Abstract
Objective
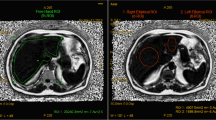
The purpose of this study was to evaluate the ability of a whole liver volume (WLV) segmentation algorithm to measure fat fraction (FF).
Methods
Twenty consecutive patients with histologically proven fatty liver disease underwent dual-echo in-phase/out-of-phase MRI and magnetic resonance spectroscopy (MRS) at 1.5 T. Two readers independently performed semiautomatic 3D liver segmentation on the out-of-phase sequences using an active contour model. FF was calculated for voxels, segments and WLV. Segmentation inter-observer reproducibility was assessed by intra-class correlation coefficient (ICC) for WLV and FF. Fat fraction correlation and agreement as determined by histology, MRS and MRI were determined.
Results
ICC was 0.999 (95% CI: 0.999-1, P < 0.001) for WLV FF calculation and 0.996 (95% CI: 0.990–0.998, P < 0.001) for whole liver volume calculations. Strong correlations were found between FF measured by histology, MRS and WLV-MRI. A Bland-Altman analysis showed a good agreement between FF measured by MRS and WLV-MRI. No systematic variations of FF was found between segments when analyzed by ANOVA (F = 1.78, P = 0.096).
Conclusion
This study shows that a reproducible whole liver volume segmentation method to measure fat fraction can be performed. This strategy may be integrated to a “one-stop shop” protocol in liver surgery planning.




Similar content being viewed by others
References
Massoptier L, Casciaro S (2008) A new fully automatic and robust algorithm for fast segmentation of liver tissue and tumors from CT scans. Eur Radiol 18:1658–1665
Lee J, Kim N, Lee H et al (2007) Efficient liver segmentation using a level-set method with optimal detection of the initial liver boundary from level-set speed images. Comput Methods Programs Biomed 88:26–38
Okada T, Shimada R, Sato Y et al (2007) Automated segmentation of the liver from 3D CT images using probabilistic atlas and multi-level statistical shape model. Med Image Comput Comput Assist Interv 10:86–93
Radtke A, Nadalin S, Sotiropoulos GC et al (2007) Computer-assisted operative planning in adult living donor liver transplantation: a new way to resolve the dilemma of the middle hepatic vein. World J Surg 31:175–185
Hermoye L, Laamari-Azjal I, Cao Z et al (2005) Liver segmentation in living liver transplant donors: comparison of semiautomatic and manual methods. Radiology 234:171–178
Gloger O, Kuhn J, Stanski A et al (2010) A fully automatic three-step liver segmentation method on LDA-based probability maps for multiple contrast MR images. Magn Reson Imaging 28(6):882–897
Ferrero A, Vigano L, Polastri R et al (2007) Postoperative liver dysfunction and future remnant liver: where is the limit? Results of a prospective study. World J Surg 31:1643–1651
Vetelainen R, van Vliet AK, van Gulik TM (2007) Severe steatosis increases hepatocellular injury and impairs liver regeneration in a rat model of partial hepatectomy. Ann Surg 245:44–50
Vetelainen R, van Vliet A, Gouma DJ et al (2007) Steatosis as a risk factor in liver surgery. Ann Surg 245:20–30
Vetelainen R, Bennink RJ, van Vliet AK et al (2007) Mild steatosis impairs functional recovery after liver resection in an experimental model. Br J Surg 94:1002–1008
Baccarani U, Adani GL, Lorenzin D et al (2009) The role of steatosis of the liver graft in the development of post-transplant biliary complications. Transpl Int 23:239
Ploeg RJ, D’Alessandro AM, Knechtle SJ et al (1993) Risk factors for primary dysfunction after liver transplantation–a multivariate analysis. Transplantation 55:807–813
Fiorini RN, Kirtz J, Periyasamy B et al (2004) Development of an unbiased method for the estimation of liver steatosis. Clin Transplant 18:700–706
El-Badry AM, Breitenstein S, Jochum W et al (2009) Assessment of hepatic steatosis by expert pathologists: the end of a gold standard. Ann Surg 250:691–697
d’Assignies G, Ruel M, Khiat A et al (2009) Noninvasive quantitation of human liver steatosis using magnetic resonance and bioassay methods. Eur Radiol 19:2033–2040
O’Regan DP, Callaghan MF, Wylezinska-Arridge M et al (2008) Liver fat content and T2*: simultaneous measurement by using breath-hold multiecho MR imaging at 3.0 T–feasibility. Radiology 247:550–557
Kim H, Taksali SE, Dufour S et al (2008) Comparative MR study of hepatic fat quantification using single-voxel proton spectroscopy, two-point dixon and three-point IDEAL. Magn Reson Med 59:521–527
Cowin GJ, Jonsson JR, Bauer JD et al (2008) Magnetic resonance imaging and spectroscopy for monitoring liver steatosis. J Magn Reson Imaging 28:937–945
Marsman H, Matsushita T, Dierkhising R et al (2004) Assessment of donor liver steatosis: pathologist or automated software? Hum Pathol 35:430–435
Szczepaniak LS, Nurenberg P, Leonard D et al (2005) Magnetic resonance spectroscopy to measure hepatic triglyceride content: prevalence of hepatic steatosis in the general population. Am J Physiol Endocrinol Metab 288:E462–468
Strunk H, Stuckmann G, Textor J et al (2003) Limitations and pitfalls of Couinaud’s segmentation of the liver in transaxial imaging. Eur Radiol 13:2472–2482
Campadelli P, Casiraghi E, Esposito A (2009) Liver segmentation from computed tomography scans: a survey and a new algorithm. Artif Intell Med 45:185–196
Cheng YF, Chen CL, Huang TL et al (2001) Single imaging modality evaluation of living donors in liver transplantation: magnetic resonance imaging. Transplantation 72:1527–1533
Joy D, Thava VR, Scott BB (2003) Diagnosis of fatty liver disease: is biopsy necessary? Eur J Gastroenterol Hepatol 15:539–543
Ratziu V, Charlotte F, Heurtier A et al (2005) Sampling variability of liver biopsy in nonalcoholic fatty liver disease. Gastroenterology 128:1898–1906
Guiu B, Petit JM, Loffroy R et al (2009) Quantification of liver fat content: comparison of triple-echo chemical shift gradient-echo imaging and in vivo proton MR spectroscopy. Radiology 250:95–102
Guiu B, Loffroy R, Petit JM et al (2009) Mapping of liver fat with triple-echo gradient echo imaging: validation against 3.0-T proton MR spectroscopy. Eur Radiol 19:1786–1793
Biglands JD (2007) Comparison of MRI and histopathalogic methods of quantifying hepatic fat fraction. ISMRM, Berlin
Longo R, Pollesello P, Ricci C et al (1995) Proton MR spectroscopy in quantitative in vivo determination of fat content in human liver steatosis. J Magn Reson Imaging 5:281–285
Belghiti J, Ogata S (2005) Assessment of hepatic reserve for the indication of hepatic resection. J Hepatobiliary Pancreat Surg 12:1–3
Frankel WL, Tranovich JG, Salter L et al (2002) The optimal number of donor biopsy sites to evaluate liver histology for transplantation. Liver Transplant 8:1044–1050
Rinella ME, McCarthy R, Thakrar K et al (2003) Dual-echo, chemical shift gradient-echo magnetic resonance imaging to quantify hepatic steatosis: Implications for living liver donation. Liver Transplant 9:851–856
Thomas EL, Hamilton G, Patel N et al (2005) Hepatic triglyceride content and its relation to body adiposity: a magnetic resonance imaging and proton magnetic resonance spectroscopy study. Gut 54:122–127
Cotler SJ, Guzman G, Layden-Almer J et al (2007) Measurement of liver fat content using selective saturation at 3.0 T. J Magn Reson Imaging 25:743–748
Bugianesi E (2007) Non-alcoholic steatohepatitis and cancer. Clin Liver Dis 11:191–207, x–xi
Fulcher AS, Szucs RA, Bassignani MJ et al (2001) Right lobe living donor liver transplantation: preoperative evaluation of the donor with MR imaging. AJR Am J Roentgenol 176:1483–1491
Acknowledgements
This work was supported by a clinical research scholarship to G.S. from Fonds de la recherche en santé du Québec (FRSQ) and to G.A. from the Société Française de Radiologie.
Author information
Authors and Affiliations
Corresponding author
Appendix: T1 and T2 correction on single-voxel proton MRS data
Appendix: T1 and T2 correction on single-voxel proton MRS data
T2 corrections were performed using the equation \( {\hbox{SI}} = {\hbox{SI}}_0\,{ \exp }\left( { - {\hbox{TE}}/{\hbox{T2}}} \right) \), SI and SI0 being the corrected and uncorrected signal intensities, respectively, TE the echo time and T2 the reported transverse relaxation times of 60 ms for lipids and 50 ms for water [20]. The T1 correction was performed using the equation \( {\hbox{SI}} = {\hbox{SI}}_0\,({1}/\left( {{1} - { \exp }\left( { - {\hbox{TR}}/{\hbox{T1}}} \right)} \right) \), TR being the repetition time and T1 the reported longitudinal relaxation time of 663 ms for water and 236 ms for lipids [29]. Data were also corrected for (1) the ratio of the number of lipid protons evaluated by MRS relative to the total number of lipid protons (0.85), (2) the proton densities of fat (110 mol/l) and water (111 mol/l), (3) the fat/water weight ratio in the liver (0.711 g fat/g water) and (4) the density of liver tissue (1.051 g/l) and the density of liver fat (0.90 g/l) using the method described by Longo et al. to yield fat fractions expressed in relative volumes[29]. The fat fraction (FF) determined by MRS corresponds to the corrected L/(L+W) ratio.
Rights and permissions
About this article
Cite this article
d’Assignies, G., Kauffmann, C., Boulanger, Y. et al. Simultaneous assessment of liver volume and whole liver fat content: a step towards one-stop shop preoperative MRI protocol. Eur Radiol 21, 301–309 (2011). https://doi.org/10.1007/s00330-010-1941-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-010-1941-1