Abstract
Objectives
Radiomic involves testing the associations of a large number of quantitative imaging features with clinical characteristics. Our aim was to extract a radiomic signature from axial T2-weighted (T2-W) magnetic resonance imaging (MRI) of the whole prostate able to predict oncological and radiological scores in prostate cancer (PCa).
Methods
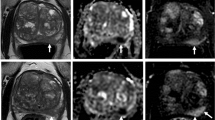
This study included 65 patients with localized PCa treated with radiotherapy (RT) between 2014 and 2018. For each patient, the T2-W MRI images were normalized with the histogram intensity scale standardization method. Features were extracted with the IBEX software. The association of each radiomic feature with risk class, T-stage, Gleason score (GS), extracapsular extension (ECE) score, and Prostate Imaging Reporting and Data System (PI-RADS v2) score was assessed by univariate and multivariate analysis.
Results
Forty-nine out of 65 patients were eligible. Among the 1702 features extracted, 3 to 6 features with the highest predictive power were selected for each outcome. This analysis showed that texture features were the most predictive for GS, PI-RADS v2 score, and risk class; intensity features were highly associated with T-stage, ECE score, and risk class, with areas under the receiver operating characteristic curve (ROC AUC) ranging from 0.74 to 0.94.
Conclusions
MRI-based radiomics is a promising tool for prediction of PCa characteristics. Although a significant association was found between the selected features and all the mentioned clinical/radiological scores, further validations on larger cohorts are needed before these findings can be applied in the clinical practice.
Key Points
• A radiomic model was used to classify PCa aggressiveness.
• Radiomic analysis was performed on T2-W magnetic resonance images of the whole prostate gland.
• The most predictive features belong to the texture (57%) and intensity (43%) domains.



Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AIRC:
-
Associazione Italiana per la Ricerca sul Cancro
- AS:
-
Active surveillance
- AUC:
-
Area under the curve
- CI:
-
Confidence intervals
- DCE:
-
Dynamic contrast-enhanced
- DIL:
-
Dominant intraprostatic lesion
- DWI:
-
Diffusion-weighted imaging
- EBRT:
-
External beam radiotherapy
- ECE:
-
Extracapsular extension
- FDR:
-
False discovery rate
- Gb-scale:
-
Generalized ball scale
- GLCM:
-
Gray-level co-occurrence matrix
- GLRLM:
-
Gray-level run length matrix
- GOH:
-
Gradient orient histogram
- GS:
-
Gleason score
- G-scale:
-
Generalized scale
- IH:
-
Intensity histogram
- HT:
-
Hormone therapy
- ID:
-
Intensity direct
- IMRT-SIB:
-
Intensity-modulated radiation therapy simultaneous integrated boost
- ISUP:
-
International Society of Urological Pathology
- KW:
-
Kruskal-Wallis
- mpMRI:
-
Multiparametric magnetic resonance imaging
- NCCN:
-
National Comprehensive Cancer Network
- NID:
-
Neighborhood intensity difference
- PC:
-
Custom normalization based on healthy tissue intensities in the prostate
- PCa:
-
Prostate cancer
- PI-RADS:
-
Prostate imaging reporting and data system
- PROMIS:
-
Prostate mr imaging study
- PROTECT:
-
Prostate testing for cancer and treatment
- PSA:
-
Prostate-specific antigen
- PTEN:
-
Phosphatase and tensin homolog
- ROC:
-
Receiver operating characteristics
- ROI:
-
Regions of interest
- RP:
-
Prostatectomy
- RT:
-
Radiotherapy
- T2-W:
-
T2-weighted
- VOI:
-
Volume of interest
References
Donovan JL, Hamdy FC, Lane JA et al (2016) Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med 375(15):1425–1437
National Comprehensive Cancer Network (NCCN). Prostate cancer. In: National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology, version 2.2019-April 24, 2019. Available at: https://www.nccn.org/professionals/physician_gls/default.aspx Accessed: May 28, 2019
Morgans (2018) Optimization of risk stratification in localized prostate cancer. J Clin Oncol 36(6):528–533
Ahmed HU, Bosaily AE-S, Brown LC et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389(10071):815–822
Ma S, Xie H, Wang H et al (2019) MRI-based radiomics signature for the preoperative prediction of extracapsular extension of prostate cancer. J Magn Reson Imaging. 50(6):1914–1925
Aerts HJ, Velazquez ER, Leijenaar RT et al (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun:1–8
Nketiah G, Elschot M, Kim E et al (2017) T2-weighted MRI-derived textural features reflect prostate cancer aggressiveness: preliminary results. Eur Radiol 27:3050–3059
Wibmer A, Hricak H, Gondo T et al (2015) Haralick texture analysis of prostate MRI: utility for differentiating non-cancerous prostate from prostate cancer and differentiating prostate cancers with different Gleason scores. Eur Radiol 25:2840–2850
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757
Timon G, Ciardo D, Bazani A et al (2018) Short-term high precision radiotherapy for early prostate cancer with concomitant boost to the dominant lesion: ad interim analysis and preliminary results of Phase II trial AIRC-IG-13218. Br J Radiol. 91(1089):20160725
Zhang L, Fried DV, Fave XJ, Hunter LA, Yang J, Court LE (2015) IBEX: an open infrastructure software platform to facilitate collaborative work in radiomics. Med Phys 42(3):1341–1353
Nyul LG, Udupa JK (1999) On standardizing the MR image intensity scale. Magn Reson Med 42:072–1081
Madabhushi A, Udupa JK, Souza A (2006) Generalized scale: theory, algorithms, and application to image inhomogeneity correction. Comput Vis Image Underst 101(2):100–121
Madabhushi A, Udupa JK (2006) New methods of mr image intensity standardization via generalized scale. Med Phys 33(9):3426–3434
Isaksson LJ, Raimondi S, Botta F et al (2020) Effects of MRI image normalization techniques in prostate cancer radiomics. Phys Med 71:7–13
Perneger TV (1999) Adjusting for multiple testing in studies is less important than other concerns. BMJ 318(7193):1288
Luzzago S, Petralia G, Musi G et al (2019) Multiparametric magnetic resonance imaging second opinion may reduce the number of unnecessary prostate biopsies: time to improve radiologists’ training program? Clin Genitourin Cancer 17(2):88–96
Sonn GA, Fan RE, Ghanouni P et al (2017) Prostate magnetic resonance imaging interpretation varies substantially across radiologists. Eur Urol Focus. 5(4):592–599
Hectors SJ, Cherny M, Yadav KK et al (2019) Radiomics features measured with multiparametric magnetic resonance imaging predict prostate cancer aggressiveness. J Urol. 02(3):498–505
Cuocolo R, Stanzione A, Ponsiglione A et al (2019) Clinically significant prostate cancer detection on MRI: a radiomic shape features study. Eur J Radiol 116:144–149
Schwier M, van Griethuysen J, Vangel MG et al (2019) Repeatability of multiparametric prostate MRI radiomics features. Sci Rep 9(1):9441
Bianchini L, Botta F, Origgi D et al (2020) PETER PHAN: an MRI phantom for the optimisation of radiomic studies of the female pelvis. Phys Med 71:71–81
Fehr D, Veeraraghavan H, Wibmer A et al (2015) Automatic classification of prostate cancer Gleason scores from multiparametric magnetic resonance images. Proc Natl Acad Sci U S A 112(46):E6265–E6273
Chaddad A, Niazi T, Probst S, Bladou F, Anidjar M, Bahoric B (2018) Predicting Gleason score of prostate cancer patients using radiomic analysis. Front Oncol 8
Abdollahi H, Mofid B, Shiri I et al (2019) Machine learning-based radiomic models to predict intensity-modulated radiation therapy response, Gleason score and stage in prostate cancer. Radiol Med. 124(6):555–567
Wang J, Wu CJ, Bao ML, Zhang J, Wang XN, Zhang YD (2017) Machine learning-based analysis of MR radiomics can help to improve the diagnostic performance of PI-RADS v2 in clinically relevant prostate cancer. Eur Radiol 27(10):4082–4090
Algohary A, Viswanath S, Shiradkar R et al (2018) Radiomic features on MRI enable risk categorization of prostate cancer patients on active surveillance: preliminary findings. J Magn Reson Imaging 48(3):818–828
Tanadini-Lang S, Bogowicz M, Veit-Haibach P et al (2018) Exploratory radiomics in computed tomography perfusion of prostate cancer. Anticancer Res 38(2):685–690
Brooks FJ, Grigsby PW (2014) The effect of small tumor volumes on studies of intratumoral heterogeneity of tracer uptake. J Nucl Med 55(1):37–42
Oommen T, Baise LG, Vogel RM (2011) Sampling bias and class imbalance in maximum-likelihood logistic regression. Math Geosci 43(1):99–120
Kasivisvanathan V, Rannikko AS, Borghi M et al (2018) MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 378(19):1767–1777
Gao J, Jiang Q, Zhou B, Chen D (2019) Convolutional neural networks for computer-aided detection or diagnosis in medical image analysis: an overview. Math Biosci Eng 16(6):6536
Acknowledgments
LJI was partially supported by Associazione Italiana per la Ricerca sul Cancro (AIRC), by project IG-13218 “Short-term High Precision Radiotherapy for Early Prostate Cancer With Concomitant Boost on the Dominant Lesion,” registered at ClinicalTrials.gov (NCT01913717), and SGG by project IG-14300 “Carbon ions boost followed by pelvic photon intensity modulated radiotherapy for high-risk prostate cancer,” registered at ClinicalTrials.gov (NCT02672449). MP was supported by a research grant from Accuray Inc. entitled “Data collection and analysis of Tomotherapy and CyberKnife breast clinical studies, breast physics studies and prostate study.” The work was also partially supported by the Italian Ministry of Health with Ricerca Corrente and 5x1000 funds. The sponsors did not play any role in the study design, collection, analysis and interpretation of data, nor in the writing of the manuscript, nor in the decision to submit the manuscript for publication. All authors have participated in (a) conception and design, or analysis and interpretation of the data; (b) drafting the article or revising it critically for important intellectual content; and (c) approval of the final version.
This manuscript has not been submitted to, nor is under review at, another journal or other publishing venue.
Funding
The study was partially funded by Associazione Italiana per la Ricerca sul Cancro (AIRC) and by Accuray Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Barbara Alicja Jereczek-Fossa.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
Two of the authors are biostatisticians.
Informed consent
Written informed consent was obtained from all patients in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Retrospective
• Experimental
• Study performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The first affiliation was the affiliation at the time of data collection for authors Simone Giovanni Gugliandolo, Delia Ciardo and Giulia Riva.
Electronic supplementary material
ESM 1
(DOCX 41 kb)
Rights and permissions
About this article
Cite this article
Gugliandolo, S.G., Pepa, M., Isaksson, L.J. et al. MRI-based radiomics signature for localized prostate cancer: a new clinical tool for cancer aggressiveness prediction? Sub-study of prospective phase II trial on ultra-hypofractionated radiotherapy (AIRC IG-13218). Eur Radiol 31, 716–728 (2021). https://doi.org/10.1007/s00330-020-07105-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07105-z