Abstract
Objective
To develop a visual rating scale for posterior atrophy (PA) assessment and to analyse whether this scale aids in the discrimination between Alzheimer’s disease (AD) and other dementias.
Methods
Magnetic resonance imaging of 118 memory clinic patients were analysed for PA (range 0–3), medial temporal lobe atrophy (MTA) (range 0–4) and global cortical atrophy (range 0–3) by different raters. Weighted-kappas were calculated for inter- and intra-rater agreement. Relationships between PA and MTA with the MMSE and age were estimated with linear-regression analysis.
Results
Intra-rater agreement ranged between 0.93 and 0.95 and inter-rater agreement between 0.65 and 0.84. Mean PA scores were higher in AD compared to controls (1.6 ± 0.9 and 0.6 ± 0.7, p < 0.01), and other dementias (0.8 ± 0.8, p < 0.01). PA was not associated with age compared to MTA (B = 1.1 (0.8) versus B = 3.1 (0.7), p < 0.01)). PA and MTA were independently negatively associated with the MMSE (B = −1.6 (0.5), p < 0.01 versus B = −1.4 (0.5), p < 0.01).
Conclusion
This robust and reproducible scale for PA assessment conveys independent information in a clinical setting and may be useful in the discrimination of AD from other dementias.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD) has been considered clinically homogenous and characterized by progressive memory impairment followed by global cognitive decline [1]. The clinical diagnosis of AD depends largely on the presenting cognitive profile. However, atypical focal presentations of AD may be more common than previously thought, which can make a clinical diagnosis difficult [1–3]. Neuroimaging, particularly magnetic resonance (MR) imaging, has an established role in excluding other, potentially treatable diagnoses, but also increasingly recognized potential to support a clinical diagnosis of AD by identifying certain atrophy patterns [4, 5]. AD is typically associated with medial temporal lobe atrophy (MTA) [6–9]. The presence of MTA on MR imaging improves the discrimination of AD from healthy controls and predicts progression to dementia in patients with mild cognitive impairment (MCI) [9, 10]. Visual MTA rating based on established rating scales, has proven to be useful for a good and reproducible assessment in clinical practice and correlates well with volumetric assessments [11, 12]. However, MTA is also present in other dementias, for example Frontotemporal lobe degeneration (FTLD), vascular dementia (VaD) and dementia with Lewy bodies (DLB) and may been seen in normal aging [13–18]. Furthermore, younger AD patients often show relatively less (or no) MTA at presentation [19].
Occasionally, AD patients present with a striking posterior atrophy pattern- often referred to as posterior cortical atrophy. Benson et al. were the first describing patients with evidence of posterior cerebral involvement and prominent visual problems and suggested possible underlying AD [20]. Early posterior cerebral involvement is emerging as an important aspect of AD, more generally with dysfunction and atrophy of the posterior cingulate gyrus, precuneus and parietal lobes, being perhaps a particular feature of early-onset (<65 years) AD (EOAD) [3, 19, 21–27]. Posterior atrophy (PA) appears to be characteristic of AD in patients with typical and atypical clinical presentations and may assist in the clinical distinction of AD from Frontotemporal lobar degeneration (FTLD) [28, 29]. Moreover, combined with relative sparing of the medial temporal lobe, PA has found to be characteristic for patients with atypical clinical presentations [3, 25, 30].
Limited data is available about the clinical relevance of PA and no visual rating scale is currently available to measure atrophy in the posterior regions, hampering systematic evaluation of PA. The purpose of this study was to develop a robust visual rating scale for PA assessment and evaluate intra- and inter-observer agreement. The second aim was to investigate the clinical relevance of PA, by comparing the degree of PA in AD, controls and other dementias and investigate the relationship with age and the mini-mental state examination (MMSE) [31].
Materials and methods
Patient selection
This study was conducted at our memory clinic and approved by the local institutional review board. Written informed consent was obtained from all participants.
We selected 120 patients from our memory clinic population based on their clinical diagnosis. The selection included 60 AD patients (35 EOAD (onset <65 years) and 25 late-onset AD), 20 age-matched patients with other dementias (10 FTLD and 10 DLB patients) and 40 age-matched patients with subjective memory complaints, without cognitive impairment. The group of other dementias did not include patients with vascular dementia, characterized by severe vascular abnormalities on MR imaging, since this could be of influence on PA measurement. The diagnostic procedure consisted of a standard battery of investigations including a patient and informant-based medical history, physical and neurological examination including the MMSE and multisequence MR imaging. The clinical diagnosis of probable AD, FTLD or DLB was made according to current criteria by a multidisciplinary team, including a neurologist, psychiatrist, neurophysiologist, neuropsychologist and specialized nurse [32–34]. The patients’ diagnosis was established on clinical criteria and not on basis of their imaging findings. Imaging findings did not change a patient’s diagnosis in our study group.
MR imaging
MR imaging was performed using 3.0 T MR (Signa HDxt, General Electric, Milwaukee). Mean time between MR imaging and diagnosis was 0.2 ±1 months. All subjects were examined according to a standard dementia MRI protocol: sagittal T1-weighted 3D fast spoiled gradient echo (FSPGR) sequences (field of view (FOV) = 250 mm; matrix = 256 × 256; 1 mm slices; echo time = 3 ms; repetition time = 7.8 ms; inversion time = 450 ms; one signal acquired), 3D fluid-attenuated inversion recovery (FLAIR) (FOV = 250 mm; matrix = 224 × 224; 1 mm slices; echo time = 125 ms; repetition time = 8,000 ms; inversion time = 2,349 ms; echo train length 230, one signal acquired) and axial fast spin-echo T2/PD sequences (FOV = 250 mm; matrix = 384 × 384; 3 mm slices; echo time = 20/109 ms; repetition time = 8,240 ms; echo train length 24, two signals acquired). Multiplanar (MPR) reconstructions of 3D-T1-weighted sequences were performed in sagittal (5 mm) and oblique-coronal orientations (3 mm slices perpendicular to the long axis of the hippocampus), using a 5 mm section thickness. MPR reconstructions of 3D-FLAIR images were performed in transverse orientation using a 3 mm section thickness.
Posterior rating scale development
To define the areas included in the scale, we evaluated 25 MR examinations from probable AD patients with a wide range of atrophy and determined the most affected posterior regions. Secondly, we assessed these areas in 10 controls, to determine whether these areas were more affected in AD patients and therefore able to represent PA. Finally, taking into account those regions mentioned in literature, the following anatomical regions were selected: the posterior cingulate sulcus, precuneus, parieto-occipital sulcus and the cortex of the parietal lobes. The MR examinations used to determine which posterior regions to rate were not further used in this study, to avoid any circularity.
To assess PA we designed a 4-point rating scale (0 = no atrophy; 1 = mild widening of the sulci without evident volume loss of the gyri; 2 = substantial widening of the sulci and volume loss of the gyri; grade 3 = severe end-stage atrophy). The overall score based on the presence of atrophy in sagittal, axial and coronal orientation, was assessed for left and right separately on sagittal and coronal MPR sections of the T1-weighted sequence and axial MPR sections of the FLAIR sequence.
Characteristics of the posterior rating scale
The following anatomical landmarks were rated in three different orientations:
-
a)
sagittal orientation: widening of the posterior cingulate- and parieto-occipital sulcus, and atrophy of the precuneus on left and right by considering paramedian-sagittal images.
-
b)
axial orientation: widening of the posterior cingulate sulcus and sulcal dilatation in parietal lobes on axial images.
-
c)
coronal orientation: widening of the posterior cingulate sulcus and parietal lobes on coronal images.
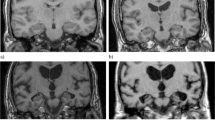
Grade 0 represents a closed posterior cingulate- and parieto-occipital sulcus and closed sulci of the parietal lobes and precuneus. Grade 1 includes a mild widening of the posterior cingulate- and parieto-occipital sulcus, with mild atrophy of the parietal lobes and precuneus. Grade 2 shows substantial widening of the posterior cingulate- and parieto-occipital sulcus, with substantial atrophy of the parietal lobes and precuneus. Grade 3 represents end-stage atrophy with evident widening of both sulci and knife-blade atrophy of the parietal lobes and precuneus (the different grades of atrophy are shown in Figure 1). In case of different scores on different orientations (e.g. score 1 on sagittal direction and score 2 on axial direction), the highest score was considered.
Visual rating scale for the posterior brain regions. In sagittal, axial and coronal orientation, this rating scale rates 0 = no atrophy, 1 = minimal atrophy, 2 = moderate atrophy and 3 = severe atrophy. In sagittal orientation, widening of posterior cingulate- and parieto-occipital sulcus and atrophy of the precuneus was evaluated. In axial orientation, the widening of the posterior cingulate sulcus and sulcal dilatation in the parietal lobes was evaluated. In coronal orientation, the widening of the posterior cingulate sulcus and sulcal dilatation in the parietal lobes was evaluated (reproduced from "Neuroimaging in Dementia" by Barkhof et al., 2011, fig.3.10 © Springer Science + Business Media). Abbreviations: PSC posterior cingulate sulcus, POS parieto-occipital sulcus, PRE precuneus and PAR parietal lobe
Image analysis
All 120 MR examinations were rated for PA by three different raters (rater 1: neuroradiologist, 8-year experience in imaging in dementia, rater 2: MD working as PhD-student and rater 3: PhD-student, both 3-year experience in imaging in dementia). All raters were blinded for the clinical diagnosis and paraclinical tests. Twenty-nine MR examinations were rated twice by two observers (rater 1 and 2) to assess intra-rater agreement. All MR examinations were rated for MTA using a 5-point rating scale (0–4) and for global cortical atrophy (GCA) by two raters (rater 1 and 2) using a 4-point (0–3) rating scale.[9, 35] GCA was assessed on axial MPR sections of the FLAIR sequence and MTA was assessed on oblique-coronal MPR sections of the T1-weighted sequence.
Statistical analysis
For statistical analysis we used averaged scores of the left and right hemisphere for the posterior scale and MTA scale. Basic demographic data were examined using Chi-square tests for categorical data and t-tests for continuous data. To analyse differences in mean atrophy scores between the groups, scores of one rater (rater 1) were used. To quantify inter- and intra-observer agreement we calculated weighted-kappa’s between each pair of observers and between the first and second session of two raters separately. For weighted-kappa values, degree of agreement was defined according to Landis and Koch [36].
Linear-regression analysis was used to analyse relationships between scores on each visual rating scale and age and MMSE score. In the first model relationships of each visual rating scale (independent variable) with either MMSE or age (dependent variable) were assessed unadjusted. In the second model the same relationships were assessed, corrected for age and sex in the analysis with the MMSE and corrected for sex in the analysis with age. In the third model both visual rating scales were entered simultaneously, together with the same covariates. Results are presented as linear-regression coefficients with standard error, B (SE). Statistical significance was set at p < 0.05.
Results
Patient characteristics
Two controls were excluded, since sagittal T1-weighted sequences were missing, resulting in a total group of 118 patients (60 AD, 20 other dementias and 38 controls).
Table 1 shows demographical and imaging characteristics of the study population. The groups were similar with respect to age. Sex was equally distributed within AD patients and controls (roughly 50% female), whereas women made up 25% of other dementias patients (p < 0.05). Disease duration (time between first symptoms and diagnosis) did not differ between AD and other dementias. Mean MTA scores were higher in AD compared with controls (1.3 ± 1.0 versus 0.4 ± 0.7, p < 0.01), but did not differ from other dementias (1.2 ± 1.0). GCA scores were higher in AD compared with controls (1.1 ± 0.7 versus 0.2 ± 0.5, p < 0.01) and other dementias (0.7 ± 0.7, p < 0.05).
Mean scores on the PA rating scale were higher in AD compared with controls (1.6 ± 0.9 and 0.6 ± 0.7, p < 0.01) and other dementias (0.8 ± 0.8, p < 0.01). PA scores did not differ between other dementias and controls.
Rater agreement
Intra-rater agreement (determined in 29 MR examinations for 2 raters) for PA was excellent with a weighted-kappa value of 0.93 (rater 1) and 0.95 (rater 2). Inter-rater agreement (determined in 118 examinations for 3 rater pairings) was best between rater 1 and 2, with a value of 0.84, followed by a value of 0.70 between rater 2 and 3 and lowest between rater 1 and 3, with a value of 0.65. The average value for inter-rater agreement (0.73) was good. Intra-rater agreement for MTA varied between 0.91 and 0.95, whereas values for inter-rater agreement varied between 0.82 (rater 1 and 3) and 0.90 (rater 1 and 2 and rater 2 and 3) (Table 2). GCA was only rated by two raters (rater 1 and 2), intra-rater agreement was 0.85 for both raters and inter-rater agreement was 0.70.
Clinical correlations
In a separate analysis we investigated relationships between PA and MTA with either age or MMSE across the entire group (Table 3). Higher ratings of MTA were associated with older age (B = 3.1 (0.7), p < 0.01) and lower MMSE scores (B = −2.0 (0.5), p < 0.01). By contrast, PA was not related to age, but there was an equally strong relationship with MMSE scores (B = −1.8 (0.5), p < 0.01)). These results remained essentially unchanged when both scales were entered into the same model, illustrating their independent effects on cognitive deterioration.
Rating scales: sensitivity and specificity
To further investigate how MTA and PA contribute to AD, we investigated the degree of MTA and PA in AD, controls and other dementias (Table 4). We therefore dichotomized scores for MTA and PA, with an average score of >1 being considered abnormal for both rating scales. Most of the controls had normal scores (≤1) for MTA and PA (N = 33, 88%). A minority had either MTA or PA, but none of the controls had abnormal scores (>1) for both regions. In AD, 28% had prominent PA (scores >1) without evident MTA, whereas only 15% had prominent MTA (scores >1) without evident PA. Minimal MTA and PA was found in 27% of the AD patients. The sensitivity of PA for AD was 58%, with a specificity of 95%, whereas MTA had a sensitivity of 45% with a specificity of 92%. Combining MTA and PA (combining patients with either MTA or PA, or a combination of both) increased sensitivity to 73% and slightly decreased specificity to 87%.
Most other dementia patients had normal scores for MTA and PA (N = 9, 45%).
When we looked at early- and late-onset AD separately, it became obvious that in late-onset AD approximately the same proportion had either prominent PA or MTA (20% versus 16%). However, in EOAD, 34% had prominent PA, whereas only 14% had prominent MTA. Disease duration did not differ between early- and late-onset AD (3.3 years ± 1.9 versus 3.0 years ± 1.6, p = 0.5).
Discussion
PA is seen in a substantial proportion of AD patients, perhaps particularly in those with an early onset. Because of the complexity of anatomy and variability in posterior regions, this atrophy may be less obvious to inspection than MTA. Moreover, a visual rating scale for PA has been lacking. With this study we aimed to develop a robust visual rating scale, to detect the wide range and complexity of PA. We found that agreement on visual assessment of PA varied between good and excellent. Furthermore, higher PA scores were found in AD compared to controls and other dementias and therefore the scale may be useful in the discrimination between AD and other dementias.
Several studies have shown that the precuneus, parietal lobes and posterior cingulate gyrus are frequently affected in (younger) AD patients and our visual rating scale was designed to include these regions [3, 19, 21–26].
Visual rating of atrophy has shown to be a quick and reproducible method to assess the degree of atrophy in a clinical setting, compared to more labour intensive techniques like volumetric analysis. For applicability in different clinical settings or by different raters, good inter- and intra-rater agreement is essential. Visual rating of MTA using a 5-point scale has become fairly established and has shown fair to good inter-rater agreement [9, 12, 37]. Visual rating of GCA seems harder to perform reliably between different raters, while despite moderate to good intra-rater agreement, poor inter-rater agreement has been reported [35]. Our PA rating scale varied between good and excellent inter- and intra-rater agreement even between raters with different degrees of expertise, suggesting its usefulness in a clinical setting as an aid in assessment of PA. However, degree of agreement was lower for PA compared to MTA (Table 2), which could be due to the fact that visual assessment of MTA is applied for many years in our memory clinic and raters gained much experience. Visual assessment of PA on the contrary has not been done before and our study is the first to assess the applicability of visual assessment of PA in a clinical setting.
MTA is one of the most important markers of AD on MR imaging. In the newly developed AD research criteria, presence of early episodic memory decline and MTA is sufficient for a diagnosis of prodromal AD [4]. MTA, however, has been described in other dementias, which makes MTA by itself less useful in the discrimination of AD from other dementias [13–16, 18]. Furthermore, there is an overlap in hippocampal volumes with normal aging, which could make the interpretation of MTA more difficult in older subjects [17].
There is increasing evidence that posterior brain regions are relatively more affected in EOAD and medial temporal lobe structures may be less involved [19, 21–24, 26]. Several clinico-pathological studies have reported involvement of the posterior cingulate gyrus, parieto-occipital-, and temporoparietal cortex in AD patients with typical as well as atypical clinical presentations, although the combination with relative sparing of the medial temporal lobe occurs especially in patients presenting without prominent memory impairment [3, 25, 28–30]. PA may be under-recognized, as clinicians may be unaware of this relatively subtle MR imaging characteristic of AD. Our visual assessment of PA was able to discriminate AD from controls and other dementias, whereas MTA failed to discriminate between AD and other dementias. We found that a considerable proportion of AD patients (28%) had prominent PA and mild or absent MTA, whereas only 15% had prominent MTA and mild or absent PA. Furthermore, combining MTA and PA increased the sensitivity for AD. This underlines the importance of evaluating PA in the diagnostic work-up of dementia.
Earlier studies showed that PA was more prominent in younger AD patients [22, 26]. We found that the proportion of patients with predominant PA was higher in EOAD compared to late-onset AD (34% versus 20%). Furthermore, patients with only prominent PA were younger than patients with only prominent MTA, although this difference did not reach statistical significance. However, we did not find a direct relation between PA and age with linear-regression analysis.
A previous study has shown that MTA is correlated with memory deficits and lower MMSE scores in AD [9]. PA seems to have clinical relevance as well, since PA was clearly associated with lower MMSE scores, independent of MTA. This is perhaps not surprising, given the importance of posterior (non-memory) aspects of the MMSE, such as calculation and visuospatial function.
A limitation of our study is lack of pathological confirmation of the diagnoses. We therefore cannot exclude other underlying abnormalities or mixed disease processes. However, all patients were carefully screened and fulfilled clinical criteria for the specific dementia types. It should be noted that our cohort consisted of relatively young AD patients, which could have influenced our results, since PA may be relatively more common in younger patients. Before generalisation of our results to older AD patients, further research is necessary to determine the prevalence of PA in large numbers of young and elderly AD patients and assess its discriminative power and the potential influence of age on its utility.
A considerable number of patients attending a memory clinic are not able to undergo MR imaging for any number of reasons (e.g. pacemaker, limited availability of MR imaging etc.) and computed tomography (CT) could be an alternative. Visual assessment of MTA has found to be comparable on CT and MR imaging and future work should address whether our rating scale is also applicable for CT [38].
In conclusion this study provides an easy applicable and robust visual rating scale to assess PA. Measurement of PA using this visual rating scale appears to add value in the discrimination of AD from controls and other dementias (FTLD and DLB) on MRI.
References
Hodges JR (2006) Alzheimer's centennial legacy: origins, landmarks and the current status of knowledge concerning cognitive aspects. Brain 129:2811–2822
Alladi S, Xuereb J, Bak T et al (2007) Focal cortical presentations of Alzheimer's disease. Brain 130:2636–2645
Galton CJ, Patterson K, Xuereb JH et al (2000) Atypical and typical presentations of Alzheimer's disease: a clinical, neuropsychological, neuroimaging and pathological study of 13 cases. Brain 123:484–498
Dubois B, Feldman HH, Jacova C et al (2007) Research criteria for the diagnosis of Alzheimer's disease: revising the NINCDS-ADRDA criteria. Lancet Neurol 6:734–746
Scheltens PH (2001) Structural neuroimaging of Alzheimer's disease and other dementias. Aging (Milano) 13:203–209
Erkinjuntti T, Lee DH, Gao F et al (1993) Temporal lobe atrophy on magnetic resonance imaging in the diagnosis of early Alzheimer's disease. Arch Neurol 50:305–310
Jack CR Jr, Petersen RC, O'Brien PC et al (1992) MR-based hippocampal volumetry in the diagnosis of Alzheimer's disease. Neurology 42:183–188
Kesslak JP, Nalcioglu O, Cotman CW (1991) Quantification of magnetic resonance scans for hippocampal and parahippocampal atrophy in Alzheimer's disease. Neurology 41:51–54
Scheltens P, Leys D, Barkhof F et al (1992) Atrophy of medial temporal lobes on MRI in "probable" Alzheimer's disease and normal ageing: diagnostic value and neuropsychological correlates. J Neurol Neurosurg Psychiatry 55:967–972
Korf ES, Wahlund LO, Visser PJ et al (2004) Medial temporal lobe atrophy on MRI predicts dementia in patients with mild cognitive impairment. Neurology 63:94–100
Bresciani L, Rossi R, Testa C et al (2005) Visual assessment of medial temporal atrophy on MR films in Alzheimer's disease: comparison with volumetry. Aging Clin Exp Res 17:8–13
Scheltens P, Launer LJ, Barkhof F et al (1995) Visual assessment of medial temporal lobe atrophy on magnetic resonance imaging: interobserver reliability. J Neurol 242:557–560
Barber R, Gholkar A, Scheltens P et al (1999) Medial temporal lobe atrophy on MRI in dementia with Lewy bodies. Neurology 52:1153–1158
Bastos-Leite AJ, van der Flier WM, van Straaten EC et al (2007) The contribution of medial temporal lobe atrophy and vascular pathology to cognitive impairment in vascular dementia. Stroke 38:3182–3185
Laakso MP, Partanen K, Riekkinen P et al (1996) Hippocampal volumes in Alzheimer's disease, Parkinson's disease with and without dementia, and in vascular dementia: an MRI study. Neurology 46:678–681
Staekenborg SS, van Straaten EC, van der Flier WM et al (2008) Small vessel versus large vessel vascular dementia: risk factors and MRI findings. J Neurol 255:1644–1651
van de Pol LA, Hensel A, Barkhof F et al (2006) Hippocampal atrophy in Alzheimer disease: age matters. Neurology 66:236–238
van de Pol LA, Hensel A, van der Flier WM et al (2006) Hippocampal atrophy on MRI in frontotemporal lobar degeneration and Alzheimer's disease. J Neurol Neurosurg Psychiatry 77:439–442
Frisoni GB, Testa C, Sabattoli F et al (2005) Structural correlates of early and late onset Alzheimer's disease: voxel based morphometric study. J Neurol Neurosurg Psychiatry 76:112–114
Benson DF, Davis RJ, Snyder BD (1988) Posterior cortical atrophy. Arch Neurol 45:789–793
Barnes J, Godbolt AK, Frost C et al (2007) Atrophy rates of the cingulate gyrus and hippocampus in AD and FTLD. Neurobiol Aging 28:20–28
Frisoni GB, Pievani M, Testa C et al (2007) The topography of grey matter involvement in early and late onset Alzheimer's disease. Brain 130:720–730
Ishii K, Kawachi T, Sasaki H et al (2005) Voxel-based morphometric comparison between early- and late-onset mild Alzheimer's disease and assessment of diagnostic performance of z score images. AJNR Am J Neuroradiol 26:333–340
Jones BF, Barnes J, Uylings HB et al (2006) Differential regional atrophy of the cingulate gyrus in Alzheimer disease: a volumetric MRI study. Cereb Cortex 16:1701–1708
Josephs KA, Whitwell JL, Duffy JR et al (2008) Progressive aphasia secondary to Alzheimer disease vs FTLD pathology. Neurology 70:25–34
Karas G, Scheltens P, Rombouts S et al (2007) Precuneus atrophy in early-onset Alzheimer's disease: a morphometric structural MRI study. Neuroradiology 49:967–976
Scahill RI, Schott JM, Stevens JM et al (2002) Mapping the evolution of regional atrophy in Alzheimer's disease: unbiased analysis of fluid-registered serial MRI. Proc Natl Acad Sci USA 99:4703–4707
Lehmann M, Rohrer JD, Clarkson MJ et al (2010) Reduced cortical thickness in the posterior cingulate gyrus is characteristic of both typical and atypical Alzheimer's disease. J Alzheimers Dis 20:587–598
Whitwell JL, Jack CR Jr, Przybelski SA et al (2009) Temporoparietal atrophy: a marker of AD pathology independent of clinical diagnosis. Neurobiol Aging. doi:10.1016/j.neurobiolaging.2009.10.012
Lehmann M, Crutch SJ, Ridgway GR et al (2009) Cortical thickness and voxel-based morphometry in posterior cortical atrophy and typical Alzheimer's disease. Neurobiol Aging 32:1466–1476, Epub 2009 Sep 25
Folstein MF, Folstein SE, McHugh PR (1975) Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
McKeith IG, Galasko D, Kosaka K et al (1996) Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology 47:1113–1124
McKhann G, Drachman D, Folstein M et al (1984) Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology 34:939–944
Neary D, Snowden JS, Gustafson L et al (1998) Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 51:1546–1554
Scheltens P, Pasquier F, Weerts JG et al (1997) Qualitative assessment of cerebral atrophy on MRI: inter- and intra-observer reproducibility in dementia and normal aging. Eur Neurol 37:95–99
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Galton CJ, Gomez-Anson B, Antoun N et al (2001) Temporal lobe rating scale: application to Alzheimer's disease and frontotemporal dementia. J Neurol Neurosurg Psychiatry 70:165–173
Wattjes MP, Henneman WJ, van der Flier WM et al (2009) Diagnostic imaging of patients in a memory clinic: comparison of MR imaging and 64-detector row CT. Radiology 253:174–183
Acknowledgement
The authors wish to thank Dr. Dirk L. Knol, Department of Epidemiology and Biostatistics, VU University Medical Center Amsterdam for his statistical advice. NCF is supported by an MRC (UK) Senior Clinical Fellowship and holds a National Institute for Health Research (NIHR) senior investigator award. ML is supported by the Alzheimer’s Society.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Koedam, E.L.G.E., Lehmann, M., van der Flier, W.M. et al. Visual assessment of posterior atrophy development of a MRI rating scale. Eur Radiol 21, 2618–2625 (2011). https://doi.org/10.1007/s00330-011-2205-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-011-2205-4