2005) Provenancing Baltic timber from art historical objects success and limitations. J Archaeol Sci 32:261–271
Hang T, Kalm V, Kihno K, Milkevičius M (2008) Pollen, diatom and plant macrofossil assemblages indicate a low water level phase of Lake Peipsi at the beginning of the Holocene. Hydrobiol 599:13–21
Hannon GE, Gaillard M-J (1997) The plant-macrofossil record of past lake-level changes. J Paleolimnol 18:15–28
Heikkilä M, Seppä H (2003) A 11,000 yr palaeotemperature reconstruction from the southern boreal zone in Finland. Quat Sci Rev 22:541–554
Heiri O, Tinner W, Lotter AF (2004) Evidence for cooler European summers during periods of changing meltwater to the North Atlantic. Proc Natl Acad Sci 101:15,285–15,288
Hessen DO, Walseng B (2008) The rarity concept and the commonness of rarity in freshwater zooplankton. Freshw Biol 53:2026–2035
Hoffmann MH, Litt T, Jäger EJ (1998) Ecology and climate of the early Weichselian flora from Gröbern (Germany). Rev Palaeobot Palynol 102:259–276
Hölzer A (2010) Die Torfmoose Südwestdeutschlands und der Nachbargebiete. Weissdorn-Verlag, Jena
Holzhauser H, Magny M, Zumbühl HJ (2005) Glacier and lake-level variations in west-central Europe over the last 3500 years. Holocene 15:789–801
Isarin RFB, Bohncke SJP (1999) Summer temperatures during the Younger Dryas in North-western and central Europe inferred from climate indicator plant species. Quat Res 51:158–173
Iversen J (1964) Plant indicators of climate, soil and other factors during the Quaternary. Report of the VIth International Congress on Quaternary, Warsaw, 1961, vol 2, pp 421–428
Janowská V, Komárek J (2000) Indicative value of Pediastrum and other coccal green algae in palaeoecology. Folia Geobot 35:59–82
Juggins S (2003) C2 User guide. Software for ecological and palaeoecological data analysis and visualisation. University of Newcastle upon Tyne, Newcastle upon Tyne
Karczewski M (2011) Archeologia środowiskowa zachodniobałtyjskiego kręgu kulturowego na pojezierzach. Bogucki Wydawnictwo Naukowe, Poznań-Białystok
Kłosowski S, Jabłońska E, Szańkowski N (2011) Aquatic vegetation as an indicator of littoral habitats and various stages of lake aging in north-eastern Poland. Ann Limnol 47:281–295
Kołaczek P, Karpińska-Kołaczek M, Petera-Zganiacz J (2012) Vegetation patterns under climate changes in the Eemian and Early Weichselian in Central Europe inferred from a palynological sequence from Ustków (central Poland). Quat Int 268:9–20
Kolstrup E (1979) Herbs as July temperature indicators for parts of the Pleniglacial and the Lateglacial in The Netherlands. Geol en Mijnbouw 59:377–380
Kolstrup E (1980) Climate and stratigraphy in Northwestern Europe between 30,000 BP and 13,000 BP, with special reference to The Netherlands. Meded Rijks Geol Dienst 32:181–253
Koprowski M (2013) Spatial distribution of introduced Norway spruce growth in lowland Poland: the influence of changing climate and extreme weather events. Quat Int 283:139–146
Korhola A, Rautio M (2001) Cladocera and other branchiopod crustaceans. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, zoological indicators. Kluwer, Dordrecht, pp 5–41
Kowalski J (2000) Chronologia grupy elbląskiej i olsztyńskiej kręgu zachodniobałtyjskiego (V-VII w.). Zarys problematyki Barbaricum 6:203–266
Kozłowski JK, Kaczanowski P (1998) Najdawniejsze dzieje ziem polskich. Fogra Oficyna Wydawnicza, Kraków
Krzywicki T (2002) The maximum ice sheet limit of the Vistulian Glaciation in north-eastern Poland and neighbouring areas. Geol Q 46:165–188
Kupryjanowicz M (2007) Postglacial development of vegetation in the vicinity of the Wigry Lake. Geochronometria 27:53–66
Kupryjanowicz M, Jurochnik A (2009) Pollen record of postglacial vegetation changes as contained in bottom deposts of Wigry Lake. In: Rutkowski J, Krzysztofiak L (eds) History of the lake in the light og geological and paleoecological studies. Stowarzyszenie “Człowiek i Przyroda”, Suwałki, pp 181–198
Kupryjanowicz M, Piotrowska N, Rutkowski J, Krzysztofiak L (2009) Podsumowanie. In: Rutkowski J, Krzysztofiak L (eds) Jezioro Wigry. Historia jeziora w świetle badań geologicznych i paleoekologicznych. Stowarzyszenie “Człowiek i Przyroda”, Suwałki, pp 256–264
Lamentowicz M, Obremska M, Mitchell EAD (2008) Autogenic succession, land-use change, and climatic influences on the Holocene development of a kettle-hole mire in Northern Poland. Rev Palaeobot Palynol 151:21–40
Lang G (1994) Quartäre Vegetationsgeschichte Europas. Fischer, Jena
Latałowa M, Van der Knaap WO (2006) Late Quaternary expansion of Norway spruce Picea abies (L.) Karst. in Europe according to pollen data. Quat Sci Rev 25:2780–2805
Lauterbach S, Brauer A, Andersen N, Danielopol DL, Dulski P, Hüls M, Milecka K, Namiotko T, Plessen B, Von Grafenstein U, DecLakes participants (2011) Multi-proxy evidence for early to mid-Holocene environmental and climatic changes in northeastern Poland. Boreas 40:57–72
Litt T, Stebich M (1999) Bio- and chronostratigraphy of the lateglacial in the Eifel region, Germany. Quat Int 61:5–16
Litt T, Brauer A, Goslar T, Merkt J, Bałaga K, Müller H, Ralska-Jasiewiczowa M, Stebich M, Negendank JFW (2001) Correlation and synchronisation of Lateglacial continental sequences in northern central Europe based on annually laminated lacustrine sediments. Quat Sci Rev 20:1,233–1,249
Lorenc H (2005) Atlas klimatu Polski. IMGW, Warszawa
Łowmiański H (1986) Studia nad dziejami Wielkiego Księstwa Litewskiego. Wydawnictwo Naukowe UAM, Poznań
Madeja J, Bałaga K, Harmata K, Nalepka D (2004) Pteridium aquilinum (L.) Kuhn–Bracken. In: Ralska-Jasiewiczowa M, Latałowa M, Wasylikowa K, Tobolski K, Madeyska T, Wright Jr HE, Turner C (eds) Late glacial and Holocene history of vegetation in Poland based on isopollen maps. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków, pp 327–335
Magny M (2004) Holocene climate variability as reflected by mid-European lake-level fluctuations and its probable impact on prehistoric human settlements. Quat Int 113:65–79
Magny M, Vannière B, De Beaulieu JL, Bègeot C, Heiri O, Millet L, Peyron O, Walter-Simonnet AV (2007) Early-Holocene climatic oscillations recorded by lake-level fluctuations in west-central Europe and in central Italy. Quat Sci Rev 26:1,951–1,964
Maj J, Gałka M (2012) Wiek i historia rozwoju torfowiska w Pakosławicach (południowo-zachodnia Opolszczyzna) na podstawie badań paleobotanicznych. (The age and development history of biogenic accumulation basin in Pakosławice (south-western Opole Province) in the light of palaeobotanical research, in Polish, English summary). Przegląd Geologiczny 60:110–116
Markowski R, Stasiak J (1988) Juncus subnodulosus Schrank. In: Jasiewicz A (ed) Materiały do poznania gatunków rzadkich i zagrożonych Polski. Fragm Flor Geobot 33:386–397
Marks L (2002) Last glacial maximum in Poland. Quat Sci Rev 21:103–110
Matusiewicz A, Rodziwonowicz T, Skłodowski K (2005) Historia Suwałk do 1945 roku. In: Kopciała J (ed) Suwałki miasto nad Czarną Hańczą. Wydawnictwo Hańcza, Suwałki, pp 89–472
Matuszkiewicz JM (2001) Zespoly leśne Polski. Wydawnictwo Naukowe PWN, Warszawa
Mauquoy D, Yeloff D, Van Geel B, Charman D, Blundell A (2008) Two decadally resolved records from north-west European peat bogs show rapid climate changes associated with solar variability during the mid-late Holocene. J Quat Sci 23:745–763
Mayewski PA, Rohling EE, Curt Stager J et al (2004) Holocene climate variability. Quat Res 62:243–255
Meusel H, Jager E, Weinert E (1965) Vergleichende Chorologie der Zentraleuropäischen Flora. Bd. I. VEB G. Fischer, Jena
Milecka K, Szeroczyńska K (2005) Changes in macrophytic flora and planktonic organisms in Lake Ostrowite, Poland, as a response to climatic and trophic fluctuations. Holocene 15:74–84
Milecka K, Kowalewski G, Szeroczyńska K (2011) Climate-related changes during the Late glacial and early Holocene in northern Poland, as derived from the sediments of Lake Sierzywk. Hydrobiol 676:187–202
Mirek Z, Piękoś-Mirkowa H, Zając A, Zając M (2002) Flowering plants and Pteridophytes of Poland. A checklist. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Moen A (1999) National atlas of Norway: vegetation. Norwegian Mapping Authority, Hönefoss
Mortensen MF, Birks HH, Christensen C, Holm J, Noe-Nygaard N, Odgaard B, Olsen J, Rasmussen KL (2011) Lateglacial vegetation development in Denmark—new evidence based on macrofossils and pollen from Slotseng, a small-scale site in southern Jutland. Quat Sci Rev 30:2,534–2,550
Moschen R, Kühl N, Peters S, Vos H, Lücke A (2011) Temperature variability at Dürres Maar, Germany during the Migration Period and at High Medieval Times, inferred from stable carbon isotopes of Sphagnum cellulose. Clim Past 7:1,011–1,026
Novik A, Punning J-M, Zernitskaya V (2010) The development of Belarusian lakes during the Late glacial and Holocene. Eston J Earth Sci 59:63–79
Nowakowski W (1986) Stan i potrzeby badań nad zachodniobałtyjskim kręgiem kulturowym na terenie Polski ze szczególnym uwzględnieniem kultury bogaczewskiej. Stan i potrzeby badań nad młodszym okresem przedrzymskim i okresem wpływów rzymskich w Polsce. Materiały z konferencji, Kraków, 14–16 listopad 1984, Kraków, pp 375–393
Okulicz-Kozaryn J (1993) Szurpiły-zespół osadnictwa od III w. p.n.e. do XIII w. n.e. Przewodnik LXIV Zjazdu Polskiego Towarzystwa Geologicznego na Ziemi Suwalskiej, 9–12 września 1993, Warszawa, pp 139–146
Ołtuszewski W (1937) Historia lasów Pojezierza Suwalsko-Augustowskiego w świetle analizy pyłkowej. Poznańskie Towarzystwo Przyjaciół Nauk. Prace Kom Matemat Przyrod B 8:1–65
Ozola I, Cernina A, Kalnina L (2010) Reconstruction of palaeovegetation and sedimentation conditions in the area of ancient Lake Burtnieks, northern Latvia. Eston J Earth Sci 59:164–179
Pip E, Simmons K (1986) Aquatic angiosperms at unusual depths in Shoal lake, Manitoba Ontario. Canad Field Natur 100:354–358
Podbielkowski Z, Tomaszewicz H (1996) Zarys hydrobotaniki. Państwowe Wydawnictwo Naukowe, Warszawa
Punning J-M, Kangur M, Koff T, Possnert G (2003) Holocene lake-level changes and their reflection in the paleolimnological records of two lakes in northern Estonia. J Paleolimnol 29:167–178
Ralska-Jasiewiczowa M (1966) Osady denne Jeziora Mikołajskiego na Pojezierzu Mazurskim w świetle badań paleobotanicznych (Bottom sediments of the Mikołajki Lake (Masurian Lake District, in Polish, English summary) in the light of palaeobotanical investigations. Acta Palaeobot 7:1–118
Ralska-Jasiewiczowa M, Latałowa M (1996) Synthesis of palaeoecological events in Poland. In: Berglund BE, Birks HJB, Ralska-Jasiewiczowa M, Wright HE (eds) Palaeoecological events during the last 15,000 years. Regional syntheses of palaeoecological studies of lakes and mires. Wiley, Chichester, pp 403–472
Ralska-Jasiewiczowa M, Nalepka D, Goslar T (2003) Some problems of forest transformation at the transition to the oligocratic/Homo sapiens phase of the Holocene interglacial in northern lowlands of central Europe. Veget Hist Archaeobot 12:233–247
Reimer PJ, Baillie MGL, Bard E, Bayliss A, Beck JW, Blackwell PG, Ramsey CB, Buck CE, Burr GS, Edwards RL, Friedrich M, Grootes PM, Guilderson TP, Hajdas I, Heaton TJ, Hogg AG, Hughen KA, Kaiser KF, Kromer B, McCormac FG, Manning SW, Reimer RW, Richards DA, Southon JR, Talamo S, Turney CSM, Van der Plicht J, Weyhenmeye CE (2009) IntCal09 and Marine09 radiocarbon age calibration curves, 0–50,000 years cal BP. Radiocarbon 51:1,111–1,150
Saarse L, Niinemets E, Amon L, Heinsalu A, Veski S, Sohar K (2009) Development of the late glacial Baltic basin and the succession of vegetation cover as revealed at Palaeolake Haljala, northern Estonia. Eston J Earth Sci 58:317–333
Salmina L (2004) Factors influencing distribution of Cladium mariscus in Latvia. Ann Bot Fenn 41:367–371
Sarmaja-Korjonen K (2001) Correlation of fluctuations in cladoceran planktonic: littoral ratio between three cores from a small lake in southern Finland: Holocene water-level changes. Holocene 11:53–63
Sarmaja-Korjonen K, Seppä H (2007) Abrupt and consistent responses of aquatic and terrestrial ecosystems to the 8200 cal. yr cold event: a lacustrine record from Lake Arapisto, Finland. Holocene 17:457–467
Seppä H, Poska A (2004) Holocene annual mean temperature changes in Estonia and their relationship to solar insolation and atmospheric circulation patterns. Quat Res 61:22–31
Siitonen S, Väliranta M, Weckström J, Juutinen S, Korhola A (2011) Comparison of Cladocera-based water-depth reconstruction against other types of proxy data in Finnish Lapland. Hydrobiol 676:155–172
Sokołowski AW (1973) Zbiorowiska leśne Suwalskiego Parku Krajobrazowego. Prace Białost Tow Nauk 19:67–83
Środoń A (1967) Świerk pospolity w czwartorzędzie Polski (The common spruce in the Quaternary of Poland). Acta Palaeobot 8:3–59
Stachowicz-Rybka R, Gałka M, Aleksandowicz WP, Aleksandrowicz SW (2009) Plant macrofossils and malacocoenoses of Quaternary mineral-organic sediments at Starunia palaeontological site and vicinity (Carpathian region, Ukraine). Ann Soc Geol Polon 79:297–313
Stančikaitė M, Kabailienė M, Ostrauskas T, Guobytė R (2002) Environment and man around Lakes Dñba and Pelesa, SE Lithuania, during the Late glacial and Holocene. Geol Q 46:391–409
Stančikaitė M, Baltrǔnas V, Šinkunas P, Kisielinė D, Ostrauskas T (2006) Human response to the Holocene environmental changes in the Biržulis Lake region, NW Lithuania. Quat Int 150:113–129
Stančikaitė M, Šinkunas P, Šeirienė V, Kisielinė D (2008) Patterns and chronology of the Lateglacial environmental development at Pamerkiai and Kašučiai, Lithuania. Quat Sci Rev 27:127–147
Stančikaitė M, Kisieliė D, Moe D, Vaikutiė G (2009) Lateglacial and early Holocene environmental changes in northeastern Lithuania. Quat Int 207:80–92
Stančikaitė M, Baltrūnas V, Karmaza B, Karmazienė D, Molodkov A, Ostrauskas T, Obukhowsky V, Sidorovich W (2011) The Late glacial history of Gornitsa foreland and Kovaltsy Palaeolithic site, W Belarus. Baltica 24:25–36
Starkel L (2011) Present-day events and the evaluation of Holocene palaeoclimatic proxy data. Quat Int 229:2–7
Starkel L, Pazdur A, Pazdur MF, Wicik B, Więckowski K (1998) Lake-level changes and palaeohydrological reconstructions during the Holocene. In: Ralska-Jasiewiczowa M, Goslar T, Madeyska T, Starkel L (eds) Lake Gościąż, Central Poland. A monographic study. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków, pp 225–229
Stasiak J (1970) Wstępne wyniki badań paleogeograficznych w Korklinach, pow. Suwalski. Rocz Białost 9:197–209
Swindles GT, Plunkett G, Roe HM (2007) A multiproxy climate record from a raised bog in County Fermanagh, Northern Ireland: a critical examination of the link between bog surface wetness and solar variability. J Quat Sci 22:667–679
Swindles GT, Blundell A, Roe HM, Hall VA (2010) A 4500-year proxy climate record from peatlands in the North of Ireland: the identification of widespread summer ‘drought phases’? Quat Sci Rev 29:1,577–1,589
Szeroczyńska K (1985) Cladocera jako wskaźnik ekologiczny w późnoczwartorzędowych osadach jeziornych Polski Północnej. (Cladocera as ecological indicator in late quaternary lacustrine sediments, Northern Poland). Acta Palaeontol Pol 30:3–69
Szeroczyńska K, Sarmaja-Korjonen K (2007) Atlas of subfossil Cladocera from central and northern Europe. Friends of the Lower Vistula Society, Świecie
Szeroczyńska K, Zawisza E (2007) Paleolimnology—history of development of Polish lake on the light of subfossil Cladocera analysis (in Polish, English abstract) Stud Lim et Tel 1:51–60
Szeroczyńska K, Zawisza E (2011) Records of the 8200 cal BP cold event reflected in the composition of subfossil Cladocera in the sediments of three lakes in Poland. Quat Int 233:185–193
Szkiruć Z (1986) Suwalski Park Krajobrazowy. Ludowa Spółdzielnia Wydawnicza, Warszawa
Terasmaa J (2011) Lake basin development in the Holocene and its impact on the sedimentation dynamics in a small lake (southern Estonia). Eston J Earth Sci 60:159–171
Tinner W, Lotter AF (2001) Central European vegetation response to abrupt climate change at 8.2 ka. Geology 29:2,551–2,554
Tobolski K (1976) Przemiany klimatyczno-ekologiczne w okresie czwartorzędu a problem zmian we florze. Phytocenosis 5:187–197
Tobolski K (1987) Holocene vegetational development based on the Kluki reference site in the Gardno-Łeba Plain. Acta Palaeobot 27:179–222
Tobolski K (2000) Przewodnik do oznaczania torfów i osadów jeziornych. Wydawnictwo Naukowe PWN, Warszawa
Tobolski K (2006) Torfowiska Parku Narodowego „Bory Tucholskie”. Park Narodowy “Bory Tucholskie”, Charzykowy
Tuittila E-S, Väliranta M, Laine J, Korhola A (2007) Quantifying patterns and controls of mire vegetation succession in a southern boreal bog in Finland using partial ordinations. J Veget Sci 18:891–902
Väliranta M, Korhola A, Seppä H, Tuittila E-S, Sarmaja-Korjonen K, Laine J, Alm J (2007) High-resolution reconstruction of wetness dynamics in a southern boreal raised bog, Finland, during the late Holocene: a quantitative approach. Holocene 17:1093–1107
Valovirta V (1962) Cladium mariscus in Finnland während der Postglazialzeit. Bull Comm Géol Finl 197:1–66
Van der Linden M, Van Geel B (2006) Late Holocene climate change and human impact recorded in a south Swedish ombrotrophic peat bog. Palaeogeogr Palaeoclimonol Palaeoecol 240:649–667
Van Geel B, Buurman J, Waterbolk HT (1996) Archeological and paleoecological indications of an abrupt climate change in The Netherlands, and evidence for climatological teleconnections around 2650 BP. J Quat Sci 11:451–460
Velichkevich FU, Zastawniak E (2006) Atlas of the Pleistocene vascular plant macrofossils of Central and Eastern Europe. Part 1—Pteridophytes and monocotyledons. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Velichkevich FU, Zastawniak E (2009) Atlas of the Pleistocene vascular plant macrofossils of Central and Eastern Europe. Part 2—Pteridophytes and monocotyledons. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Voigt R, Grüger G, Baier J, Meischner D (2008) Seasonal variability of Holocene climate: a palaeolimnological study on varved sediments in Lake Jues (Harz Mountains, Germany). J Paleolimnol 40:1,021–1,052
Vuorela I (1986) Palynological and historical evidence of slash-and-burn cultivation in South Finland. In: Behre K-E (ed) Anthropogenic indicators in pollen diagrams. Balkema, Rotterdam, pp 53–64
Wacnik A (2009) Vegetation development in the Lake Miłkowskie area, north-eastern Poland, from the Plenivistulian to the late Holocene. Acta Palaeobot 49:287–335
Wacnik A, Goslar T, Czernik J (2012) Vegetation changes caused by agricultural societies in the Great Mazurian Lake District. Acta Palaeobot 52:59–104
Wasylikowa K (1964) Roślinność i klimat późnego glacjału w środkowej Polsce na podstawie badań w Witowie koło Łęczycy. Biul Peryglac 13:261–417
Ważny T (2005) The origin, assortments and transport of Baltic Timber. In: Van de Velde C, Beeckman H, Van der Acker J, Verhaeghe F (eds) Constructing wooden images. Brussels University Press, Brussels, pp 115–126
Weninger B, Alram-Stern E, Bauer E, Clare L, Danzeglocke UJ, Ris O, Kubatzki C, Rollefson G, Todorova H (2006) Climate forcing due to the 8200 cal BP event observed at Early Neolithic sites in the eastern Mediterranean. Quat Res 66:401–420
Wiliński K (1984) Walki polsko-pruskie w X-XIII w. Acta Univer Lodziensis. (Folia Hist 15) Łódź
Wingfield RA, Murphy KJ, Hollingswordth P, Gaywood MJ (2004) The ecology of Najas flexilis. Scottish Natural Heritage and Commission Report No. 017 (ROAME No. F98PA02)
Woś A (1999) Klimat Polski. Wydawnictwo Naukowe PWN, Warszawa
Wróblewski W, Sawicka L (2012) Relikty osadnictwa jaćwieskiego w obrębie Suwalskiego Parku Krajobrazowego. In: Rant-Tanajewska M, Świerubska T, Dziubiak M, Buczyński P (eds) XXXV lat Suwalskiego Parku Krajobrazowego. Suwalski Park Krajobrazowy, Jeleniewo, pp 39–44
Yu GE, Harrison SP (1995) Holocene changes in atmospheric circulation patterns as shown by lake status changes in northern Europe. Boreas 24:260–268
Zachowicz J, Ralska-Jasiewiczowa M, Miotk-Szpiganowicz G, Nalepka D (2004) Ulmus L.-Elm. In: Ralska-Jasiewiczowa M, Latałowa M, Wasylikowa K, Tobolski K, Madeyska T, Wright Jr HE, Turner C (eds) Late glacial and Holocene history of vegetation in Poland based on isopollen maps. W. Szafer Institute of Botany, Polish Academy of Sciences, Kraków, pp 224–235
Zalewska-Gałosz J (2008) Rodzaj Potamogeton L. w Polsce—taksonomia i rozmieszczenie. The genus Potamogeton L. in Poland—taxonomy and distribution. Instytut Botaniki Uniwersytetu Jagiellońskiego, Kraków
Zarzycki K, Trzcińska-Tacik H, Różański W, Szeląg Z, Wołek J, Korzeniak U (2002) Ecological indicator values of vascular plants of Poland. Biodiversity of Poland, Kraków
Zawisza E, Szeroczyńska K (2007) The development history of Wigry Lake as shown by subfossil Cladocera. Geochronometria 27:67–74
Zernitskaya V, Mikhailov N (2009) Evidence of early farming in the Holocene pollen spectra of Belarus. Quat Int 203:91–104
Acknowledgments
The scientific work financed by the Ministerstwo Nauki i Szkolnictwa Wyższego (Ministry of Science and Higher Education) as research project N N305 3259 33, and was performed during 2007–2010. We express our gratitude to employees of the Suwalski Landscape Park for assistance in the field work and sediment sampling, Małgorzata Suchorska for assistance in sample preparation, Milena Obremska for drawing up the pollen diagrams, and Krystyna Milecka for constructive comments on an earlier version of the paper. We would like to thank the anonymous reviewers and Karl-Ernst Behre for their comprehensive reading of the manuscript and constructive suggestions for its improvement.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K.-E. Behre.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Gałka, M., Tobolski, K., Zawisza, E. et al. Postglacial history of vegetation, human activity and lake-level changes at Jezioro Linówek in northeast Poland, based on multi-proxy data. Veget Hist Archaeobot 23, 123–152 (2014). https://doi.org/10.1007/s00334-013-0401-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-013-0401-7