Abstract
Objectives
To assess the prognostic value of pre-therapeutic computed tomography (CT) attenuation of liver metastases for overall survival (OS) in metastatic colorectal cancer (mCRC).
Methods
In the open-label, randomised, prospective phase-III FIRE-3 trial, patients with histologically confirmed mCRC received fluorouracil (5-FU), leucovorin and irinotecan (FOLFIRI) with either cetuximab or bevacizumab. Participating patients gave written informed consent prior to study entry. In CT at baseline (portal venous phase, slice thickness ≤5 mm), mean attenuation [Hounsfield units (HU)] of liver metastases was retrospectively assessed by semi-automated volumetry. Its prognostic influence on OS was analysed in Kaplan-Meier-analysis and Cox proportional hazard regression and an optimal threshold was determined.
Results
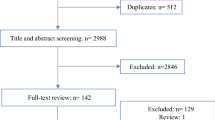
In FIRE-3, 592 patients were enrolled between 2007 and 2012. Among the 347 patients eligible for liver volumetry, median baseline CT attenuation of liver metastases was 59.67 HU [interquartile range (IQR), 49.13, 68.85]. Increased attenuation was associated with longer OS {per 10 HU: hazard ratio (HR), 0.85 [95% confidence interval (CI), 0.78, 0.93], p < 0.001}. The optimised threshold (≥61.62 HU) was a strong predictor for increased OS [median, 21.3 vs 30.6 months; HR, 0.61 (95% CI, 0.47, 0.80), p < 0.001]. Multivariate regression controlling for correlated and further prognostic factors confirmed this [HR, 0.60 (95% CI, 0.45, 0.81), p = 0.001]. Furthermore, mean attenuation ≥61.62 HU was significantly associated with increased early tumour shrinkage (p = 0.002) and increased depth of response (p = 0.012).
Conclusions
Increased mean baseline CT attenuation of liver metastases may identify mCRC patients with prolonged OS and better tumour response.
Key Points
• In colorectal cancer, increased attenuation of liver metastases in baseline computed tomography is a prognostic factor for prolonged OS (p < 0.001).
• A threshold of ≥61.62 HU was determined as optimal cut-off to identify patients with prolonged OS (p < 0.001), early tumour shrinkage (p = 0.002) and increased depth of response (p = 0.012).




Similar content being viewed by others
Abbreviations
- AP:
-
Alkaline phosphatase
- CPH:
-
Cox proportional hazard
- DpR:
-
Depth of response
- ECOG:
-
Eastern cooperative oncology group performance score
- EGFR:
-
Epidermal growth factor receptor
- ETS:
-
Early tumour shrinkage
- KRAS:
-
Kirsten rat sarcoma gene family
- mCRC:
-
Metastatic colorectal cancer
- OS:
-
Overall survival
- RAS:
-
Rat sarcoma gene family
- RECIST:
-
Response evaluation criteria in solid tumours
- VEGF:
-
Vascular endothelial growth factor
- WBC:
-
White blood cell
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65:5–29
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J et al (2013) Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 49:1374–1403
Köhne C-H, Cunningham D, Di Costanzo F et al (2002) Clinical determinants of survival in patients with 5-fluorouracil-based treatment for metastatic colorectal cancer: results of a multivariate analysis of 3825 patients. Ann Oncol 13:308–317
Giessen C, Graeven U, Laubender RP et al (2013) Prognostic factors for 60-day mortality in first-line treatment of metastatic colorectal cancer (mCRC): individual patient analysis of four randomised, controlled trials by the AIO colorectal cancer study group. Ann Oncol 24:3051–3055
Modest DP, Ricard I, Heinemann V et al (2016) Outcome according to KRAS-, NRAS- and BRAF-mutation as well as KRAS mutation variants : pooled analysis of five randomized trials in metastatic colorectal cancer by the AIO colorectal cancer study group. Ann Oncol 27:1746–1753
Douillard J-Y, Oliner KS, Siena S et al (2013) Panitumumab-FOLFOX4 treatment and RAS mutations in colorectal cancer. N Engl J Med 369:1023–1034
Riihimaki M, Hemminki A, Sundquist J, Hemminki K (2016) Patterns of metastasis in colon and rectal cancer. Sci Rep 6:29765
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Chun YS, Vauthey J-N, Boonsirikamchai P et al (2009) Association of computed tomography morphologic criteria with pathologic response and survival in patients treated with bevacizumab for colorectal liver metastases. JAMA 302:2338–2344
Goh V, Padhani AR, Rasheed S (2007) Functional imaging of colorectal cancer angiogenesis. Lancet Oncol 8:245–255
Van Cutsem E, Cervantes A, Nordlinger B, Arnold D, Group EGW (2014) Metastatic colorectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 25:iii1-9
Wu B, Yao Y, Zhang K, Ma X (2017) RAS testing and cetuximab treatment for metastatic colorectal cancer: a cost-effectiveness analysis in a setting with limited health resources. Oncotarget 8(41):71164–71172
Auer RC, White RR, Kemeny NE et al (2010) Predictors of a true complete response among disappearing liver metastases from colorectal cancer after chemotherapy. Cancer 116:1502–1509
Sommer WH, Ceelen F, García-Albéniz X et al (2013) Defining predictors for long progression-free survival after radioembolisation of hepatic metastases of neuroendocrine origin. Eur Radiol 23:3094–3103
Heinemann V, Weikersthal LFV, Decker T et al (2014) FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial. Lancet Oncol 15:1065–1075
Sorich MJ, Wiese M, Rowland A, Kichenadasse G, McKinnon RA, Karapetis C (2014) Extended RAS mutations and anti-EGFR monoclonal antibody survival benefit in metastatic colorectal cancer: a meta-analysis of randomized, controlled trials. Ann Oncol 26:13–21
Heinemann V, Stintzing S, Modest DP, Giessen-Jung C, Michl M, Mansmann UR (2015) Early tumour shrinkage (ETS) and depth of response (DpR) in the treatment of patients with metastatic colorectal cancer (mCRC). Eur J Cancer 51:1927–1936
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
R Core Team (2017) R: a language and environment for statistical computing. Available via: https://www.R-project.org/. Accessed 23 Jan 2017
Huellner MW, Hennedige TP, Winterhalder R et al (2012) Prognostic value of different CT measurements in early therapy response evaluation in patients with metastatic colorectal cancer. Cancer Imaging 12:212–224
Dohan A, Gallix B, Guiu B et al (2017) Survival prediction in patients treated by FOLFIRI and bevacizumab for metastatic colorectal cancer (PRODIGE 9) using contrast-enhanced CT texture analysis (SPECTRA). J Clin Oncol 35:3601–3601
Choi H (2008) Response evaluation of gastrointestinal stromal tumors. Oncologist 13:4–7
Van Der Veldt A, Meijerink M, Van Den Eertwegh A, Haanen J, Boven E (2010) Choi response criteria for early prediction of clinical outcome in patients with metastatic renal cell cancer treated with sunitinib. Br J Cancer 102:803–809
Lencioni R, Llovet JM (2010) Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis 30:52–60
Brufau BP, Cerqueda CS, Villalba LB, Izquierdo RS, González BM, Molina CN (2013) Metastatic renal cell carcinoma: radiologic findings and assessment of response to targeted antiangiogenic therapy by using multidetector CT. Radiographics 33:1691–1716
Aschoff AJ, Catalano C, Kirchin MA, Krix M, Albrecht T (2017) Low radiation dose in computed tomography: the role of iodine. Br J Radiol 90:20170079
Funding
The FIRE-3 study was sponsored by University Hospital Grosshadern, Ludwig-Maximilians-University of Munich and received financial support from Merck Serono GmbH, an affiliate of Merck KGaA, Darmstadt, Germany, Pfizer Pharma GmbH, Karlsruhe, Germany and the German Consortium of Translational Cancer Research, Heidelberg, Germany.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Dr. Wieland Sommer.
Conflict of interest
The authors of this manuscript declare relationships with the following companies:
M.F.F. disclosed no relevant relationships.
V.H. has received honoraria from Merck KGaA, Roche AG, Amgen, Sanofi, SIRTEX and BAXALTA and has received travel support from Merck KGaA, Roche AG, Amgen, SIRTEX and Baxalta and has served on advisory boards for Merck KGaA, Roche AG, Amgen, Sanofi, Lilly, SIRTEX, Böhringer Ingelheim, Baxalta, Taiho and Merrimack.
W.H.S. disclosed no relevant relationships.
J.W.H. has received travel support from Novartis and has served on advisory boards for Roche.
F.S. disclosed no relevant relationships.
N.H. has received financial support from Merck KGaA.
A.B.B. has received financial support from Merck KGaA.
W.G.K. disclosed no relevant relationships.
M.F.R. disclosed no relevant relationships.
J.R. disclosed no relevant relationships.
M.D. disclosed no relevant relationships.
S.S. has received honoraria for talks, advisory boards and travel expenses by Roche, Merck KgaA, Amgen, Bayer, Lilly, Sanofi and Sirtex.
D. P. M. has received honoraria from Merck KGaA, Amgen, Bayer, Servier and Roche and travel support from Merck KGaA, Roche, Amgen, Bayer, Sanofi, Servier.
P.M.K. disclosed no relevant relationships.
F.O.H. disclosed no relevant relationships.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Some FIRE-3 study subjects or cohorts have been previously reported in several journals, e.g. Heinemann et al. [15].
However, results concerning HU-attenuation of liver metastases in this trial have not been published in another journal before.
Methodology
• retrospective
• multicentre study
Electronic supplementary material
ESM 1
(PDF 3.16 mb)
Rights and permissions
About this article
Cite this article
Froelich, M.F., Heinemann, V., Sommer, W.H. et al. CT attenuation of liver metastases before targeted therapy is a prognostic factor of overall survival in colorectal cancer patients. Results from the randomised, open-label FIRE-3/AIO KRK0306 trial. Eur Radiol 28, 5284–5292 (2018). https://doi.org/10.1007/s00330-018-5454-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5454-7