Abstract
A 50 m-long radiocarbon dated core was studied through sediment and pollen analysis to reconstruct the Holocene mangrove and environmental changes at a coastal site Pakhiralaya in the Sundarban Biosphere Reserve in the western Ganga–Brahmaputra Delta, India. This biosphere reserve harbours a diverse mangrove ecosystem and supports a large number of people living in the area. Pollen and stratigraphic data indicate the existence of a brackish water estuarine mangrove swamp forest in this area during the last 9880 cal yr b.p. The development of the mangrove forest is not shown continuously in the Holocene record. Rapid transgression of the sea (9240 cal yr b.p.) halted the development of the mangrove. After about 8420 cal yr b.p. mangrove recolonised the area and persisted until 7560 cal yr b.p. as a result of a balance between the sedimentation and sea level fluctuation. The mangrove disappeared again from the site until 4800 cal yr b.p. because of a high sedimentation rate and possible delta progradation with loss of habitats. The reappearance of mangrove at the study site occurred with a return of a brackish water estuarine environment and the site then gradually became supra tidal during the mid-late Holocene. The continuity of the mangrove development and dynamics was interrupted by the fluctuating sea levels. Climatic fluctuations were viewed as an indirect factor influencing the mangrove ecosystem.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
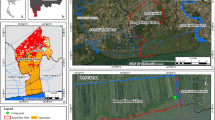
The coastal area of the Ganga–Brahmaputra delta of India and Bangladesh is ecologically unique for its diverse mangrove habitats known as the Sundarban mangrove forest. Most part of this forest is located on the low-lying southern delta plain (Fig. 1) at an elevation of about 1–2 m above present-day mean sea level (Delft Hydraulics 1989; Milliman et al. 1989; Jelgersma 1994; Broadus 1996). This forest constitutes the largest single area of mangroves in the world (Chanda 1977; Hussain and Acharya 1994; Sanyal 1996; Naskar and Mandal 1999). The Sundarban mangrove forest is also the only mangrove tiger habitat in the world and is internationally recognised as a world heritage site for its diverse mangrove flora and fauna. The major plant taxa of the present day Sundarban forest are Avicennia spp., Excoecaria sp., Sonneratia spp., Rhizophora spp., Aegiceras sp., Acanthus sp., Acrostichum sp., Bruguiera spp., Ceriops spp., Heritiera sp., Kandelia sp., Xylocarpus spp., Nypa sp., Phoenix sp., Suaeda sp. and Porteresia sp. Avicennia is the dominant tree in the forest followed by Excoecaria, Phoenix and Ceriops.
The mangrove forest of the Sundarbans acts as a buffer zone that helps to reduce the impact of landward driven tides, storms and cyclones and thus protects the hinterland from environmental and economic hazards. The rapidly growing population of this region also is heavily dependent on this forest for their livelihood (Naskar 1988; Milliman et al. 1989; Hussain and Acharya 1994; Chaudhuri and Choudhury 1994). Currently, this ecosystem is under stress through anthropogenic factors in the form of land reclamation and decreased river flow influx. Protection of this mangrove forest is essential and urgent.
The mangroves are physiologically distinct low-lying coastal vegetation highly adapted to the coastal environment of the tropics and sub-tropics. The physiography of the area, frequency and duration of flooding, the nature of the soil and the degree of salinity are the important environmental factors that determine the growth, development, richness, diversity and zonation of the mangrove at the local scale (Lugo and Snedaker 1974; Hutchings and Saenger 1987; Naskar and Guha Bakshi 1987; Woodroffe 1990; Nazrul-Islam 1993; Bunt 1996; Blasco et al. 1996; Ellison 2005; Naskar and Mandal 1999; Duke et al. 1998; Grindrod et al. 2002).
Owing to their intertidal habitats mangrove ecosystems are sensitive to any change in sea level and are considered as a sea level indicator. They can move landward or seaward according to sea transgression or regression, respectively. Therefore this ecosystem is sometimes referred to as a dancing ecosystem (Sanyal 1990). Mangrove pollen recovered from the sediments has been used by several workers as an indicator of sea level changes in the coastal areas (Woodroffe et al. 1985; Sen and Banerjee 1990; Chanda and Hait 1996; Ellison 1994, 2005; Kamaludin 1993; Hait et al. 1996a, b; Blasco et al. 1996; Islam and Tooley 1999; Lézine 1996; Lézine et al. 2002; Nguyen et al. 2000; Behling et al. 2001, 2004; Campo and Bengo 2004; Yulianto et al. 2004, 2005).
Our knowledge of the last 7,000 years of vegetation history of the areas north and north west of the present study site is well documented (Gupta 1981; Barui and Chanda 1992; Sen and Banerjee 1990; Chanda and Hait 1996; Hait et al. 1994a, b, Hait et al. 1996a, b). All these studies indicate the presence of a humid climate in these areas for the last 7,000 years. A marine core from off Karwar, western India, indicates a less humid climate from ca. 3,500 year b.p. (Caratini et al. 1994).
However, the Holocene vegetation and environmental history of the coastal areas of the western Ganga–Brahmaputra Delta is poorly understood. The analysis of the Holocene coastal environmental history is important in order to understand the modern and future mangrove development and dynamics in this area for coastal management and conservation. In this study we report the first, although discontinuous, record of Holocene vegetation and environmental history based on a 50 m radiocarbon dated coastal core from Pakhiralaya.
Study site
Pakhiralaya (22°09′ N, 88° 47′E) is located in the lower flood plain of the southwestern Ganga–Brahmaputra Delta (Fig. 1). The plain is about 2 m above the present sea level and the topography is gentle. Several rivers flow through the plain and discharge into the bay. Before discharging into the bay, they form an intricate network of islands, creeks and creeklets. These are the areas that are inhabited by extensive mangrove forests. The Vidya river is the main river system in the study area, separating the site from the present day mangrove forest and the tiger project areas.
The study area receives about 1,800 mm rainfall per year. The area is inundated during the monsoon period (June–September) and becomes dry during the winter (November–January). Mean annual maximum and minimum temperatures are 29 and 20°C, respectively. The humidity is between 70 and 80% (Chaudhuri and Choudhury 1994). The site was once extensively covered by mangroves. In recent times, the area has largely been converted into settlement, agricultural and aquacultural farms (Naskar 1988; Guha Bakshi and Naskar 1994; Hussain and Acharya 1994; Chaudhuri and Choudhury 1994; Stanley and Hait 2000). Today there is some secondary vegetation, consisting mainly of Avicennia trees, growing along the fringe of the study area.
Numerous records of sea level changes during the last glacial-deglacial transition and the Holocene from all over the world are available (Pirazzoli 1991, 1996; Fleming et al. 1998). It is now established that the sea level was ca. 125 ± 5 m lower at the time of the last glacial maximum. The early Holocene rapid rise in sea level of 10 m Ka−1 is mostly attributed to climatically controlled glacio-eustatic factors and there has been a very little change of eustatic sea level for the last 7,000 years. However, the actual scenario on a regional/local basis can depart significantly from the global pattern because of tectonic and glacio-hydro-isostatic effects. Neotectonic factors were mainly responsible for changes in the sea level during the Holocene period at the study site (Stanley and Hait 2000) and in the adjacent Bangladesh region (Islam 2001).
Material and methods
A 50 m sediment core was recovered through boring by a combination of augering and direct rotary coring from the study site (Fig. 1). For palynological studies, 0.5 cm3 sediment samples were taken at 10 cm intervals along the core. Several sand samples were analysed and proved barren or had only a few palynomorphs and were not used for palynological study. On this basis not all the sand samples were processed. Samples were prepared following standard pollen analytical techniques including acetolysis (Erdtman 1969; Faegri and Iversen 1989). Sample residues were mounted on microscope slides in a glycerine jelly medium. Published pollen morphological descriptions were consulted for identification of pollen grains and spores (Thanikaimoni 1987). The reference collection of the second author was used for identification purposes. The pollen counts were made with a Carl Zeiss microscope at a magnification of 400×. Most of the pollen samples were counted to a minimum of 300 grains including arboreal and non-arboreal pollen. The pollen sum excludes fern and fungal spores, and micro foraminifera. The palynological data are presented in pollen diagrams as percentages of the pollen sum. Identified taxa were grouped into mangrove, non-mangrove trees, herbs, hypersaline herbs (HSB), aquatics, ferns, mangrove ferns (MF), fungi and marine (foraminifera) on the basis of their ecological requirement and life form. The software TILIA was used for calculations, CONISS for the cluster analysis of pollen taxa and TGVIEW to plot the pollen diagrams (Grimm 1987).
Results
Stratigraphy
The dominant lithological unit is grey silty clay (Table 1). Variations between this and grey clayey silt are frequent. The sequences of grey silty clay at the bottom (49.8–40.8 m) and grey clayey silt in the middle part of the core are rich in decomposed wood fragments. The grey fine sand (40.8–30.8 m) contains fragments of shells. Alternating sequences of grey silty mud and grey sandy silt are seen between the depths of 30.6 and 26.4 m. A large piece of wood was also found in situ in the core at a depth of 22.44–22.12 m. The sediments of the upper part of the core (1.5–0 m) appear to be disturbed and oxidised, and contain plant material related to farming.
Chronology
Five radiocarbon dates provide time control (Tables 2, 3). The dates were calibrated for calendrical age using the program CalPal (Weninger et al. 2004). Because of the mostly terrestrial deposits and the dating mainly of wood fragments, marine reservoir corrections (Southon et al. 2002) were not applied. According to the radiocarbon dates, the studied sediment core deposits did not accumulate continuously. Gaps exist in the 50 m sediment core and only parts of the core had sufficient pollen content for analysis. Therefore only three periods of the Holocene mangrove development are recorded. Based on the radiocarbon dates, the deposits between 49.85 and 41.00 m core depth have an age of between about 9880 and 9240 cal yr b.p. (Zone P I). The age of the deposits between 24.80 and 24.60 m (Zone P II) is difficult to estimate. Linear interpolation between the dates 9240 ± 110 cal yr b.p. at 41.30 m and 8310 ± 89 cal yr b.p. at 22.30 m depth in the core would give an age of about 8420 cal yr b.p. for this 0.20 m of deposit. The age could be much greater. The estimated age for the 19.80 m depth is based on linear extrapolation of the radiocarbon age of 8310 ± 89 cal yr b.p. at 22.30 m and 7950 ± 68 cal yr b.p. at 21.15 m depth in the core. The deposits between the depths of 24.60 and 19.80 m then have an age between 8420 and 7560 cal yr b.p. (Zone P III). The deposits between the depths of 2.30 and 1.50 m (Zone P IV) are of an uncertain late Holocene age.
Palynological analysis
About 161 samples were analysed palynologically, of which 124 samples had sufficient pollen content for analysis of a minimum of 300 pollen grains. Microscopic studies recorded 31 taxa, including 25 angiosperms, 3 pteridophytes, 1 fungus, 1 foraminifer and 1 acritarch. Some of these types have been identified to species level (Nypa fruiticans, Acrostichum aureum). The overall frequency of occurrence reveals that genera like Sonneratia and Rhizophora dominate the assemblages throughout the core. Taxa such as Acanthus, Avicennia, Excoecaria, Bruguiera, Xylocarpus, Heritiera, Nypa fruiticans (mangrove palm), Poaceae, Suaeda and Phoenix palludosa (mangrove palm) are abundant in restricted stratigraphic sequences. Acrostichum aureum, the only mangrove fern, is abundant in some stratigraphic sequences and particularly in the upper part of the core. The remaining taxa ranged from common to rare. Most of the dispersed palynomorphs recorded are comparable with the extant mangrove vegetation of the modern Sundarbans (Thanikaimoni 1987).
Four pollen zones (Zone P I–P IV) are recognised (Figs. 2, 3, 4). The zonation is based on marked changes in the pollen assemblages and stratigraphically restricted occurrence of the taxa. Taxa that are restricted stratigraphically and important ecologically are also mentioned in the pollen zone.
Pollen zones
Three levels with a significant pollen content have been found in the 50 m sediment core. These are described as Pollen Zones P I, II, III and IV:
Zone P I (49.85–41 m, 9880–9240 cal yr b.p., 80 samples, important taxa: Rhizophora, Sonneratia, Bruguiera, Avicennia, Aegiceras, Heritiera, Excoecaria, Acanthus). This zone is represented by large abundances of different mangrove pollen taxa. Non-mangrove pollen such as Borassus and Bombax, and aquatic pollen are rare while grass pollen (Poaceae) is common. Monolete fern spores are also present in high numbers together with the mangrove fern spore Acrostichum aureum. Marine taxa (foraminifera) are also recorded in this zone. Some samples contain pollen belonging to the hypersaline herb (salt tolerant) Suaeda.
Zone P II (24.8–24.6 m, about 8420 cal yr b.p. (linear interpolated), 2 samples, important taxa: Phoenix, Cyperaceae, Poaceae).
This zone is separated from zone I by a thick, mostly sandy deposit and is distinguished from the previous zones by a lower representation of mangrove pollen taxa. The complete absence of mangrove fern, hypersaline herbs, aquatics, fungi and marine taxa is noticeable in this zone. Phoenix paludosa is dominant in this zone among the mangroves along with a very high representation of Cyperaceae pollen grains. Baringtonia represents the non-mangrove pollen taxa.
Zone P III (24.6–19.8 m, about 8420–7560 cal yr b.p. (linear interpolated), 37 samples, important taxa: Sonneratia, Rhizophora, Xylocarpus, Nypa and abundant fern spores).
A high diversity of mangrove pollen is again noticeable in this zone. Non-mangrove, fungus and marine taxa are rare, herbs and ferns are common and aquatics are absent in this zone. Sonneratia pollen regains its supremacy in this zone followed by Rhizophora. The other mangrove pollen including Nypa is also present in lower percentages. Xylocarpus has a higher representation in the upper part of the zone. Non-mangrove pollen is represented only by Cocos. Herbs are represented by Poaceae and the hypersaline herb Suaeda. The sediment of this zone is also marked by the occurrence of a large piece of wood (cf. Heritiera) in situ.
Zone P IV (2.3–1.5 m, late Holocene, five samples, important taxa: Phoenix, Poaceae, Excoecaria, Suaeda, Acrostichum).
This zone is separated from the previous pollen zone by a thick silty and sandy deposit while the representation of mangrove pollen is lower in comparison to herbs, hypersaline herbs and mangrove fern. Non-mangroves are represented in relatively high numbers. Aquatics are rare while fungi and marine taxa are absent in this zone which is characterised by the overwhelming representation of Poaceae pollen followed by Cyperaceae and the salt tolerant Suaeda. Phoenix paludosa pollen is the dominant mangrove pollen taxon followed by Excoecaria, Avicennia, Acanthus, Bruguiera and Rhizophora. Acrostichum spores are abundant.
Interpretation and discussion
The sediments of the 50 m deep core were deposited, with interruptions, over the last 9880 cal yr b.p. by brackish water estuarine mangrove swamps, shallow marine and fluvial environments (Figs. 2, 3, 4). A model of the Holocene mangrove development and dynamics, and coastal environmental changes is given in Fig. 5a–e.
Diagramatic representation of the Holocene mangrove development, dynamics and coastal environment at Pakhiralaya, western Ganga–Brahmaputra Delta, India. a Development of mangrove at the site as a result of the early Holocene sea level rise. Drowning of the area provided the accommodation space for the development of the mangrove ecosystem, b mangrove could not keep pace with the rapid transgression of the sea and therefore disappeared, c the relative stability of the sea level was responsible for the re-colonisation of the mangrove at the site, d intense fluvial activity together with the deceleration of the sea level rise was responsible for vertical accretion of sediment beyond the tidal limit and hence the destruction of the mangrove habitat, e Present day situation––the site is outside the mangrove zone; the tidal intensity and frequency is low; the area has been reclaimed during the last century for agriculture and settlement
Early Holocene coastal environment and mangrove dynamics
Pollen and stratigraphic data indicate the existence of brackish water estuarine mangrove swamp at a depth of about 49.80 m at the study site since 9880 cal yr b.p. (Table 1, Figs. 2, 4, 5a). This mangrove ecosystem developed as a result of the early Holocene sea level rise.
Drowning of the area provided the accommodation space for the development of this mangrove ecosystem. The rich and diverse mangrove pollen in the sediment (49.8–40.8 m core depth) indicate that mangroves became established, and the high occurrence of Rhizophora and Sonneratia in Zone P I suggests an intertidal environment with a mangrove forest in the study area (Fig. 5a). Variations in abundance of the major mangrove taxa in this zone are related to the local environmental conditions; namely physiography, salinity, frequency and duration of inundation. Absence of mangrove peat in this horizon indicates a high rate of sedimentation of mangrove swamp sediments in a highly energised lower part of the intertidal zone. Mangrove persisted at the site for about 650 years from 9880 to 9240 cal yr b.p. (Figs. 2, 4).
After this period, the area was drowned as a result of a rapid transgression by the sea. The mangrove could not keep pace with this rise in sea level and it therefore disappeared (Fig. 5b). The sediments deposited between 40.8 and 30.8 m are characterised by fine-grained grey sand with very poor representation of pollen grains, the frequent occurrence of fragments of marine shells suggesting shallow marine conditions. The acceleration slowed and fluctuations in relative sea level may have occurred in the upper part as it is evident from the alternation of sandy silt and silty mud between 30.8 and 26.5 m (Figs. 2, 4). These alternations suggest a variable influence of fluvial processes on the system.
The signature of fluvial processes in the estuarine marine shallows is also observable during Zone P II, which is characterised by the frequent occurrence of Cyperaceae and the non-mangrove tree Barringtonia. Mangrove trees are present but rare except for the mangrove palm Phoenix paludosa, which at the present day grows at or just above the usual tide level. The decrease in Cyperaceae pollen is abrupt, indicating the loss of the transitional sequence from marine to fluvio-marine, possibly because of erosion.
Zones P II and P III (24.8–19.8 m) show the return of an intertidal habitat (Fig. 5c). The relative stability of the sea level was responsible for the re-colonisation by mangrove of the site from about 8420 until ca. 7560 cal yr b.p. All taxa recorded in this zone had already occurred in Zone PI. The dominant mangrove taxon in this zone was Sonneratia. Rhizophora and Xylocarpus were among the other mangrove trees present during this period.
The occurrence of an early mangrove swamp at a depth of 49.80–41.00 m (9880–9240 cal yr b.p., Zone P I) and 24.8–19.8 m (about 8420–7560 cal yr b.p., Zones P II–III) indicates the unstable nature of the area. The frequently used global sea level curve (Chappel and Shackleton 1986; Fairbanks 1989) indicates that the position of the early Holocene sea level during 9880 cal yr b.p. was at a depth of about 33 m below the present-day sea level. This indicates that a post depositional subsidence has taken place at the study site. The core also records at a depth of 22 m the occurrence of a mangrove stump (cf. Heritiera) in situ, indicative of subsidence. Geological, geomorphological and sedimentological studies also indicate neotectonic activities during the Holocene in these areas (Stanley and Hait 2000).
Early-mid Holocene delta progradation
An abrupt environmental change seems to have occurred after the deposition of Zone P III and as a result the mangrove was rapidly replaced by a fluvial environment (Fig. 5d). The sediment of this phase is characterised by medium to fine grained yellowish sand and contains very few or no palynomorphs. About 15 m of these sediments were deposited, probably by fluvial activities, during a period of about 2800 years between 7560 and 4800 cal yr b.p. (Figs. 2, 3, 4, 5). This intense fluvial activity together with the deceleration of the sea level rise was responsible for vertical accretion of sediments above the tidal limit and hence the destruction of the mangrove habitat. The consequence was the disappearance of mangroves at the core site. This thick layer of sediments indicates large river inputs. A change in climate and eventual increase in rainfall in the upper reaches of the river system during this period generated a huge amount of sediment that was eventually transferred to this area by the Ganga River (Goodbred 2003) and indirectly influenced the mangrove development. This process continued until about 4800 cal yr b.p. The provenance study also indicates the presence of sediment input from the Himalaya via the river Hooghly, a distributory of the large Ganga river system, which is still active today (Chanda et al. 1996).
Similar types of mid Holocene coastal progradation are reported from other areas of the world, namely in northern Australia (Woodroffe et al. 1985), in the Atralto delta in Colombia (West 1956), in the Nile delta in Egypt (Stanley and Warne 1993, 1994), in the Philippines (Berdin et al. 2003), Indonesia (Atmadja and Soerojo 1994; Yulianto et al. 2004), Bangladesh (Umitsu 1993; Islam and Tooley 1999), Puerto Rico (Lugo and Cintron 1975) and in the Mississippi delta in USA (Coleman and Roberts 1988). This suggests a world-wide phenomenon due to environmental changes caused by a combination of the factors of regional climate and local tectonics.
Late Holocene mangrove dynamics
Pollen Zone P IV indicates the return of an intertidal habitat (Fig. 5e). The age of this late Holocene period is uncertain because of the limited available radiocarbon dates. However, the tidal intensity and frequency were low as is evidenced by Excoecaria and Phoenix which grow at the upper limit of the intertidal zone. This zone is also characterised by a higher representation of Acrostichum and the hypersaline herb Suaeda.
The upper part of the section is disturbed and barren of pollen (Figs. 2, 3, 4). This might be related to anthropogenic activities such as farming, aquaculture and settlement during the last century (Naskar 1988; Hussain and Acharya 1994; Chaudhuri and Choudhury 1994; Guha Bakshi and Naskar 1994; Stanley and Hait 2000).
Currently this ecosystem is under stress because of land reclamation and a decreased fresh water supply (Milliman et al. 1989; Sanyal 1990; Nazrul-Islam 1993; Alam 1996; Blasco et al. 1996; Allison 1998; Stanley and Hait 2000). This is also shown by this palynological study. Heritiera and Nypa are two important mangrove plants that prefer to grow in areas that receive a sufficient amount of fresh water (Sanyal 1990, 2001; Chaudhuri and Choudhury 1994; Naskar and Mandal 1999). The presence of both Heritiera and Nypa indicates that there was a balance between fresh water and salt water in the areas until the late Holocene. However, in recent times the balance has been altered because of a lower supply of freshwater to the system, resulting in a poor population growth and density of Heritiera and Nypa in the modern vegetation distribution of the Indian Sundarban areas. The migration of outer estuarine mangrove species further inland and further development of adaptive morphological features by some species are also indications of environmental change. The changes in the species composition and morpho-anatomical features are mainly because of the change in salinity (Sanyal and Ball 1986; Sanyal 2001).
Conclusions
-
1.
Palynological analysis of a radiocarbon-dated 50 m deep core provided the basis for reconstruction of the history of the vegetation and coastal environment in the western Ganga–Brahmaputra Delta.
-
2.
A diverse mangrove forest existed at the study site during the early Holocene at about 9880 cal yr b.p.
-
3.
This early Holocene mangrove forest disappeared due to the changes in the sea level (transgression) after 9240 cal yr b.p. and reappeared as a result of the stabilization of the sea level at the study site around 7560 cal yr b.p. and its less rapid increase from about 5000 cal yr b.p. onwards.
-
4.
The loss of the intertidal habitat, because of the accumulation of a huge quantity of sediment from the sea level regression, accompanied by a high rate of fluvial sedimentation, triggered the disappearance of the mangrove from the site between 7560 and 4800 cal yr b.p. The intertidal habitat reappeared again after 4800 cal yr b.p. at the study site as is evidenced by the pollen record (Zone P IV).
-
5.
Currently, anthropogenic factors prevail over natural factors and the mangrove forest is being altered because of large-scale land reclamation and reduced fresh water inflow into the system.
References
Alam M (1996) Subsidence of the Ganges–Brahmaputra delta of Bangladesh and associated drainage, sedimentation and salinity. In: Milliman JD, Haq BU (eds) Sea level rise and coastal subsidence. Kluwer, Dordrecht, pp 169–192
Allison MA (1998) Geologic framework and environmental status of the Ganges–Brahmaputra delta. J Coastal Res 14:826–836
Atmadja WS, Soerojo A (1994) Mangrove status of Indonesia. In: Wilkinson C, Sudara S, Ming CL (eds) Proceedings third ASEAN––Australian symposium on living coastal resources. Status reviews, vol 1. Australian Institute of Marine Science, Townsville, pp 210–207
Barui NC, Chanda S (1992) Late Quaternary pollen analysis in relation to paleoecology, biostratigraphy and dating of Calcutta peat. Proc Indian Natl Sci Acad B58(4):191–200
Behling H, Cohen MCL, Lara RJ (2001) Studies on Holocene mangrove ecosystem dynamics of the Braganca peninsulain north-eastern Para, Brazil. Palaeogeogr Palaeoclimatol Palaeoecol 167:225–242
Behling H, Cohen MCL, Lara RJ (2004) Late Holocene mangrove dynamics of Marajo island in Amazonia, northern Brazil. Veget Hist Archaeobot 13:73–80
Berdin RD, Siringan FP, Maeda Y (2003) Holocene relative sea-level changes and mangrove response in southwest Bohol, Philipines. J Coastal Res 19:304–313
Blasco F, Saenger P, Janodet E (1996) Mangroves as indicator of coastal change. Catena 27:167–178
Broadus JM (1996) Economising human responses to subsidence and rising sea level. In: Milliman JD, Haq BU (eds) Sea level rise and coastal subsidence: causes, consequences and strategies. Kluwer, Dordrecht, pp 313–325
Bunt JS (1996) Mangrove zonation: an examination of data from seventeen riverine estuaries in tropical Australia. Ann Bot 78:333–341
Campo EV, Bengo MD (2004) Marine palynology in recent sediments of Cameroon. Marine Geol 208:315–330
Caratini C, Bentaleb I, Fontugne M, Morzadec-Kerfourn MT, Pascal JP, Tissot C (1994) A less humid climate since ca. 3500 year b.p. from marine cores off Karwar, western India. Palaeogeogr Palaeoclimatol Palaeoecol 109:371–384
Chanda S (1977) An eco-floristic survey of the mangroves of Sundarbans, West Bengal, India. Trans Bose Res Inst 40:5–14
Chanda S, Hait AK (1996) Aspects and appraisal of late Quaternary vegetation of lower Bengal Basin. Palaeobotanist 45:117–124
Chanda S, Saha AK, Roy A, Hait AK, Das H, Ghosh S, Chakraborti S (1996) Paleoclimate and paleoenvironment of the Bengal Basin during the Holocene. Center for study of man and environment (unpublished report)
Chappel J, Shackleton NJ (1986) Oxygen isotope and sea level. Nature 324:137–140
Chaudhuri AB, Choudhury A (1994) Mangroves of the Sundarbans, IUCN––The World Conservation Union, vol 1, India, Bangkok, Thailand
Coleman JM, Roberts HH (1988) Sedimentary development of the Louisiana continental shelf related to sea level cycles. Part I––sedimentary sequences. Geo Mar Lett 8:63–108
Delft Hydraulics (1989) Criteria for assessing vulnerability to sea-level rise. a global inventory to high risk areas, United Nations environment programme, Delft
Duke NC, Ball MC, Ellison JC (1998) Factors influencing biodiversity and distributional gradients in mangroves. Glob Ecol Biogeogr Lett 7:27–47
Ellison JC (1994) Climate change and sea level rise impacts on mangrove ecosystems. In: Pernetta J, Leemans R, Elder D, Humphrey S (eds) Impacts of climate change on ecosystems and species: marine and coastal ecosystems. International union for conservation of nature and natural resources, Gland, pp 45–57
Ellison JC (2005) Holocene palynology and sea-level change in two estuaries in southern Irian Jaya. Palaeogeogr Palaeoclimatol Palaeoecol 220:291–309
Erdtman G (1969) Handbook of palynology, Munksgaard, Copenhagen
Fairbanks RG (1989) A 17,000 year glacio-eustatic sea-level record: influence of glacial melting rates on the younger Dryas event and deep ocean circulation. Nature 342:637–642
Faegri K, Iversen J (1989) Textbook of pollen analysis. Wiley, Chichester
Fleming K, Johnston P, Zwartz D, Yokoyama Y, Lambeck K, Chappell J (1998) Refining the eustatic sea-level curve since the last glacial maximum using far- and intermediate-field sites. Earth Planet Sci Lett 163:327–342
Goodbred SL Jr (2003) Response of the Ganges dispersal system to climate change: a source-to-sink view since the last interstade. Sed Geol 162:83–104
Grimm EC (1987) CONISS: a FORTRAN 77 program for stratigraphically constrained cluster analysis by the method of the incremental sum of squares. Comput Geosci 13:13–35
Grindrod J, Moss P, van der Kaars S (2002) Late Quaternary mangrove pollen records from continental shelf and ocean cores in the North Australian–Indonesian region. In: Kershaw P, David B, Tapper N, Penny D, Brown J (eds) Bridging Wallace’s line: the environmental and cultural history and dynamics of the SE-Asian–Australian region. Catena, Reiskirchen, pp 119–146
Guha Bakshi DN, Naskar K (1994) Studies of the anthropogenic effects on mangrove ecology of the Sundarbans in West Bengal. In: Ray M (ed) Recent research in ecology. environment and pollution. Today and Tomorrow’s Printers and Publishers, New Delhi, pp 85–90
Gupta HP (1981) Paleoenvironments during Holocene time in Bengal Basin as reflected by palynostratigraphy. Paleobotanist 27:138–160
Hait AK, Das HK, Chakraborti S, Ray AK, Chanda S (1994a) On the Holocene paleoecological changes in coastal Digha––a note based on floral and faunal record. Indian J Earth Sci 21:192–198
Hait AK, Das HK, Ray AK, Chanda S (1994b) On the occurrence of mangrove biota and some faunal remains from the sediments of Haldia. W.B. in relation to its past environment. J Palynol 30:73–78
Hait AK, Das HK, Ghosh S, Ray AK, Saha AK, Chanda S (1996a) New dates of Pleisto-Holocene subcrop samples from south Bengal, India. Indian J Earth Sci 23:79–82
Hait AK, Das HK, Ghosh S, Ray AK, Saha AK, Chanda S (1996b) Evidence of paleoenvironmental variations during late Quaternary of Kolaghat, Midnapur district, West Bengal. Curr Sci 70:1089–1093
Hutchings P, Saenger P (1987) Ecology of mangroves. Queensland University Press, St. Lucia
Hussain Z, Acharya G (1994) Mangroves of the Sundarbans. IUCN–the world conservation union, vol 2, Bangkok, Bangladesh, Thailand
Islam MS (2001) Sea level changes in Bangladesh: The last ten thousand years. Asiatic Society of Bangladesh, Dhaka
Islam MS, Tooley MJ (1999) Coastal and sea level changes during the Holocene in Bangladesh. Quat Int 55:61–75
Jelgersma S (1994) Examples of the geological past: Evolution of the coastal sedimentation sequences during Holocene sea-level rise. In: Pernetta J, Leemans R, Elder D, Humphrey S (eds) Impacts of climate change on ecosystems and species: marine and coastal ecosystems. International union for conservation of nature and natural resources, Gland, pp 45–57
Kamaludin BH (1993) The change of mangrove shorelines in Kuala Kurau, Peninsular Malaysia. Sediment Geol 83:187–197
Lézine A-M (1996) La mangrove ouest-africaine, signal des variations du niveau marin et des conditions régionales du climat au cours de la dernière déglaciation. Bulletin de la Société Géologique de France 6:743–752
Lézine A-M, Saliege J-F, Mathieu R, Tagliatela TL, Mery S, Charpentier V, Cleuziou S (2002) Mangroves of Oman during the late Holocene: climatic implications and impact on human settlements. Veget Hist Archaeobot 11:221–232
Lugo AE, Cintron G (1975) The mangrove forests of Puerto Rico and their management. In: Walsh G, Snedaker SC, Teas H (eds) Proceedings of the international symposium on biology and management of mangroves. University of Florida, Gainesville, pp 825–846
Lugo AE, Snedaker SC (1974) The ecology of mangroves. Ann Rev Ecol Syst 5:39–64
Milliman JD, Broadus JM, Gable F (1989) Environmental and economic implications of rising sea-level and subsiding deltas: the Nile and Bengal examples. Ambio 18:340–345
Naskar KR (1988) Economic potentialities of the tidal mangrove forest of Sundarbans in India. J Indian Soc Coastal Agric Res 6:149–157
Naskar KR, Guha Bakshi DN (1987) Mangrove swamps of the Sundarbans––an ecological perspective. Naya Prokash, Calcutta
Naskar KR, Mandal RN (1999) Ecology and biodiversity of Indian mangroves. Naya Prokash, Calcutta
Nazrul-Islam AKM (1993) Environment and vegetation of mangroves. In: Lieth H, Al Masoom A (eds) Towards the rational use of high salinity tolerant plants. Kluwer, Dordrecht, pp 81–88
Nguyen VL, Ta TKO, Tateishi M (2000) Late Holocene depositional environments and coastal evolution of the Mekong river delta, southern Vietnam. J Asian Earth Sci 18:427–439
Pirazzoli PA (1991) World atlas of Holocene sea-level changes. Elsevier Oceanography Series 58:1–280
Pirazzoli PA (1996) Sea level changes––the last 20,000 years. Wiley, Chichester
Sanyal P, Ball AK (1986) Some observations on abnormal adaptations of mangrove in Indian Sundarbans. J Indian Soc Coastal Agric Res 4:9–15
Sanyal P (1990) Ecological problems of Sundarbans mangrove. In: Proceeding of the workshop on Wasteland Development Programme. Jadavpur University, Calcutta, pp 1–6
Sanyal P (1996) Sundarbans, the largest mangrove diversity on globe. In: Proceeding of the William Roxburgh memorial seminar on Sundarbans Mangals. Calcutta Wildlife Society, Calcutta, pp 11–15
Sanyal P (2001) Effect of global climate change on Sundarban. Banabithi 2:5–8
Sen PK, Banerjee M (1990) Palyno-plankton stratigraphy and environment changes during the Holocene in the Bengal Basin, India. Rev Palaeobot Palynol 65:25–35
Stanley DJ, Warne AG (1993) Nile delta: recent geological evolution and human impact. Science 260:628–634
Stanley DJ, Warne AG (1994) Worldwide initiation of Holocene marine deltas by deceleration of sea level rise. Science 265:228–231
Stanley DJ, Hait AK (2000) Holocene depositional patterns, Neotectonics and Sundarban mangroves in the western Ganges–Brahmaputra delta. J Coastal Res 16:26–39
Southon J, Kashgarian M, Fontugne M, Metivier B, Yim Wyss W-S (2002) Marine reservoir corrections for the Indian Ocean and Southeast Asia. Radiocarbon 44:167–180
Thanikaimoni G (1987) Mangrove palynology. UNDP/UNESCO regional project on training and research on mangrove ecosystems, RAS/79/002, and the French Institute, Pondicherry
Umitsu M (1993) Late quaternary sedimentary environments and landforms in the Ganges delta. Sediment Geol 83:117–186
Weninger B, Jöris O, Danzeglocke U (2004) Cal Pal––the Cologne radiocarbon CALibration and Palaeoclimate research package, http://www.calpal.de
West RC (1956) Mangrove swamps of the pacific coast of Colombia. Ann Am Assoc Geogr 46:98–121
Woodroffe CD (1990) The impact of sea-level rise on mangrove shorelines. Prog Phys Geogr 14:483–520
Woodroffe CD, Thom BC, Chappel J (1985) Development of widespread mangrove swamps in mid-Holocene times in northern Australia. Nature 317:711–713
Yulianto E, Sukapti WS, Rahardjo AT, Noeradi D, Siregar DA, Suparan P, Hirakawa K (2004) Mangrove shoreline responses to Holocene environmental change, Makassar strait, Indonesia. Rev Paleobot Palynol 131:251–268
Yulianto E, Rahardjo WS, Noeradi AT, Siregar DA, Hirakawa K (2005) A Holocene pollen record of vegetation and coastal environmental changes in coastal swamp forest at Batulicin, south Kalimantan, Indonesia. J Asian Earth Sci 25:1–8
Acknowledgments
Appreciation is expressed to S. Chanda, Center for Study of Man and Environment, Principal and Head, Department of Botany, City College of Calcutta; V. Ittekot, Zentrum für Marine Tropenökologie (ZMT); G. Wefer, Geowissenchaften, and B. Zolitschka, Geomorphologie und Polarforschung (GEOPOLAR), both from the University of Bremen, Germany for encouragement to undertake this study. Lydie Dupont and one anonymous reviewer are thanked for their critical comments on an earlier draft of this manuscript. The Department of Science and Technology, Government of India provided the funding for coring. The first author expresses his sincere gratitude to Hanse Wissenschaftskolleg (HWK) for support to complete this study in Germany.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by F. Bittmann.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Hait, A.K., Behling, H. Holocene mangrove and coastal environmental changes in the western Ganga–Brahmaputra Delta, India. Veget Hist Archaeobot 18, 159–169 (2009). https://doi.org/10.1007/s00334-008-0203-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-008-0203-5