Abstract
Objectives
To investigate the effect of CT image acquisition parameters on the performance of radiomics in classifying benign and malignant pulmonary nodules (PNs) with respect to nodule size.
Methods
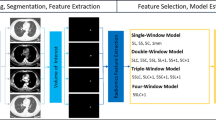
We retrospectively collected CT images of 696 patients with PNs from March 2015 to March 2018. PNs were grouped by nodule diameter: T1a (diameter ≤ 1.0 cm), T1b (1.0 cm < diameter ≤ 2.0 cm), and T1c (2.0 cm < diameter ≤ 3.0 cm). CT images were divided into four settings according to slice-thickness-convolution-kernels: setting 1 (slice thickness/reconstruction type: 1.25 mm sharp), setting 2 (5 mm sharp), setting 3 (5 mm smooth), and random setting. We created twelve groups from two interacting conditions. Each PN was segmented and had 1160 radiomics features extracted. Non-redundant features with high predictive ability in training were selected to build a distinct model under each of the twelve subsets.
Results
The performance (AUCs) on predicting PN malignancy were as follows: T1a group: 0.84, 0.64, 0.68, and 0.68; T1b group: 0.68, 0.74, 0.76, and 0.70; T1c group: 0.66, 0.64, 0.63, and 0.70, for the setting 1, setting 2, setting 3, and random setting, respectively. In the T1a group, the AUC of radiomics model in setting 1 was statistically significantly higher than all others; In the T1b group, AUCs of radiomics models in setting 3 were statistically significantly higher than some; and in the T1c group, there were no statistically significant differences among models.
Conclusions
For PNs less than 1 cm, CT image acquisition parameters have a significant influence on diagnostic performance of radiomics in predicting malignancy, and a model created using images reconstructed with thin section and a sharp kernel algorithm achieved the best performance. For PNs larger than 1 cm, CT reconstruction parameters did not affect diagnostic performance substantially.
Key Points
• CT image acquisition parameters have a significant influence on the diagnostic performance of radiomics in pulmonary nodules less than 1 cm.
• In pulmonary nodules less than 1 cm, a radiomics model created by using images reconstructed with thin section and a sharp kernel algorithm achieved the best diagnostic performance.
• For PNs larger than 1 cm, CT image acquisition parameters do not affect diagnostic performance substantially.


Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the receiver operating curve
- CCC:
-
Concordance correlation coefficient
- CT:
-
Computed tomography
- IFS:
-
Incremental forward search
- IPN:
-
Indeterminate pulmonary nodules
- LDCT:
-
Low-dose computed tomography
- mRMR:
-
Minimum redundancy maximum relevance
- NLST:
-
National Lung Screening Trial
- PN:
-
Pulmonary nodules
- RF:
-
Random forest
References
National Lung Screening Trial Research Team (2013) Church TR, Black WC, et al Results of initial low-dose computed tomographic screening for lung cancer. N Engl J Med 368(21):1980–1991. https://doi.org/10.1056/NEJMoa1209120
Paul R, Hawkins SH, Schabath MB, Gillies RJ, Hall LO, Goldgof DB (2018) Predicting malignant nodules by fusing deep features with classical radiomics features. J Med Imaging (Bellingham) 5(1):011021. https://doi.org/10.1117/1.JMI.5.1.011021
Patz EF Jr, Pinsky P, Gatsonis C et al (2014) Overdiagnosis in low-dose computed tomography screening for lung cancer [published correction appears in JAMA Intern Med. 2014 May;174(5):828]. JAMA Intern Med 174(2):269–274. https://doi.org/10.1001/jamainternmed.2013.12738
Aberle DR, DeMello S, Berg CD et al (2013) Results of the two incidence screenings in the National Lung Screening Trial. N Engl J Med 369(10):920–931. https://doi.org/10.1056/NEJMoa1208962
Takiguchi Y, Sekine I, Iwasawa S (2013) Overdiagnosis in lung cancer screening with low-dose computed tomography. J Thorac Oncol 8(11):e101–e102. https://doi.org/10.1097/JTO.0b013e3182a476d4
Kim H, Park CM, Goo JM, Wildberger JE, Kauczor HU (2015) Quantitative computed tomography imaging biomarkers in the diagnosis and management of lung cancer. Invest Radiol 50(9):571–583. https://doi.org/10.1097/RLI.0000000000000152
Birchard KR, Hoang JK, Herndon JE Jr, Patz EF Jr (2009) Early changes in tumor size in patients treated for advanced stage nonsmall cell lung cancer do not correlate with survival. Cancer 115(3):581–586. https://doi.org/10.1002/cncr.24060
Veronesi G, Maisonneuve P, Bellomi M et al (2012) Estimating overdiagnosis in low-dose computed tomography screening for lung cancer: a cohort study. Ann Intern Med 157(11):776–784. https://doi.org/10.7326/0003-4819-157-11-201212040-00005
Bach PB, Mirkin JN, Oliver TK et al (2012) Benefits and harms of CT screening for lung cancer: a systematic review [published correction appears in JAMA. 2012 Oct 3;308(13):1324] [published correction appears in JAMA. 2013 Jun 5;309(21):2212]. JAMA 307(22):2418–2429. https://doi.org/10.1001/jama.2012.5521
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48(4):441–446. https://doi.org/10.1016/j.ejca.2011.11.036
Kumar V, Gu Y, Basu S et al (2012) Radiomics: the process and the challenges. Magn Reson Imaging 30(9):1234–1248. https://doi.org/10.1016/j.mri.2012.06.010
Chen CH, Chang CK, Tu CY et al (2018) Radiomic features analysis in computed tomography images of lung nodule classification. PLoS One 13(2):e0192002. https://doi.org/10.1371/journal.pone.0192002
Ma J, Zhou Z, Ren Y et al (2017) Computerized detection of lung nodules through radiomics. Med Phys 44(8):4148–4158. https://doi.org/10.1002/mp.12331
Rizzo S, Botta F, Raimondi S et al (2018) Radiomics: the facts and the challenges of image analysis. Eur Radiol Exp 2(1):36. https://doi.org/10.1186/s41747-018-0068-z
Xu Y, Lu L, E LN et al (2019) Application of radiomics in predicting the malignancy of pulmonary nodules in different sizes. AJR Am J Roentgenol 213(6):1213–1220. https://doi.org/10.2214/AJR.19.21490
Ma J, Wang Q, Ren Y et al (2016) Automatic lung nodule classification with radiomics approach. Proc Proc SPIE 9879. 117/12.2220768
Hawkins S, Wang H, Liu Y et al (2016) Predicting malignant nodules from screening CT scans [published correction appears in J Thorac Oncol. 2018 Feb;13(2):280–281]. J Thorac Oncol 11(12):2120–2128. https://doi.org/10.1016/j.jtho.2016.07.002
Alahmari SS, Cherezov D, Goldgof D, Hall L, Gillies RJ, Schabath MB (2018) Delta radiomics improves pulmonary nodule malignancy prediction in lung cancer screening. IEEE Access 6:77796–77806. https://doi.org/10.1109/ACCESS.2018.2884126
Khawaja A, Bartholmai BJ, Rajagopalan S et al (2020) Do we need to see to believe?-radiomics for lung nodule classification and lung cancer risk stratification. J Thorac Dis 12(6):3303–3316. https://doi.org/10.21037/jtd.2020.03.105
E L, Lu L, Li L, Yang H, Schwartz LH, Zhao B (2019) Radiomics for classification of lung cancer histological subtypes based on nonenhanced computed tomography. Acad Radiol 26(9):1245–1252. https://doi.org/10.1016/j.acra.2018.10.013
E L, Lu L, Li L, Yang H, Schwartz LH, Zhao B (2019) Radiomics for classifying histological subtypes of lung cancer based on multiphasic contrast-enhanced computed tomography. J Comput Assist Tomogr 43(2):300–306. https://doi.org/10.1097/RCT.0000000000000836
Patil R, Mahadevaiah G, Dekker A (2016) An approach toward automatic classification of tumor histopathology of non-small cell lung cancer based on radiomic features. Tomography 2(4):374–377. https://doi.org/10.18383/j.tom.2016.00244
Trebeschi S, Drago SG, Birkbak NJ et al (2019) Predicting response to cancer immunotherapy using noninvasive radiomic biomarkers. Ann Oncol 30(6):998–1004. https://doi.org/10.1093/annonc/mdz108
Shi L, He Y, Yuan Z et al (2018) Radiomics for response and outcome assessment for non-small cell lung cancer. Technol Cancer Res Treat 17:1533033818782788. https://doi.org/10.1177/1533033818782788
Li H, Galperin-Aizenberg M, Pryma D, Simone CB 2nd, Fan Y (2018) Unsupervised machine learning of radiomic features for predicting treatment response and overall survival of early stage non-small cell lung cancer patients treated with stereotactic body radiation therapy. Radiother Oncol 129(2):218–226. https://doi.org/10.1016/j.radonc.2018.06.025
Lee H, Chen Y-PP (2015) Image Based Computer Aided Diagnosis System for Cancer Detection. Expert Syst Appl. 42(12):5356–5365. https://doi.org/10.5555/2781921.2782466
Zhao B, Tan Y, Tsai WY et al (2016) Reproducibility of radiomics for deciphering tumor phenotype with imaging. Sci Rep 6:23428. https://doi.org/10.1038/srep23428
Solomon J, Mileto A, Nelson RC, Roy Choudhury K, Samei E (2016) Quantitative features of liver lesions, lung nodules, and renal stones at multi-detector row CT examinations: dependency on radiation dose and reconstruction algorithm. Radiology 279(1):185–194. https://doi.org/10.1148/radiol.2015150892
Meyer M, Ronald J, Vernuccio F et al (2019) Reproducibility of CT radiomic features within the same patient: influence of radiation dose and CT reconstruction settings. Radiology 293(3):583–591. https://doi.org/10.1148/radiol.2019190928
Berenguer R, Pastor-Juan MDR, Canales-Vázquez J et al (2018) Radiomics of CT features may be nonreproducible and redundant: influence of CT acquisition parameters. Radiology 288(2):407–415. https://doi.org/10.1148/radiol.2018172361
Mackin D, Fave X, Zhang L et al (2015) Measuring computed tomography scanner variability of radiomics features. Invest Radiol 50(11):757–765. https://doi.org/10.1097/RLI.0000000000000180
Zhao B, Tan Y, Tsai WY, Schwartz LH, Lu L (2014) Exploring variability in CT characterization of tumors: a preliminary phantom study [published correction appears in Transl Oncol. 2014 Oct;7(5):656] [published correction appears in Transl Oncol. 2014 Oct;7(5):656]. Transl Oncol 7(1):88–93. https://doi.org/10.1593/tlo.13865
Shafiq-Ul-Hassan M, Latifi K, Zhang G, Ullah G, Gillies R, Moros E (2018) Voxel size and gray level normalization of CT radiomic features in lung cancer. Sci Rep. 8(1):10545. https://doi.org/10.1038/s41598-018-28895-9
Escudero Sanchez L, Rundo L, Gill AB, Hoare M, Mendes Serrao E, Sala E (2021) Robustness of radiomic features in CT images with different slice thickness, comparing liver tumour and muscle. Sci Rep. 11(1):8262. https://doi.org/10.1038/s41598-021-87598-w
Shafiq-Ul-Hassan M, Zhang GG, Latifi K et al (2017) Intrinsic dependencies of CT radiomic features on voxel size and number of gray levels. Med Phys 44(3):1050–1062. https://doi.org/10.1002/mp.12123
He L, Huang Y, Ma Z, Liang C, Liang C, Liu Z (2016) Effects of contrast-enhancement, reconstruction slice thickness and convolution kernel on the diagnostic performance of radiomics signature in solitary pulmonary nodule. Sci Rep 6:34921. https://doi.org/10.1038/srep34921
Li Y, Lu L, Xiao M et al (2018) CT Slice thickness and convolution kernel affect performance of a radiomic model for predicting EGFR status in non-small cell lung cancer: a preliminary study. Sci Rep 8(1):17913. https://doi.org/10.1038/s41598-018-36421-0
Zhao B (2011) Understanding sources of variation to improve the reproducibility of radiomics. Front Oncol 11:633176. https://doi.org/10.3389/fonc.2021.633176
Felix A, Oliveira M, Machado A (2016) Using 3D texture and margin sharpness features on classification of small pulmonary nodules. 29th SIBGRAPI Conf Graph Patterns Images. p. 394–400. https://doi.org/10.1109/SIBGRAPI.2016.061
Chen C, Chang C, Tu C, Liao W, Wu B (2018) Radiomic features analysis in computed tomography images of lung nodule classification. PLoS One 13:e0192002. https://doi.org/10.1371/journal.pone.0192002
Lee SH, Cho HH, Lee HY, Park H (2019) Clinical impact of variability on CT radiomics and suggestions for suitable feature selection: a focus on lung cancer. Cancer Imaging 19(1):54. https://doi.org/10.1186/s40644-019-0239-z
Raptis CA, Robb CL, Bhalla S (2018) Tumor staging of lung cancer: essential concepts for the radiologist. Radiol Clin North Am 56(3):391–398. https://doi.org/10.1016/j.rcl.2018.01.006
Yang H, Schwartz LH, Zhao B (2016) A response assessment platform for development and validation of imaging biomarkers in oncology. Tomography 2(4):406–410. https://doi.org/10.18383/j.tom.2016.00223
Tan Y, Schwartz LH, Zhao B (2013) Segmentation of lung lesions on CT scans using watershed, active contours, and Markov random field. Med Phys 40(4):043502. https://doi.org/10.1118/1.4793409
Lu L, Wang D, Wang L et al (2020) A quantitative imaging biomarker for predicting disease-free-survival-associated histologic subgroups in lung adenocarcinoma [published online ahead of print, 2020 Feb 21]. Eur Radiol. https://doi.org/10.1007/s00330-020-06663-6
Zhao B, James LP, Moskowitz CS et al (2009) Evaluating variability in tumor measurements from same-day repeat CT scans of patients with non-small cell lung cancer. Radiology 252:263–272
Lin LI (1989) A concordance correlation coefficient to evaluate reproducibility. Biometrics 45:255–268. https://doi.org/10.2307/2532051
Lu L, Wang D, Wang L et al (2020) A quantitative imaging biomarker for predicting disease-free-survival-associated histologic subgroups in lung adenocarcinoma [published correction appears in Eur Radiol. 2020 Jul 22]. Eur Radiol 30(7):3614–3623. https://doi.org/10.1007/s00330-020-06663-6
Peng H, Long F, Ding C (2005) Feature selection based on mutual information: criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans Pattern Anal Mach Intell 27:1226–1238. https://doi.org/10.1109/TPAMI.2005.159
Breiman L (2001) Random forests. Mach Learn 45:5–32. https://doi.org/10.1023/A:1010933404324
Chawla NV, Bowyer KW, Hall LO, Kegelmeyer WP (2002) SMOTE: synthetic minority over-sampling technique. J Art Intell Res 16:321–357. https://doi.org/10.1613/jair.953
Aerts HJ, Velazquez ER, Leijenaar RT et al (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach [published correction appears in Nat Commun. 2014;5:4644. Cavalho, Sara [corrected to Carvalho, Sara]]. Nat Commun 5:4006. Published 2014 Jun 3. https://doi.org/10.1038/ncomms5006
Youden WJ (1950) Index for rating diagnostic tests. Cancer 3(1):32–35. https://doi.org/10.1002/1097-0142(1950)3:1<32::aid-cncr2820030106>3.0.co;2-3
Humphrey LL, Deffebach M, Pappas M et al (2013) Screening for lung cancer with low-dose computed tomography: a systematic review to update the US Preventive services task force recommendation. Ann Intern Med 159(6):411–420. https://doi.org/10.7326/0003-4819-159-6-201309170-00690
Callister ME, Baldwin DR, Akram AR et al (2015) British Thoracic Society guidelines for the investigation and management of pulmonary nodules [published correction appears in Thorax. 2015 Dec;70(12):1188]. Thorax 70 Suppl 2:ii1-ii54. https://doi.org/10.1136/thoraxjnl-2015-207168
MacMahon H, Naidich DP, Goo JM et al (2017) Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology 284(1):228–243. https://doi.org/10.1148/radiol.2017161659
Scrivener M, de Jong EEC, van Timmeren JE, Pieters T, Ghaye B, Geets X. Radiomics applied to lung cancer: a review. Transl Cancer Res Vol 5, No 4 (August 2016) Transl Cancer Res (Focused Issue Radiomics Radiat Oncol. Published online 2016. http://tcr.amegroups.com/article/view/8536. https://doi.org/10.21037/tcr.2016.06.18
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14(12):749–762. https://doi.org/10.1038/nrclinonc.2017.141
Tan Y, Guo P, Mann H et al (2012) Assessing the effect of CT slice interval on unidimensional, bidimensional and volumetric measurements of solid tumours. Cancer Imaging. 12(3):497–505. https://doi.org/10.1102/1470-7330.2012.0046
Zhao B, Tan Y, Bell DJ et al (2013) Exploring intra- and inter-reader variability in uni-dimensional, bi-dimensional, and volumetric measurements of solid tumors on CT scans reconstructed at different slice intervals. Eur J Radiol 82(6):959–968. https://doi.org/10.1016/j.ejrad.2013.02.018
Barrett JF, Keat N (2004) Artifacts in CT: recognition and avoidance. Radiographics 24(6):1679–1691. https://doi.org/10.1148/rg.246045065
Park S, Lee SM, Do KH et al (2019) Deep learning algorithm for reducing CT slice thickness: effect on reproducibility of radiomic features in lung cancer. Korean J Radiol 20(10):1431–1440. https://doi.org/10.3348/kjr.2019.0212
Choe J, Lee SM, Do KH et al (2019) Deep learning-based image conversion of CT reconstruction kernels improves radiomics reproducibility for pulmonary nodules or masses. Radiology 292(2):365–373. https://doi.org/10.1148/radiol.2019181960
Acknowledgements
This study was supported by the Beijing Natural Science Foundation, China [Grant No. 7182040], and in part by Grant U01 CA225431 from the National Cancer Institute (NCI). The content is solely the responsibility of the authors and does not necessarily represent the funding sources.
Funding
This study was supported by the Beijing Natural Science Foundation, China [Grant No. 7182040], and in part by Grant U01 CA225431 from the National Cancer Institute (NCI). The content is solely the responsibility of the authors and does not necessarily represent the funding sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Binsheng Zhao.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
Several authors have significant statistical and machine learning expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained. The protocols in this study were approved by the Institutional Review Board of Beijing Friendship Hospital, Capital Medical University (Beijing, China) (2018-P2-100-01) and waived the requirement for informed consent because we retrospectively collected the patient data.
Study subjects or cohorts overlap
The content of this manuscript has been presented in part at the IASLC 2019 World Conference on Lung Cancer, Xu Y, Lu L, Lian W, Schwartz L, Yang Z, Zhao B. P2.11-30 Effects of the size of nodules, reconstruction slice thickness and convolution kernel on radiomics model in classifying pulmonary nodules. J Thorac Oncol. 2019;14(10, Supplement): S804-S805. https://doi.org/10.1016/j.jtho.2019.08.1730. Pars of the data have been used in the study published in AJR as “Xu Y, Lu L, E LN, et al Application of radiomics in predicting the malignancy of pulmonary nodules in different sizes. AJR Am J Roentgenol. 2019;213(6):1213-1220. doi:10.2214/AJR.19.21490.”
Methodology
• Retrospective.
• case-controlled study.
•performed at one institution.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, Y., Lu, L., Sun, S.H. et al. Effect of CT image acquisition parameters on diagnostic performance of radiomics in predicting malignancy of pulmonary nodules of different sizes. Eur Radiol 32, 1517–1527 (2022). https://doi.org/10.1007/s00330-021-08274-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08274-1