Abstract
Objectives
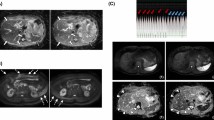
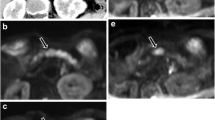
To compare respiratory-triggered, free-breathing, and breath-hold DWI techniques regarding (1) image quality, and (2) signal intensity (SI) and ADC measurements in pancreatic ductal adenocarcinoma (PDAC).
Methods
Fifteen patients with histopathologically proven PDAC underwent DWI prospectively at 1.5 T (b = 0, 50, 300, 600 and 1,000 s/mm2) with the three techniques. Two radiologists, independently and blindly, assigned total image quality scores [sum of rating diffusion images (lesion detection, anatomy, presence of artefacts) and ADC maps (lesion characterisation, overall image quality)] per technique and ranked them. The lesion SI, signal-to-noise ratio, mean ADC and coefficient of variation (CV) were compared.
Results
Total image quality scores for respiratory-triggered, free-breathing and breath-hold techniques were 17.9, 16.5 and 17.1 respectively (respiratory-triggered was significantly higher than free-breathing but not breath-hold). The respiratory-triggered technique had a significantly higher ranking. Lesion SI on all b-values and signal-to-noise ratio on b300 and b600 were significantly higher for the respiratory-triggered technique. For respiratory-triggered, free-breathing and breath-hold techniques the mean ADCs were 1.201, 1.132 and 1.253 × 10-3 mm2/s, and mean CVs were 8.9, 10.8 and 14.1 % respectively (respiratory-triggered and free-breathing techniques had a significantly lower mean CV than the breath-hold technique).
Conclusion
In both analyses, respiratory-triggered DWI showed superiority and seems the optimal DWI technique for demonstrating PDAC.
Key Points
• Diffusion-weighted magnetic resonance imaging is increasingly used to detect pancreatic cancer
• Images are acquired using various breathing techniques and multiple b-values
• Breathing techniques used: respiratory-triggering, free-breathing and breath-hold
• Respiratory-triggering seems the optimal breathing technique for demonstrating pancreatic cancer


Similar content being viewed by others
References
Taouli B, Chouli M, Martin AJ, Qayyum A, Coakley FV, Vilgrain V (2008) Chronic hepatitis: role of diffusion-weighted imaging and diffusion tensor imaging for the diagnosis of liver fibrosis and inflammation. J Magn Reson Imaging 28:89–95
Sandrasegaran K, Akisik FM, Lin C et al (2009) Value of diffusion-weighted MRI for assessing liver fibrosis and cirrhosis. AJR Am J Roentgenol 193:1556–1560
Do RK, Chandarana H, Felker E et al (2010) Diagnosis of liver fibrosis and cirrhosis with diffusion-weighted imaging: value of normalized apparent diffusion coefficient using the spleen as reference organ. AJR Am J Roentgenol 195:671–676
Lewin M, Poujol-Robert A, Boëlle PY et al (2007) Diffusion-weighted magnetic resonance imaging for the assessment of fibrosis in chronic hepatitis C. Hepatology 46:658–665
Akisik MF, Aisen AM, Sandrasegaran K et al (2009) Assessment of chronic pancreatitis: utility of diffusion-weighted MR imaging with secretin enhancement. Radiology 250:103–109
Erturk SM, Ichikawa T, Motosugi U, Sou H, Araki T (2006) Diffusion-weighted MR imaging in the evaluation of pancreatic exocrine function before and after secretin stimulation. Am J Gastroenterol 101:133–136
Kamisawa T, Takuma K, Anjiki H et al (2010) Differentiation of autoimmune pancreatitis from pancreatic cancer by diffusion-weighted MRI. Am J Gastroenterol 105:1870–1875
Bruegel M, Holzapfel K, Gaa J et al (2008) Characterization of focal liver lesions by ADC measurements using a respiratory triggered diffusion-weighted single-shot echoplanar MR imaging technique. Eur Radiol 18:477–485
Taouli B, Vilgrain V, Dumont E, Daire JL, Fan B, Menu Y (2003) Evaluation of liver diffusion isotropy and characterization of focal hepatic lesions with two single-shot echo-planar MR imaging sequences: prospective study in 66 patients. Radiology 226:71–78
Bakir B, Salmaslioğlu A, Poyanli A, Rozanes I, Acunas B (2010) Diffusion weighted MR imaging of pancreatic islet cell tumors. Eur J Radiol 74:214–220
Ichikawa T, Erturk SM, Motosugi U et al (2007) High-b value diffusion-weighted MRI for detecting pancreatic adenocarcinoma: preliminary results. AJR Am J Roentgenol 188:409–414
Kartalis N, Lindholm TL, Aspelin P, Permert J, Albiin N (2009) Diffusion-weighted magnetic resonance imaging of pancreas tumours. Eur Radiol 19:1981–1990
Koh DM, Scurr E, Collins D et al (2007) Predicting response of colorectal hepatic metastasis: value of pretreatment apparent diffusion coefficients. AJR Am J Roentgenol 188:1001–1008
Cui Y, Zhang XP, Sun YS, Tang L, Shen L (2008) Apparent diffusion coefficient: potential imaging biomarker for prediction and early detection of response to chemotherapy in hepatic metastases. Radiology 248:894–900
Mannelli L, Kim S, Hajdu CH, Babb JS, Clark TW, Taouli B (2009) Assessment of tumor necrosis of hepatocellular carcinoma after chemoembolization: diffusion-weighted and contrast-enhanced MRI with histopathologic correlation of the explanted liver. AJR Am J Roentgenol 193:1044–1052
Kamel IR, Liapi E, Reyes DK, Zahurak M, Bluemke DA, Geschwind JF (2009) Unresectable hepatocellular carcinoma: serial early vascular and cellular changes after transarterial chemoembolization as detected with MR imaging. Radiology 250:466–473
Taouli B, Koh DM (2010) Diffusion-weighted MR imaging of the liver. Radiology 254:47–66
Taouli B, Sandberg A, Stemmer A et al (2009) Diffusion-weighted imaging of the liver: comparison of navigator triggered and breathhold acquisitions. J Magn Reson Imaging 30:561–568
Kim SY, Lee SS, Byun JH et al (2010) Malignant hepatic tumors: short-term reproducibility of apparent diffusion coefficients with breath-hold and respiratory-triggered diffusion-weighted MR imaging. Radiology 255:815–823
Kandpal H, Sharma R, Madhusudhan KS, Kapoor KS (2009) Respiratory-triggered versus breath-hold diffusion-weighted MRI of liver lesions: comparison of image quality and apparent diffusion coefficient values. AJR Am J Roentgenol 192:915–922
Kwee TC, Takahara T, Koh DM, Nievelstein RA, Luijten PR (2008) Comparison and reproducibility of ADC measurements in breathhold, respiratory triggered, and free-breathing diffusion-weighted MR imaging of the liver. J Magn Reson Imaging 28:1141–1148
Rosenkrantz AB, Oei M, Babb JS, Niver BE, Taouli B (2011) Diffusion-weighted imaging of the abdomen at 3.0 Tesla: image quality and apparent diffusion coefficient reproducibility compared with 1.5 Tesla. J Magn Reson Imaging 33:128–135
Heverhagen JT (2007) Noise measurement and estimation in MR imaging experiments. Radiology 245:638–639
Padhani AR, Liu G, Koh DM et al (2009) Diffusion-weighted magnetic resonance imaging as a cancer biomarker: consensus and recommendations. Neoplasia 11:102–125
Acknowledgements
The authors would like to thank M. Karlsson and L. Erlandsson Nordin for their technical support.
This work was presented as an oral lecture at ECR 2012 (control no.: 12-P-2599-ECR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kartalis, N., Loizou, L., Edsborg, N. et al. Optimising diffusion-weighted MR imaging for demonstrating pancreatic cancer: a comparison of respiratory-triggered, free-breathing and breath-hold techniques. Eur Radiol 22, 2186–2192 (2012). https://doi.org/10.1007/s00330-012-2469-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-012-2469-3