Abstract
Objectives
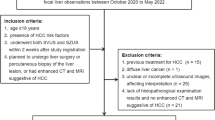
To evaluate the diagnostic accuracy of perfluorobutane contrast-enhanced ultrasonography (CEUS) for hepatocellular carcinoma (HCC) and to explore how accuracy can be improved compared to conventional diagnostic criteria in at-risk patients.
Methods
A total of 123 hepatic nodules (≥ 1 cm) from 123 at-risk patients who underwent perfluorobutane CEUS between 2013 and 2020 at three institutions were retrospectively analyzed. Ninety-three percent of subjects had pathological results, except benign lesions stable in follow-up images. We evaluated presence of arterial phase hyperenhancement (APHE), washout time and degree, and Kupffer phase (KP) defects. KP defects are defined as hypoenhancing lesions relative to the liver in KP. HCC was diagnosed in two ways: (1) Liver Imaging Reporting and Data System (LI-RADS) criteria defined as APHE and late (≥ 60 s)/mild washout, and (2) APHE and Kupffer (AK) criteria defined as APHE and KP defect. We explored grayscale features that cause misdiagnosis of HCC and reflected in the adjustment. Diagnostic performance was compared using McNemar’s test.
Results
There were 77 HCCs, 15 non-HCC malignancies, and 31 benign lesions. An ill-defined margin without hypoechoic halo on grayscale applied as a finding that did not suggest HCC. Regarding diagnosis of HCC, sensitivity of AK criteria (83.1%; 95% confidence interval [CI]: 72.9–90.7%) was higher than that of LI-RADS criteria (75.3%; 95% CI: 64.2–84.4%; p = 0.041). Specificity was 91.3% (95% CI: 79.2–97.6%) in both groups.
Conclusion
On perfluorobutane CEUS, diagnostic criteria for HCC using KP defect with adjustment by grayscale findings had higher diagnostic performance than conventional criteria without losing specificity.
Key Points
• Applying Kupffer phase defect instead of late/mild washout and adjusting with grayscale findings can improve the diagnostic performance of perfluorobutane-enhanced US for HCC.
• Adjustment with ill-defined margins without a hypoechoic halo for features unlikely to be HCC decreases false positives for HCC diagnosis using the perfluorobutane-enhanced US.
• After adjustment with grayscale findings, the sensitivity and accuracy of the APHE and Kupffer criteria were higher than those of the LI-RADS criteria; specificity was 91.3% for both.




Similar content being viewed by others
Abbreviations
- AP:
-
Arterial phase
- APHE:
-
Arterial phase hyperenhancement
- CEUS:
-
Contrast-enhanced ultrasonography
- CI:
-
Confidence interval
- combined HCC-CC:
-
Combined hepatocellular-cholangiocarcinoma
- DN:
-
Dysplastic nodule
- HCC:
-
Hepatocellular carcinoma
- IHCC:
-
Intrahepatic cholangiocarcinomas
- KP:
-
Kupffer phase
- LI-RADS:
-
Liver Imaging Reporting and Data System
- LP:
-
Late phase
- PVP:
-
Portal venous phase
- SD:
-
Standard deviation
- SPIO:
-
Superparamagnetic iron oxide
References
Yanagisawa K, Moriyasu F, Miyahara T, Yuki M, Iijima H (2007) Phagocytosis of ultrasound contrast agent microbubbles by Kupffer cells. Ultrasound Med Biol 33:318–325
Maruyama H, Sekimoto T, Yokosuka O (2016) Role of contrast-enhanced ultrasonography with Sonazoid for hepatocellular carcinoma: evidence from a 10-year experience. J Gastroenterol 51:421–433
Jo PC, Jang HJ, Burns PN, Burak KW, Kim TK, Wilson SR (2017) Integration of contrast-enhanced US into a multimodality approach to imaging of nodules in a cirrhotic liver: how i do it. Radiology 282:317–331
Shunichi S, Hiroko I, Fuminori M, Waki H (2009) Definition of contrast enhancement phases of the liver using a perfluoro-based microbubble agent, perflubutane microbubbles. Ultrasound Med Biol 35:1819–1827
American College of Radiology CEUS LI-RADS® v2017. https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS/CEUS-LI-RADS-v2017. Accessed 13 Aug 2021
Zhai HY, Liang P, Yu J et al (2019) Comparison of Sonazoid and SonoVue in the diagnosis of focal liver lesions: a preliminary study. J Ultrasound Med 38:2417–2425
Kang HJ, Lee JM, Yoon JH, Lee K, Kim H, Han JK (2020) Contrast-enhanced US with sulfur hexafluoride and perfluorobutane for the diagnosis of hepatocellular carcinoma in individuals with high risk. Radiology 297:108–116
Hwang JA, Jeong WK, Min JH, Kim YY, Heo NH, Lim HK (2021) Sonazoid-enhanced ultrasonography: comparison with CT/MRI Liver Imaging Reporting and Data System in patients with suspected hepatocellular carcinoma. Ultrasonography 40:486–498
Korean Liver Cancer Association (KLCA); National Cancer Center (NCC), Goyang, Korea (2019) 2018 Korean Liver Cancer Association-National Cancer Center Korea Practice Guidelines for the Management of Hepatocellular Carcinoma. Korean J Radiol 20:1042–1113
European Association for the Study of the Liver (2018) EASL Clinical Practice Guidelines: management of hepatocellular carcinoma. J Hepatol 69:182–236
Omata M, Cheng AL, Kokudo N et al (2017) Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int 11:317–370
Kudo M (2011) Diagnostic imaging of hepatocellular carcinoma: recent progress. Oncology 81(Suppl 1):73–85
Sugimoto K, Kakegawa T, Takahashi H et al (2020) Usefulness of modified CEUS LI-RADS for the diagnosis of hepatocellular carcinoma using Sonazoid. Diagnostics (Basel) 10:828
Numata K, Luo W, Morimoto M et al (2010) Contrast enhanced ultrasound of hepatocellular carcinoma. World J Radiol 2:68–82
Lee JY, Minami Y, Choi BI et al (2020) The AFSUMB consensus statements and recommendations for the clinical practice of contrast-enhanced ultrasound using Sonazoid. Ultrasonography 39:191–220
Kim TK, Noh SY, Wilson SR et al (2017) Contrast-enhanced ultrasound (CEUS) liver imaging reporting and data system (LI-RADS) 2017 - a review of important differences compared to the CT/MRI system. Clin Mol Hepatol 23:280–289
Hatanaka K, Kudo M, Minami Y et al (2008) Differential diagnosis of hepatic tumors: value of contrast-enhanced harmonic sonography using the newly developed contrast agent, Sonazoid. Intervirology 51(Suppl 1):61–69
Ichikawa T, Saito K, Yoshioka N et al (2010) Detection and characterization of focal liver lesions: a Japanese phase III, multicenter comparison between gadoxetic acid disodium-enhanced magnetic resonance imaging and contrast-enhanced computed tomography predominantly in patients with hepatocellular carcinoma and chronic liver disease. Invest Radiol 45:133–141
Mita K, Kim SR, Kudo M et al (2010) Diagnostic sensitivity of imaging modalities for hepatocellular carcinoma smaller than 2 cm. World J Gastroenterol 16:4187–4192
Choi BI, Takayasu K, Han MC (1993) Small hepatocellular carcinomas and associated nodular lesions of the liver: pathology, pathogenesis, and imaging findings. AJR Am J Roentgenol 160:1177–1187
Wernecke K, Henke L, Vassallo P et al (1992) Pathologic explanation for hypoechoic halo seen on sonograms of malignant liver tumors: an in vitro correlative study. AJR Am J Roentgenol 159:1011–1016
Tada T, Kumada T, Toyoda H et al (2016) Utility of combined gray-scale and perflubutane contrast-enhanced ultrasound for diagnosing early hepatocellular carcinomas: comparison of well differentiated and distinctly nodular types. Hepatol Res 46:1214–1225
Martins-Filho SN, Paiva C, Azevedo RS, Alves VAF (2017) Histological grading of hepatocellular carcinoma-a systematic review of literature. Front Med (Lausanne) 4:193
Kim SH, Lee WJ, Lim HK, Park CK (2009) SPIO-enhanced MRI findings of well-differentiated hepatocellular carcinomas: correlation with MDCT findings. Korean J Radiol 10:112–120
Mandai M, Koda M, Matono T et al (2011) Assessment of hepatocellular carcinoma by contrast-enhanced ultrasound with perfluorobutane microbubbles: comparison with dynamic CT. Br J Radiol 84:499–507
Kudo M, Hatanaka K, Inoue T, Maekawa K (2010) Depiction of portal supply in early hepatocellular carcinoma and dysplastic nodule: value of pure arterial ultrasound imaging in hepatocellular carcinoma. Oncology 78(Suppl 1):60–67
Takahashi M, Maruyama H, Ishibashi H, Yoshikawa M, Yokosuka O (2011) Contrast-enhanced ultrasound with perflubutane microbubble agent: evaluation of differentiation of hepatocellular carcinoma. AJR Am J Roentgenol 196:W123–W131
Inoue T, Kudo M, Maenishi O et al (2009) Value of liver parenchymal phase contrast-enhanced sonography to diagnose premalignant and borderline lesions and overt hepatocellular carcinoma. AJR Am J Roentgenol 192:698–705
Min JH, Lim HK, Lim S et al (2014) Radiofrequency ablation of very-early-stage hepatocellular carcinoma inconspicuous on fusion imaging with B-mode US: value of fusion imaging with contrast-enhanced US. Clin Mol Hepatol 20:61–70
Shin J, Lee S, Bae H et al (2020) Contrast-enhanced ultrasound liver imaging reporting and data system for diagnosing hepatocellular carcinoma: a meta-analysis. Liver Int 40:2345–2352
Kan M, Hiraoka A, Uehara T et al (2010) Evaluation of contrast-enhanced ultrasonography using perfluorobutane (Sonazoid((R))) in patients with small hepatocellular carcinoma: comparison with dynamic computed tomography. Oncol Lett 1:485–488
Hu J, Bhayana D, Burak KW, Wilson SR (2020) Resolution of indeterminate MRI with CEUS in patients at high risk for hepatocellular carcinoma. Abdom Radiol (NY) 45:123–133
Wang DC, Jang HJ, Kim TK (2020) Characterization of indeterminate liver lesions on CT and MRI with contrast-enhanced ultrasound: what is the evidence? AJR Am J Roentgenol 214:1295–1304
Ohama H, Imai Y, Nakashima O et al (2014) Images of Sonazoid-enhanced ultrasonography in multistep hepatocarcinogenesis: comparison with Gd-EOB-DTPA-enhanced MRI. J Gastroenterol 49:1081–1093
Korenaga K, Korenaga M, Furukawa M, Yamasaki T, Sakaida I (2009) Usefulness of Sonazoid contrast-enhanced ultrasonography for hepatocellular carcinoma: comparison with pathological diagnosis and superparamagnetic iron oxide magnetic resonance images. J Gastroenterol 44:733–741
Board WCoTE (2019) Digestive system tumours, WHO Classification of Tumours, 5th ed. International Agency for Research on Cancer, Lyon, France, pp 229-239
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantors of this publication are Woo Kyoung Jeong and Jeong Min Lee.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
A total of 62 study subjects have been previously reported. However, the purpose of the previous research was different from this study.
Thirty-six subjects were reported in a previous paper (Kang et al Contrast-enhanced US with sulfur hexafluoride and perfluorobutane for the diagnosis of hepatocellular carcinoma in individuals with high risk. Radiology 2020; 297:108–116). That study investigated contrast-enhanced US Liver Imaging Reporting and Data System (LI-RADS) v2017 HCC categorization by comparing perfluorobutane-enhanced US and sulfur hexafluoride–enhanced US in participants at high risk for HCC.
Twenty-six subjects were reported in a previous paper (Hwang et al Sonazoid-enhanced ultrasonography: comparison with CT/MRI Liver Imaging Reporting and Data System in patients with suspected hepatocellular carcinoma. Ultrasonography 2021;40:486-498). The aim of that study was to evaluate the association of CEUS features using Sonazoid for liver nodules with LI-RADS categories and to identify the usefulness of Kupffer phase images.
Methodology
• retrospective
• diagnostic or prognostic study
• multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 50 kb)
Rights and permissions
About this article
Cite this article
Hwang, J.A., Jeong, W.K., Kang, HJ. et al. Perfluorobutane-enhanced ultrasonography with a Kupffer phase: improved diagnostic sensitivity for hepatocellular carcinoma. Eur Radiol 32, 8507–8517 (2022). https://doi.org/10.1007/s00330-022-08900-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08900-6