Abstract
Objectives
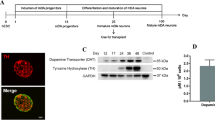
We evaluated the effects of bone marrow-derived mesenchymal stem cells (BMSCs) in a model of Parkinson’s disease (PD) using serial F-18 fluoropropylcarbomethoxyiodophenylnortropane (FP-CIT) PET.
Methods
Hemiparkinsonian rats were treated with intravenously injected BMSCs, and animals without stem cell therapy were used as the controls. Serial FP-CIT PET was performed after therapy. The ratio of FP-CIT uptake in the lesion side to uptake in the normal side was measured. The changes in FP-CIT uptake were also analyzed using SPM. Behavioural and histological changes were observed using the rotational test and tyrosine hydroxylase (TH)-reactive cells.
Results
FP-CIT uptake ratio was significantly different in the BMSCs treated group (n = 28) at each time point. In contrast, there was no difference in the ratio in control rats (n = 25) at any time point. SPM analysis also revealed that dopamine transporter binding activity was enhanced in the right basal ganglia area in only the BMSC therapy group. In addition, rats that received BMSC therapy also exhibited significantly improved rotational behaviour and preservation of TH-positive neurons compared to controls.
Conclusions
The therapeutic effect of intravenously injected BMSCs in a rat model of PD was confirmed by dopamine transporter PET imaging, rotational functional studies, and histopathological evaluation.
Key Points
• Mesenchymal stem cells were intravenously injected to treat the PD rats
• Dopamine transporter binding activity was improved after stem cell therapy
• Stem cell therapy induced functional recovery and preservation of dopaminergic neurons
• The effect of stem cells was confirmed by FP-CIT PET






Similar content being viewed by others
Abbreviations
- PD:
-
Parkinson’s disease
- MSCs:
-
Mesenchymal stem cells
- BMSCs:
-
Bone marrow-derived mesenchymal stem cells
- DAT:
-
Dopamine transporter
- 6-OHDA:
-
6-hydroxydopamine
- FP-CIT:
-
F-18 fluoropropylcarbomethoxyiodophenylnortropane
- SD:
-
Sprague–Dawley
- PBS:
-
Phosphate buffered saline
- FWHM:
-
Full width half maximum
- ROIs:
-
Regions of interest
- SPM:
-
Statistical parametric mapping
- TBS:
-
Tris-buffered saline
- TH:
-
Tyrosine hydroxylase
- F-18 FDOPA:
-
6-[F-18]-fluoro-L-4-dihydroxyphenylalanine
- GDNF:
-
Glial-cell-line-derived neurotrophic factor
- BDNF:
-
Brain-derived neurotrophic factor
- CNTF:
-
Ciliary neurotrophic factor
- ECM:
-
Extracellular matrix proteins
References
Beitz JM (2014) Parkinson’s disease: a review. Front Biosci (Schol Ed) 6:65–74
Toulouse A, Sullivan AM (2008) Progress in Parkinson’s disease-where do we stand? Prog Neurobiol 85:376–392
Glavaski-Joksimovic A, Bohn MC (2013) Mesenchymal stem cells and neuroregeneration in Parkinson’s disease. Exp Neurol 247:25–38
Ali F, Stott SR, Barker RA (2014) Stem cells and the treatment of Parkinson’s disease. Exp Neurol 260:3–11
Yang M, Donaldson AE, Jiang Y, Iacovitti L (2003) Factors influencing the differentiation of dopaminergic traits in transplanted neural stem cells. Cell Mol Neurobiol 23:851–864
Bjorklund LM, Sanchez-Pernaute R, Chung S et al (2002) Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a Parkinson rat model. Proc Natl Acad Sci U S A 99:2344–2349
Bouchez G, Sensebe L, Vourc’h P et al (2008) Partial recovery of dopaminergic pathway after graft of adult mesenchymal stem cells in a rat model of Parkinson’s disease. Neurochem Int 52:1332–1342
Clarkson ED (2001) Fetal tissue transplantation for patients with Parkinson’s disease: a database of published clinical results. Drugs Aging 18:773–785
Wang F, Yasuhara T, Shingo T et al (2010) Intravenous administration of mesenchymal stem cells exerts therapeutic effects on parkinsonian model of rats: focusing on neuroprotective effects of stromal cell-derived factor-1alpha. BMC Neurosci 11:52
Pittenger MF, Mackay AM, Beck SC et al (1999) Multilineage potential of adult human mesenchymal stem cells. Sci 284:143–147
Venkataramana NK, Kumar SK, Balaraju S et al (2010) Open-labeled study of unilateral autologous bone-marrow-derived mesenchymal stem cell transplantation in Parkinson’s disease. Transl Res 155:62–70
Danielyan L, Schafer R, von Ameln-Mayerhofer A et al (2011) Therapeutic efficacy of intranasally delivered mesenchymal stem cells in a rat model of Parkinson disease. Rejuvenation Res 14:3–16
Xiong N, Zhang Z, Huang J et al (2011) VEGF-expressing human umbilical cord mesenchymal stem cells, an improved therapy strategy for Parkinson’s disease. Gene Ther 18:394–402
Scherfler C, Donnemiller E, Schocke M et al (2002) Evaluation of striatal dopamine transporter function in rats by in vivo beta-[123I]CIT pinhole SPECT. Neuroimage 17:128–141
Booij J, Speelman JD, Horstink MW, Wolters EC (2001) The clinical benefit of imaging striatal dopamine transporters with [123I]FP-CIT SPET in differentiating patients with presynaptic parkinsonism from those with other forms of parkinsonism. Eur J Nucl Med 28:266–272
Winogrodzka A, Bergmans P, Booij J, van Royen EA, Janssen AG, Wolters EC (2001) [123I]FP-CIT SPECT is a useful method to monitor the rate of dopaminergic degeneration in early-stage Parkinson’s disease. J Neural Transm 108:1011–1019
Nakano Y, Hirko AC, Smith AD et al (2004) Presynaptic dopaminergic properties of differentiated mouse embryonic stem cells. Neurochem Int 45:1067–1073
Kyono K, Takashima T, Katayama Y et al (2011) Use of [18F]FDOPA-PET for in vivo evaluation of dopaminergic dysfunction in unilaterally 6-OHDA-lesioned rats. EJNMMI Res 1:25
Mahmood A, Lu D, Chopp M (2004) Intravenous administration of marrow stromal cells (MSCs) increases the expression of growth factors in rat brain after traumatic brain injury. J Neurotrauma 21:33–39
Gutierrez-Fernandez M, Rodriguez-Frutos B, Ramos-Cejudo J et al (2013) Effects of intravenous administration of allogenic bone marrow- and adipose tissue-derived mesenchymal stem cells on functional recovery and brain repair markers in experimental ischemic stroke. Stem Cell Res Ther 4:11
Casteels C, Vermaelen P, Nuyts J et al (2006) Construction and evaluation of multitracer small-animal PET probabilistic atlases for voxel-based functional mapping of the rat brain. J Nucl Med 47:1858–1866
Nie B, Chen K, Zhao S et al (2013) A rat brain MRI template with digital stereotaxic atlas of fine anatomical delineations in paxinos space and its automated application in voxel-wise analysis. Hum Brain Mapp 34:1306–1318
Dabbeni-Sala F, Di Santo S, Franceschini D, Skaper SD, Giusti P (2001) Melatonin protects against 6-OHDA-induced neurotoxicity in rats: a role for mitochondrial complex I activity. FASEB J 15:164–170
Nagatsu T, Levitt M, Udenfriend S (1964) Tyrosine hydroxylase. The initial step in norepinephrine biosynthesis. J Biol Chem 239:2910–2917
Alvarez-Fischer D, Blessmann G, Trosowski C et al (2007) Quantitative [(123)I]FP-CIT pinhole SPECT imaging predicts striatal dopamine levels, but not number of nigral neurons in different mouse models of Parkinson’s disease. Neuroimage 38:5–12
Cheng D, Wang Y, Liu X et al (2010) Comparison of 18F PET and 99mTc SPECT imaging in phantoms and in tumored mice. Bioconjug Chem 21:1565–1570
Hutchins GD, Miller MA, Soon VC, Receveur T (2008) Small animal PET imaging. ILAR J 49:54–65
McLarty K, Reilly RM (2007) Molecular imaging as a tool for personalized and targeted anticancer therapy. Clin Pharmacol Ther 81:420–424
Nurmi E, Ruottinen HM, Bergman J et al (2001) Rate of progression in Parkinson’s disease: a 6-[18F]fluoro-L-dopa PET study. Mov Disord 16:608–615
Ungerstedt U (1971) Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiol Scand Suppl 367:69–93
Kawasaki H, Mizuseki K, Nishikawa S et al (2000) Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28:31–40
Brederlau A, Correia AS, Anisimov SV et al (2006) Transplantation of human embryonic stem cell-derived cells to a rat model of Parkinson’s disease: effect of in vitro differentiation on graft survival and teratoma formation. Stem Cells 24:1433–1440
Kim JH, Auerbach JM, Rodriguez-Gomez JA et al (2002) Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson’s disease. Nat 418:50–56
Li Y, Chen J, Wang L, Zhang L, Lu M, Chopp M (2001) Intracerebral transplantation of bone marrow stromal cells in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease. Neurosci Lett 316:67–70
Hellmann MA, Panet H, Barhum Y, Melamed E, Offen D (2006) Increased survival and migration of engrafted mesenchymal bone marrow stem cells in 6-hydroxydopamine-lesioned rodents. Neurosci Lett 395:124–128
Park HJ, Lee PH, Bang OY, Lee G, Ahn YH (2008) Mesenchymal stem cells therapy exerts neuroprotection in a progressive animal model of Parkinson’s disease. J Neurochem 107:141–151
Fu YS, Cheng YC, Lin MY et al (2006) Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: potential therapeutic application for Parkinsonism. Stem Cells 24:115–124
Khoo ML, Tao H, Meedeniya AC, Mackay-Sim A, Ma DD (2011) Transplantation of neuronal-primed human bone marrow mesenchymal stem cells in hemiparkinsonian rodents. PLoS One 6:e19025
Phinney DG, Prockop DJ (2007) Concise review: mesenchymal stem/multipotent stromal cells: the state of transdifferentiation and modes of tissue repair–current views. Stem Cells 25:2896–2902
Blandini F, Cova L, Armentero MT et al (2010) Transplantation of undifferentiated human mesenchymal stem cells protects against 6-hydroxydopamine neurotoxicity in the rat. Cell Transplant 19:203–217
Aizman I, Tate CC, McGrogan M, Case CC (2009) Extracellular matrix produced by bone marrow stromal cells and by their derivative, SB623 cells, supports neural cell growth. J Neurosci Res 87:3198–3206
Kozlowska H, Jablonka J, Janowski M, Jurga M, Kossut M, Domanska-Janik K (2007) Transplantation of a novel human cord blood-derived neural-like stem cell line in a rat model of cortical infarct. Stem Cells Dev 16:481–488
Kraitchman DL, Tatsumi M, Gilson WD et al (2005) Dynamic imaging of allogeneic mesenchymal stem cells trafficking to myocardial infarction. Circ 112:1451–1461
Yoon JK, Park BN, Shim WY, Shin JY, Lee G, Ahn YH (2010) In vivo tracking of 111In-labeled bone marrow mesenchymal stem cells in acute brain trauma model. Nucl Med Biol 37:381–388
Gleave JA, Farncombe TH, Saab C, Doering LC (2011) Correlative single photon emission computed tomography imaging of [123I]altropane binding in the rat model of Parkinson’s. Nucl Med Biol 38:741–749
Acknowledgments
The scientific guarantor of this publication is Joon-Kee Yoon. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. This study has received funding by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST) (NRF-2011-0007935). No complex statistical methods were necessary for this paper.
Ethical Committee
The Care of Experimental Animals Committee of Ajou University School of Medicine approved all experimental procedures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, BN., Kim, JH., Lee, K. et al. Improved dopamine transporter binding activity after bone marrow mesenchymal stem cell transplantation in a rat model of Parkinson’s disease: small animal positron emission tomography study with F-18 FP-CIT. Eur Radiol 25, 1487–1496 (2015). https://doi.org/10.1007/s00330-014-3549-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3549-3