Abstract
Objective
To meta-analyse the diagnostic accuracy of US, CT, MRI and 1H-MRS for the evaluation of hepatic steatosis.
Methods
From a comprehensive literature search in MEDLINE, EMBASE, CINAHL and Cochrane (up to November 2009), articles were selected that investigated the diagnostic performance imaging techniques for evaluating hepatic steatosis with histopathology as the reference standard. Cut-off values for the presence of steatosis on liver biopsy were subdivided into four groups: (1) >0, >2 and >5% steatosis; (2) >10, >15 and >20%; (3) >25, >30 and >33%; (4) >50, >60 and >66%. Per group, summary estimates for sensitivity and specificity were calculated. The natural-logarithm of the diagnostic odds ratio (lnDOR) was used as a single indicator of test performance.
Results
46 articles were included. Mean sensitivity estimates for subgroups were 73.3–90.5% (US), 46.1–72.0% (CT), 82.0–97.4% (MRI) and 72.7–88.5% (1H-MRS). Mean specificity ranges were 69.6–85.2% (US), 88.1–94.6% (CT), 76.1–95.3% (MRI) and 92.0–95.7% (1H-MRS). Overall performance (lnDOR) of MRI and 1H-MRS was better than that for US and CT for all subgroups, with significant differences in groups 1 and 2.
Conclusion
MRI and 1H-MRS can be considered techniques of choice for accurate evaluation of hepatic steatosis.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
The prevalence of hepatic steatosis is increasing rapidly worldwide. This is largely attributed to the association with obesity and insulin resistance in non-alcoholic fatty liver disease (NAFLD) [1, 2].
Detection and quantification of hepatic steatosis is clinically important. In NAFLD, steatosis is the hepatic manifestation of the metabolic syndrome and the earliest biomarker for the development of liver fibrosis in the more severe condition of non-alcoholic steatohepatitis (NASH). Early diagnosis and treatment of NASH can prevent the potential development of cirrhosis and hepatocellular carcinoma (HCC) [3–5]. In hepatitis C, steatosis is associated with more severe fibrosis and rapid disease progression [6, 7]. In liver transplantation surgery, the presence of steatosis impairs the regenerative capacity of the liver in both donor and recipient [8, 9].
Liver biopsy remains the reference test for the evaluation of hepatic steatosis, despite well-established drawbacks regarding its invasiveness and sampling error due to small sample size and inter-observer variability [10].
Many studies have focused on the role of imaging techniques as a non-invasive alternative to liver biopsy for detecting and quantifying hepatic steatosis [11–13]. The reported sensitivities and specificities between different imaging techniques and between different studies investigating the same technique vary substantially. Although magnetic resonance spectroscopy (1H-MRS)—generally considered the best technique—is increasingly used as a reference standard instead of liver biopsy, no evidence-based consensus currently exists on this topic.
The purpose of this systematic review therefore was to summarise the available literature on the accuracy of ultrasound (US), computed tomography (CT), magnetic resonance imaging (MRI) and 1H-MRS for the evaluation of hepatic steatosis with histopathology as the reference test. Subsequently, we aimed to identify the most accurate technique by meta-analysis.
Materials and methods
Literature search and study selection
We searched the MEDLINE (January 1966–November 2009), EMBASE (January 1980–November 2009), CINAHL and Cochrane databases without language restrictions with the assistance of an experienced clinical librarian. We combined Medical Subject Headings (MeSH) terms and accompanying entry terms for the patient group (patients with hepatic steatosis) and the index test (US, CT, MRI, 1H-MRS). The search strategy is described in detail in Online Resource 1.
Two reviewers (A.B. and J.v.W) read the titles and abstracts of all the articles obtained to select potentially relevant papers (original papers that addressed the diagnostic accuracy of US, CT, MRI or 1H-MRS for detecting hepatic steatosis in humans with histopathology as the reference test in ≥10 individuals). The reference lists of selected papers and of narrative reviews were screened for search completion.
The full texts of potentially relevant papers were reviewed for inclusion by the same reviewers independently. Inclusion criteria were: (a) hepatic steatosis was evaluated with US, CT, MRI and/or 1H-MRS; (b) imaging techniques met the minimum technical requirements of grey scale and real time for US; ≤120 kV for CT and ≥1 Tesla for MRI; (c) histopathology as the reference test; (d) evaluation of ≥10 human individuals; (e) criteria for a positive index test were clearly explained; (f) examination method of steatosis on liver biopsy was clearly explained; (g) data on diagnostic accuracy were reported. Exclusion criteria were: (a) duplicate publication; (b) reporting of combined data for different imaging techniques or data on the single technique could not be extracted; (c) no original research. Papers were not blinded with regard to authors’ names, affiliations or journal. The reviewers resolved all disagreements about inclusion and data extraction by consensus after face-to-face discussion.
Data extraction
From the articles included, the reviewers (A.B. and J.v.W.) independently recorded data using a standardised form. Papers were translated if necessary.
Methodological quality
Methodological quality was assessed based on the Quality Assessment of Studies of Diagnostic Accuracy included in Systematic Reviews (QUADAS) guidelines [14]. To be reasonably sure that the condition of the liver did not change between the two tests we chose a period of 1 month as a quality indicator [15, 16]. Additionally, we noted whether the study design was prospective or retrospective.
Patient characteristics
For each study, we extracted data on (a) sample size; (b) male-female ratio; (c) patient age; (d) body mass index (BMI) and (e) patient spectrum.
Imaging features and evaluation
For each imaging technique we recorded (a) the number of patients; (b) the criteria used for steatosis evaluation and (c) the cut-off values used. We noted whether cut-off values were defined prospectively or retrospectively. Additionally, for US we noted the type and frequency of the probe(s) used. For CT we noted: (a) the type of CT and (b) the imaging parameters (kV and mAs). For MRI, we noted: (a) magnetic field strength; (b) imaging sequence; (c) imaging parameters used; (d) whether breath holds were used; and (e) correction for T2* effects. For 1H-MRS, we noted: (a) magnetic field strength; (b) imaging sequence; (c) imaging parameters; (d) voxel size; (e) whether breath holds were used and (f) correction for T1 or T2 effects.
Reference test
For liver biopsy we included data on: (a) the number of patients biopsied; (b) cut-off values for steatosis grades and whether the presence of (c) fibrosis (d) inflammation and (e) iron was evaluated in the biopsy specimen.
Data for calculation of diagnostic accuracy
We extracted available data on true-positives (TP), false-negatives (FN), false-positives (FP) and true-negatives (TN) for detecting steatosis with the selected imaging technique to construct 2 × 2 contingency tables. Available 3 × 3 and 4 × 4 tables were dichotomised. Many different cut-off values for positive results (steatosis present) on liver biopsy were compared to the imaging techniques. We therefore grouped accuracy results from cut-off values that were almost equal into four subgroups to enable meta-analysis:
-
Group 1:
Cut-off values of >0%, >2% and >5% steatosis on biopsy;
-
Group 2:
Cut-off values of >10%, >15% and >20% steatosis on biopsy;
-
Group 3:
Cut-off values of >25%, >30% and >33% steatosis on biopsy and the qualitative designation of “moderate or severe” steatosis;
-
Group 4:
Cut-off values of >50%, >60% and >66% steatosis on biopsy and the qualitative designation of “severe” steatosis.
During analysis we corrected for dependent data such as results presented for different readers or for multiple imaging techniques within one study population. For CT and MRI, we did not use data obtained by subjective visual evaluation of examinations for analysis. If raw data in terms of 2 × 2 tables were unavailable, we attempted to contact authors for completion or verification of data.
Data analysis
We performed bivariate random-effects analysis for the pooled sensitivities and specificities per cut-off value group for each imaging technique [17]. In this analysis, the logit-transformed sensitivities and logit-transformed specificities from individual studies in a meta-analysis are assumed to follow a bivariate normal distribution around a mean logit sensitivity and a mean logit specificity. After antilogit transformation, summary estimates of sensitivity and specificity with 95% confidence intervals were calculated. Additionally, we calculated the natural logarithm (ln) of the diagnostic odds ratio (DOR): [logit sensitivity + logit specificity]. The DOR is a single indicator of test performance [18]. A higher lnDOR value indicates a better discriminatory test performance. If the lnDOR is not significantly different from 0, a test does not discriminate between patients with the disorder and those without it. We performed z-tests to compare sensitivities, specificities and lnDORs between imaging techniques. A p value < 0.05 was considered statistically significant. All meta-analyses were performed with SAS statistical software (version 9.1, SAS institute, Cary, NC, USA).
Results
Literature search and study selection
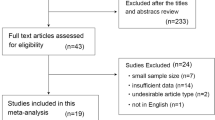
The literature search yielded 6992 unique references (Fig. 1). The reviewers selected 179 potentially relevant articles after reading the titles and abstracts of which 46 papers were finally included [15, 19–63]. Twenty-eight evaluated US, twelve evaluated CT, ten evaluated MRI and five evaluated 1H-MRS. Eight papers compared two imaging techniques [22, 23, 43, 47, 54, 58, 61, 62] and one evaluated three techniques within the same population [56] (Table 1). Two studies were published in German [28, 45] and the remaining in English.
Data extraction
Methodological quality
An overview of the results is given in Fig. 2. In general, 13 out of 46 (28%) studies fulfilled at least 10 of the 13 methodological criteria [19, 20, 25, 29, 33, 34, 36, 38, 41, 44, 48, 49, 52]. A complete table of individual study scores is available upon request from the authors.
Patient characteristics
The 46 papers comprised 4715 patients with a median study size of 81 patients (range 20–589). Mean age reported by 37 studies was 44.5 years (range 11–89). Mean BMI reported by 18 studies was 26.6 kg/m2 (range 15–54 kg/m2). The male-to-female ratio reported in 40 studies was 1.62:1. Potential living liver donors constituted 34% of the total population (1593/4715). If specified, the disease spectrum comprised most frequently chronic hepatitis C (n = 1040) and NAFLD/NASH (n = 710). See also Table 1.
Imaging features and evaluation
US imaging features are outlined in Online Resource 2. More than half (15/28) of the studies used the widely accepted criteria for subjective visual steatosis evaluation of bright liver with increased liver-kidney contrast; blurring of intrahepatic vessels and diaphragm and loss of echoes of posterior hepatic segments [57, 60]. Two studies evaluated quantitative methods to assess liver steatosis [30, 59].
CT imaging features are shown in Online Resource 3. Two papers evaluated both contrast-enhanced CT and unenhanced CT [22, 38]. All other papers evaluated unenhanced CT. Average Hounsfield Units (HU) in selected regions of interests (ROIs) from the liver were compared with average HU values of ROIs from the spleen. The spleen was used as an internal reference in all the papers included, either by measuring the liver-minus-spleen attenuation value (L-S) or the liver-to-spleen ratio (L/S). Two studies also evaluated steatosis by subtracting hepatic blood attenuation from the total hepatic attenuation using an algorithm. Four papers had defined cut-off values prospectively [22, 43, 58, 61]. Six papers defined optimal cut-off values retrospectively [15, 35, 38, 44, 52, 62]. The chosen cut-off values, however, varied substantially.
MRI characteristics are outlined in Online Resource 4. Magnetic field strength for all included papers was 1.5 Tesla. Sequences used were T1-weighted dual spin echo, T1-weighted dual gradient echo or T1-weighted spoiled gradient echo for in-phase and out-of-phase (IP/OP) chemical shift imaging. Two studies also evaluated T2-weighted fast spin echo imaging with and without fat suppression (±FS). Liver steatosis was evaluated by the amount of signal intensity (SI) loss on OP images compared with IP images and by SI difference between FS and non-FS images. Exact measuring methods, however, differed. Correction for T2* effects was performed by d’Assignies et al [24]. Cho et al were the only authors to define cut-off values prospectively [22].
1H-MRS imaging characteristics are outlined in Online Resource 5. Magnetic field strengths were 1.5 T(3/5) and 3 T (2/5). Four papers used a point-resolved spectroscopic sequence (PRESS), one in combination with chemical-shift selective water suppression (CHESS). Krššák et al used a stimulated echo acquisition mode (STEAM) sequence [41]. Voxel sizes varied from 18 × 18 × 18 mm to 30 × 30 × 40 mm. Hepatic steatosis was evaluated by the ratio of lipid versus water peaks and by the choline-to-lipid ratio. One paper each corrected for T2 effects or T1 and T2 effects [24, 41]. All included studies defined cut-off values retrospectively.
Reference test
Details of the reference test are outlined in Online Resource 6. Cut-off values for grading steatosis severity differed among the articles included, complicating their comparison. Two studies compared semi-quantitative visual analysis of steatosis with the automatic vacuole segmentation method [24] or gas-liquid chromatography [41]. Thirty studies examined the presence of fibrosis, 24 examined the presence of inflammatory activity and eight studies examined the presence of iron.
Data for calculation of diagnostic accuracy
Extraction of 2 × 2 accuracy data resulted in 48 complete data sets for US; 33 for CT; 15 for MRI and 7 for 1H-MRS. For both CT and MRI, 5 datasets were not included for analysis because the examinations were visually assessed. This resulted in 28 datasets being analysed for CT and 10 datasets being analysed for MRI. Datasets with TP and FN only were also included for analysis: 4 for US and 1 for CT. The number of datasets per cut-off group is noted in Table 2. Six authors were contacted for completion or verification of data; three answered of which one supplied additional datasets. Three studies reported data-sets for multiple readers [30, 31, 39].
Data-analysis
Sensitivity and specificity values including 95% confidence intervals (CI) and significant differences (p < 0.05) for the imaging techniques are presented in Table 2 and in more detail in the Online Resources 7–10.
Group 1 (cut-off values of >0%, >2% and >5% steatosis)
Sensitivity and specificity estimates were 73.3% and 84.4% for US; 46.1% and 93.5% for CT; 82.0% and 89.9% for MRI and 88.5% and 92.0% for 1H-MRS, respectively. The sensitivity of 1H-MRS was significantly higher than that of US (p = 0.04) and CT (p < 0.01) and the sensitivity of MRI was significantly higher than that of CT (p = 0.02). No significant differences in specificity were found. The lnDOR of 1H-MRS was significantly higher compared with US (p = 0.02) and CT (p = 0.04).
Group 2 (cut-off values of >10%, >15% and >20% steatosis)
Sensitivity and specificity estimates were 90.5% and 69.6% for US; 57.0% and 88.1% for CT; 90.0% and 95.3% for MRI and 82.6% and 94.3% for 1H-MRS, respectively. CT had a significantly lower sensitivity compared with US, MRI and 1H-MRS (p < 0.01, p < 0.01 and p = 0.02 respectively). Although US had a sensitivity comparable to MRI and 1H-MRS, the specificity was significantly lower than CT (p < 0.01), MRI (p < 0.01) and 1H-MRS (p = 0.01). The lnDOR of MRI was significantly higher than the lnDOR for both US (p = 0.05) and CT (p < 0.01). 1H-MRS had a significantly higher lnDOR than CT (p = 0.03).
Group 3 (cut-off values of >25%, >30% and >33% steatosis)
Sensitivity and specificity estimates were 85.7% and 85.2% for US; 72.0% and 94.6% for CT; 97.4% and 76.1% for MRI and 72.7% and 95.7% for 1H-MRS, respectively. The sensitivity of MRI was significantly higher than CT (p = 0.01) and 1H-MRS (p = 0.03). The specificity of MRI however was significantly lower than both CT (p = 0.02) and 1H-MRS (p = 0.04). Further, the sensitivity for US was significantly higher than for CT (p = 0.03), the specificity for US was significantly lower than for CT (p = 0.03). Analysis of the lnDOR did not show any significant differences.
Group 4 (cut-off values of >50%, >60% and >66% steatosis)
For this group, data analysis was possible for US only. Sensitivity and specificity estimates were 91.1% and 91.9% respectively.
Figure 3 shows the diagnostic performances (lnDOR) of all imaging techniques per cut-off value group, illustrating the better performance for both MRI and 1H-MRS compared with US and CT.
Discussion
Our results show that MRI and 1H-MRS perform better than US and CT over the total range of cut-off values that were analysed. For the lower cut-off ranges, we found significant differences in favour of both MRI and 1H-MRS.
These findings suggest that MRI and 1H-MRS also perform better than US and CT for detecting separate disease grades, especially for mild disease (<30% steatosis). This is of value in clinical practice when an accurate estimation of the amount of hepatic steatosis is needed. Additional benefits of MRI and 1H-MRS over US are the quantitative measurements which are less subject to inter- and intraobserver variability [64]. For CT, drawbacks are the radiation exposure and factors affecting the accuracy of the results, such as imaging parameters or iron accumulation [11, 65].
Several limitations of our study must be considered. First, the studies included showed great heterogeneity regarding patient spectrum, reference test, index test and data reporting. Therefore, comparison of separate disease grades and sub-analysis of different aetiologies of steatosis (e.g. NALFD/NASH versus HCV) was precluded. Standardisation of future study designs is needed to enable these comparisons. Moreover, no studies compared all four imaging techniques within the same population, which would be the ideal study design. We were therefore restricted to summarising accuracy data for each technique separately across all the studies included. These indirect comparisons of studies, which showed substantial methodological heterogeneity, might have biased our results.
Second, we had to make the decision to group accuracy results from different cut-off values into four subgroups to enable meta-analysis and to reduce the number of summary estimates and comparisons. The ideal situation would have been to analyse accuracy results for each cut-off value separately.
Third, a standard method for meta-analysis of diagnostic studies is the summary Receiver Operating Characteristic (sROC). For the sROC approach, a negative correlation between the logit sensitivity and the logit specificity is required [17]. As we did not find this negative correlation in our data, plotting of sROC curves was not possible. We therefore used the lnDOR to summarise our results.
Fourth, we did not analyse 3 × 3 or 4 × 4 data as the reporting thereof was scarce. By dichotomising the results, we lost information on the capability of imaging techniques to diagnose the degree of steatosis.
A fifth limitation was that we chose to exclude articles with 1H-MRS as the reference standard [66–73]. 1H-MRS is increasingly used as a reference standard for steatosis quantification since the results from the Dallas Heart Study were published by Szczepaniak et al in 2005 [74]. However, no clear consensus on this topic currently exists. The articles that were excluded all compared MRI with 1H-MRS and showed good correlations. Therefore, only a small number of datasets were available for analysis of MRI. Additionally, the included articles for MRI did not evaluate triple-echo, multi-echo or multi-interference techniques, whereas the aforementioned excluded articles did. Guiu et al recently suggested that these new techniques should replace the classical dual-echo chemical shift imaging methods, which are not reliable for quantification of liver fat in the case of liver iron overload because of T2* effects [75]. We believe that the small number of available data in combination with the techniques used could have negatively influenced our accuracy results for MRI.
We therefore recommend that consensus on the role of 1H-MRS as the reference standard needs to be established. For liver biopsy evaluation, we recommend using the classification from Kleiner et al for a uniform grading of hepatic steatosis [76].
In conclusion, we have shown that MRI and 1H-MRS are most accurate for the detection of hepatic steatosis. For future research, it is important to improve the study design and reporting of accuracy results.
References
Angulo P (2002) Nonalcoholic fatty liver disease. N Engl J Med 346:1221–1231
Williams R (2006) Global challenges in liver disease. Hepatology 44:521–526
Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A, Angulo P (2005) The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology 129:113–121
Farrell GC, Larter CZ (2006) Nonalcoholic fatty liver disease: from steatosis to cirrhosis. Hepatology 43:S99–S112
Rector RS, Thyfault JP, Wei Y, Ibdah JA (2008) Non-alcoholic fatty liver disease and the metabolic syndrome: an update. World J Gastroenterol 14:185–192
Adinolfi LE, Gambardella M, Andreana A, Tripodi MF, Utili R, Ruggiero G (2001) Steatosis accelerates the progression of liver damage of chronic hepatitis C patients and correlates with specific HCV genotype and visceral obesity. Hepatology 33:1358–1364
Rubbia-Brandt L, Fabris P, Paganin S, Leandro G, Male PJ, Giostra E, Carlotto A, Bozzola L, Smedile A, Negro F (2004) Steatosis affects chronic hepatitis C progression in a genotype specific way. Gut 53:406–412
Ploeg RJ, D’Alessandro AM, Knechtle SJ, Stegall MD, Pirsch JD, Hoffmann RM, Sasaki T, Sollinger HW, Belzer FO, Kalayoglu M (1993) Risk factors for primary dysfunction after liver transplantation–a multivariate analysis. Transplantation 55:807–813
Vetelainen R, van Vliet A, Gouma DJ, van Gulik TM (2007) Steatosis as a risk factor in liver surgery. Ann Surg 245:20–30
Bravo AA, Sheth SG, Chopra S (2001) Liver biopsy. N Engl J Med 344:495–500
Charatcharoenwitthaya P, Lindor KD (2007) Role of radiologic modalities in the management of non-alcoholic steatohepatitis. Clin Liver Dis 11:37–54
Hamer OW, Aguirre DA, Casola G, Lavine JE, Woenckhaus M, Sirlin CB (2006) Fatty liver: imaging patterns and pitfalls. Radiographics 26:1637–1653
Joseph AE, Saverymuttu SH (1991) Ultrasound in the assessment of diffuse parenchymal liver disease. Clin Radiol 44:219–221
Whiting P, Rutjes AW, Reitsma JB, Bossuyt PM, Kleijnen J (2003) The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol 3:25
Limanond P, Raman SS, Lassman C, Sayre J, Ghobrial RM, Busuttil RW, Saab S, Lu DS (2004) Macrovesicular hepatic steatosis in living related liver donors: correlation between CT and histologic findings. Radiology 230:276–280
van Werven JR, Hoogduin JM, Nederveen AJ, van Vliet AA, Wajs E, Vandenberk P, Stroes ES, Stoker J (2009) Reproducibility of 3.0 Tesla magnetic resonance spectroscopy for measuring hepatic fat content. J Magn Reson Imaging 30:444–448
Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH (2005) Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 58:982–990
Glas AS, Lijmer JG, Prins MH, Bonsel GJ, Bossuyt PM (2003) The diagnostic odds ratio: a single indicator of test performance. J Clin Epidemiol 56:1129–1135
Bahl M, Qayyum A, Westphalen AC, Noworolski SM, Chu PW, Ferrell L, Tien PC, Bass NM, Merriman RB (2008) Liver steatosis: investigation of opposed-phase T1-weighted liver MR signal intensity loss and visceral fat measurement as biomarkers. Radiology 249:160–166
Caturelli E, Squillante MM, Andriulli A, Cedrone A, Cellerino C, Pompili M, Manoja ER, Rapaccini GL (1992) Hypoechoic lesions in the ‘bright liver’: a reliable indicator of fatty change. A prospective study. Gastroenterol Hepatol 7:469–472
Chen C-H, Lin S-T, Yang C-C, Yeh Y-H, Kuo C-L, Nien C-K (2008) The accuracy of sonography in predicting steatosis and fibrosis in chronic hepatitis C. Dig Dis Sci 53:1699–1706
Cho CS, Curran S, Schwartz LH, Kooby DA, Klimstra DS, Shia J, Munoz A, Fong Y, Jarnagin WR, DeMatteo RP, Blumgart LH, D’Angelica MI (2008) Preoperative radiographic assessment of hepatic steatosis with histologic correlation. J Am Coll Surg 206:480–488
Crum-Cianflone N, Dilay A, Collins G, Asher D, Campin R, Medina S, Goodman Z, Parker R, Lifson A, Capozza T, Bavaro M, Hale B, Hames C (2009) Nonalcoholic fatty liver disease Among HIV-infected persons. J Acquir Immune Defic Syndr 50:464–473
D’Assignies G, Ruel M, Khiat A, Lepanto L, Chagnon M, Kauffmann C, Tang A, Gaboury L, Boulanger Y (2009) Noninvasive quantitation of human liver steatosis using magnetic resonance and bioassay methods. Eur Radiol 19:2033–2040
Dasarathy S, Dasarathy J, Khiyami A, Joseph R, Lopez R, McCullough AJ (2009) Validity of real time ultrasound in the diagnosis of hepatic steatosis: a prospective study. J Hepatol 51:1061–1067
de Korte PJ, van der Loos TL, van den Tweel JG, Cremers PT, Veldhuijzen van Zanten GO, Lustermans FA (1986) Interpretation of a ‘bright’ liver in ultrasound examination. Neth J Med 29:5–7
de Moura Almeida A, Cotrim HP, Barbosa DB, de Athayde LG, Santos AS, Bitencourt AG, de Freitas LA, Rios A, Alves E (2008) Fatty liver disease in severe obese patients: diagnostic value of abdominal ultrasound. World J Gastroenterol 14:1415–1418
Dietrich CF, Wehrmann T, Zeuzem S, Braden B, Caspary WF, Lembcke B (1999) Analysis of hepatic echo patterns in chronic hepatitis C. Ultraschall Med 20:9–14
Friedrich-Rust M, Muller C, Winckler A, Kriener S, Herrmann E, Holtmeier J, Poynard T, Vogl TJ, Zeuzem S, Hammerstingl R, Sarrazin C (2010) Assessment of liver fibrosis and steatosis in PBC with FibroScan, MRI, MR-spectroscopy, and Serum Markers. J Clin Gastroenterol 44:58–65
Graif M, Yanuka M, Baraz M, Blank A, Moshkovitz M, Kessler A, Gilat T, Weiss J, Walach E, Amazeen P, Irving CS (2000) Quantitative estimation of attenuation in ultrasound video images: correlation with histology in diffuse liver disease. Invest Radiol 35:319–324
Hamaguchi M, Kojima T, Itoh Y, Harano Y, Fujii K, Nakajima T, Kato T, Takeda N, Okuda J, Ida K, Kawahito Y, Yoshikawa T, Okanoue T (2007) The severity of ultrasonographic findings in nonalcoholic fatty liver disease reflects the metabolic syndrome and visceral fat accumulation. Am J Gastroenterol 102:2708–2715
Hepburn MJ, Vos JA, Fillman EP, Lawitz EJ (2005) The accuracy of the report of hepatic steatosis on ultrasonography in patients infected with hepatitis C in a clinical setting: a retrospective observational study. BMC Gastroenterol 5:14
Hirche TO, Ignee A, Hirche H, Schneider A, Dietrich CF (2007) Evaluation of hepatic steatosis by ultrasound in patients with chronic hepatitis C virus infection. Liver Int 27:748–757
Hultcrantz R, Gabrielsson N (1993) Patients with persistent elevation of aminotransferases: investigation with ultrasonography, radionuclide imaging and liver biopsy. J Intern Med 233:7–12
Iwasaki M, Takada Y, Hayashi M, Minamiguchi S, Haga H, Maetani Y, Fujii K, Kiuchi T, Tanaka K (2004) Noninvasive evaluation of graft steatosis in living donor liver transplantation. Transplantation 78:1501–1505
Joseph AE, Saverymuttu SH, al-Sam S, Cook MG, Maxwell JD (1991) Comparison of liver histology with ultrasonography in assessing diffuse parenchymal liver disease. Clin Radiol 43:26–31
Kichian K, McLean R, Gramlich LM, Bailey RJ, Bain VG (2003) Nonalcoholic fatty liver disease in patients investigated for elevated liver enzymes. Can J Gastroenterol 17:38–42
Kim DY, Park SH, Lee SS, Kim HJ, Kim SY, Kim MY, Lee Y, Kim TK, Khalili K, Bae MH, Lee JY, Lee SG, Yu ES (2010) Contrast-enhanced computed tomography for the diagnosis of fatty liver: prospective study with same-day biopsy used as the reference standard. Eur Radiol 20:359–366
Kim SH, Jeong ML, Jong HK, Kwang GK, Joon KH, Kyoung HL, Seong HP, Yi N-J, Suh K-S, Su KA, Young JK, Kyu RS, Hye SL, Byung IC (2005) Appropriateness of a donor liver with respect to macrosteatosis: application of artificial neural networks to US images—Initial experience. Radiology 234:793–803
Kim SH, Lee JM, Han JK, Lee JY, Lee KH, Han CJ, Jo JY, Yi NJ, Suh KS, Shin KS, Jo SY, Choi BI (2006) Hepatic macrosteatosis: predicting appropriateness of liver donation by using MR imaging–correlation with histopathologic findings. Radiology 240:116–129
Krššák M, Hofer H, Wrba F, Meyerspeer M, Brehm A, Lohninger A, Steindl-Munda P, Moser E, Ferenci P, Roden M (Epub 2009) Non-invasive assessment of hepatic fat accumulation in chronic hepatitis C by (1)H magnetic resonance spectroscopy. Eur J Radiol. doi:10.1016/j.ejrad.2009.03.062
Kutcher R, Smith GS, Sen F, Gelman SF, Mitsudo S, Thung SN, Reinus JF (1998) Comparison of sonograms and liver histologic findings in patients with chronic hepatitis C virus infection. J Ultrasound Med 17:321–325
Lee JY, Kim KM, Lee SG, Yu E, Lim YS, Lee HC, Chung YH, Lee YS, Suh DJ (2007) Prevalence and risk factors of non-alcoholic fatty liver disease in potential living liver donors in Korea: a review of 589 consecutive liver biopsies in a single center. J Hepatol 47:239–244
Lee SW, Park SH, Kim KW, Choi EK, Shin YM, Kim PN, Lee KH, Yu ES, Hwang S, Lee SG (2007) Unenhanced CT for assessment of macrovesicular hepatic steatosis in living liver donors: comparison of visual grading with liver attenuation index. Radiology 244:479–485
Lossner C, Cuno S, Kleine S, Kleine FD (1988) Value of ultrasound tomography in the diagnosis and follow-up of fatty liver. [German]. Dtsch Z Verdau Stoffwechselkr 48:22–26
Mathiesen UL, Franzen LE, Aselius H, Resjo M, Jacobsson L, Foberg U, Fryden A, Bodemar G (2002) Increased liver echogenicity at ultrasound examination reflects degree of steatosis but not of fibrosis in asymptomatic patients with mild/moderate abnormalities of liver transaminases. Dig Liver Dis 34:516–522
McPherson S, Jonsson JR, Cowin GJ, O’Rourke P, Clouston AD, Volp A, Horsfall L, Jothimani D, Fawcett J, Galloway GJ, Benson M, Powell EE (2009) Magnetic resonance imaging and spectroscopy accurately estimate the severity of steatosis provided the stage of fibrosis is considered. J Hepatol 51:389–397
Mennesson N, Dumortier J, Hervieu V, Milot L, Guillaud O, Scoazec JY, Pilleul F (2009) Liver steatosis quantification using magnetic resonance imaging: a prospective comparative study with liver biopsy. J Comput Assist Tomogr 33:672–677
O’Rourke TR, Welsh FK, Tekkis PP, Lyle N, Mustajab A, John TG, Peppercorn D, Rees M (2009) Accuracy of liver-specific magnetic resonance imaging as a predictor of chemotherapy-associated hepatic cellular injury prior to liver resection. Eur J Surg Oncol 35:1085–1091
Orlacchio A, Bolacchi F, Cadioli M, Bergamini A, Cozzolino V, Angelico M, Simonetti G (2008) Evaluation of the severity of chronic hepatitis C with 3-T1H-MR spectroscopy. AJR Am J Roentgenol 190:1331–1339
Palmentieri B, de Sio I, La Mura V, Masarone M, Vecchione R, Bruno S, Torella R, Persico M (2006) The role of bright liver echo pattern on ultrasound B-mode examination in the diagnosis of liver steatosis. Dig Liver Dis 38:485–489
Park SH, Kim PN, Kim KW, Lee SW, Yoon SE, Park SW, Ha HK, Lee MG, Hwang S, Lee SG, Yu ES, Cho EY (2006) Macrovesicular hepatic steatosis in living liver donors: use of CT for quantitative and qualitative assessment. Radiology 239:105–112
Perez NE, Siddiqui FA, Mutchnick MG, Dhar R, Tobi M, Ullah N, Saksouk FA, Wheeler DE, Ehrinpreis MN (2007) Ultrasound diagnosis of fatty liver in patients with chronic liver disease: a retrospective observational study. J Clin Gastroenterol 41:624–629
Rinella ME, Alonso E, Rao S, Whitington P, Fryer J, Abecassis M, Superina R, Flamm SL, Blei AT (2001) Body mass index as a predictor of hepatic steatosis in living liver donors. Liver Transplant 7:409–414
Rinella ME, McCarthy R, Thakrar K, Finn JP, Rao SM, Koffron AJ, Abecassis M, Blei AT (2003) Dual-echo, chemical shift gradient-echo magnetic resonance imaging to quantify hepatic steatosis: implications for living liver donation. Liver Transplant 9:851–856
Saadeh S, Younossi ZM, Remer EM, Gramlich T, Ong JP, Hurley M, Mullen KD, Cooper JN, Sheridan MJ (2002) The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 123:745–750
Saverymuttu SH, Joseph AE, Maxwell JD (1986) Ultrasound scanning in the detection of hepatic fibrosis and steatosis. Br Med J Clin Res Ed 292:13–15
Tobari M, Hashimoto E, Yatsuji S, Torii N, Shiratori K (2009) Imaging of nonalcoholic steatohepatitis: advantages and pitfalls of ultrasonography and computed tomography. Intern Med 48:739–746
Webb M, Yeshua H, Zelber-Sagi S, Santo E, Brazowski E, Halpern Z, Oren R (2009) Diagnostic value of a computerized hepatorenal index for sonographic quantification of liver steatosis. AJR Am J Roentgenol 192:909–914
Yajima Y, Ohta K, Narui T, Abe R, Suzuki H, Ohtsuki M (1983) Ultrasonographical diagnosis of fatty liver: significance of the liver-kidney contrast. Tohoku J Exp Med 139:43–50
Yamashiki N, Sugawara Y, Tamura S, Kaneko J, Matsui Y, Togashi J, Ohki T, Yoshida H, Omata M, Makuuchi M, Kokudo N (2009) Noninvasive estimation of hepatic steatosis in living liver donors: usefulness of visceral fat area measurement. Transplantation 88:575–581
Yoshimitsu K, Kuroda Y, Nakamuta M, Taketomi A, Irie H, Tajima T, Hirakawa M, Ishigami K, Ushijima Y, Yamada T, Honda H (2008) Noninvasive estimation of hepatic steatosis using plain CT vs. chemical-shift MR imaging: significance for living donors. J Magn Reson Imaging 28:678–684
Yu W, Hu S, Qi Y, Li B (2009) The correlation between sonographic diagnosis and laparoscopic observations on fatty liver. J Laparoendosc Adv Surg Tech 19:163–169
Strauss S, Gavish E, Gottlieb P, Katsnelson L (2007) Interobserver and intraobserver variability in the sonographic assessment of fatty liver. Am J Roentgenol 189:W320–W323
Ma X, Holalkere NS, Kambadakone RA, Mino-Kenudson M, Hahn PF, Sahani DV (2009) Imaging-based quantification of hepatic fat: methods and clinical applications. Radiographics 29:1253–1277
Borra RJ, Salo S, Dean K, Lautamaki R, Nuutila P, Komu M, Parkkola R (2009) Nonalcoholic fatty liver disease: rapid evaluation of liver fat content with in-phase and out-of-phase MR imaging. Radiology 250:130–136
Guiu B, Petit JM, Loffroy R, Ben SD, Aho S, Masson D, Hillon P, Krause D, Cercueil JP (2009) Quantification of liver fat content: comparison of triple-echo chemical shift gradient-echo imaging and in vivo proton MR spectroscopy. Radiology 250:95–102
Guiu B, Loffroy R, Petit JM, Aho S, Ben SD, Masson D, Hillon P, Cercueil JP, Krause D (2009) Mapping of liver fat with triple-echo gradient echo imaging: validation against 3.0-T proton MR spectroscopy. Eur Radiol 19:1786–1793
Irwan R, Edens MA, Sijens PE (2008) Assessment of the variations in fat content in normal liver using a fast MR imaging method in comparison with results obtained by spectroscopic imaging. Eur Radiol 18:806–813
Kim H, Taksali SE, Dufour S, Befroy D, Goodman TR, Petersen KF, Shulman GI, Caprio S, Constable RT (2008) Comparative MR study of hepatic fat quantification using single-voxel proton spectroscopy, two-point dixon and three-point IDEAL. Magn Reson Med 59:521–527
O’Regan DP, Callaghan MF, Wylezinska-Arridge M, Fitzpatrick J, Naoumova RP, Hajnal JV, Schmitz SA (2008) Liver fat content and T2*: simultaneous measurement by using breath-hold multiecho MR imaging at 3.0 T–feasibility. Radiology 247:550–557
Reeder SB, Robson PM, Yu H, Shimakawa A, Hines CD, McKenzie CA, Brittain JH (2009) Quantification of hepatic steatosis with MRI: the effects of accurate fat spectral modeling. J Magn Reson Imaging 29:1332–1339
Yokoo T, Bydder M, Hamilton G, Middleton MS, Gamst AC, Wolfson T, Hassanein T, Patton HM, Lavine JE, Schwimmer JB, Sirlin CB (2009) Nonalcoholic fatty liver disease: diagnostic and fat-grading accuracy of low-flip-angle multiecho gradient-recalled-echo MR imaging at 1.5 T. Radiology 251:67–76
Szczepaniak LS, Nurenberg P, Leonard D, Browning JD, Reingold JS, Grundy S, Hobbs HH, Dobbins RL (2005) Magnetic resonance spectroscopy to measure hepatic triglyceride content: prevalence of hepatic steatosis in the general population. Am J Physiol Endocrinol Metab 288:E462–468
Guiu B, Loffroy R, Hillon P, Petit JM (2009) Magnetic resonance imaging and spectroscopy for quantification of hepatic steatosis: urgent need for standardization! J Hepatol 51:1082–1083
Kleiner DE, Brunt EM, Van NM, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A, Yeh M, McCullough AJ, Sanyal AJ (2005) Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41:1313–1321
Acknowledgements
J.G. Daams (clinical librarian) for the electronic database search. L. Alvarez-Herrero, S.I. Goncalves, C. Lavini of Academic Medical Center, University of Amsterdam, The Netherlands and T. Takahara of University Medical Center Utrecht, The Netherlands for translating articles.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
Details of search strategy (DOC 45 kb)
ESM 2
US imaging features (DOC 62 kb)
ESM 3
CT imaging features (DOC 54 kb)
ESM 4
MR imaging features (DOC 49 kb)
ESM 5
1H-MRS imaging features (DOC 36 kb)
ESM 6
Characteristics of liver biopsy examination (DOC 76 kb)
ESM 7
US accuracy (DOC 192 kb)
ESM 8
CT accuracy (DOC 129 kb)
ESM 9
MRI accuracy (DOC 107 kb)
ESM 10
1H-MRS accuracy (DOC 74 kb)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Bohte, A.E., van Werven, J.R., Bipat, S. et al. The diagnostic accuracy of US, CT, MRI and 1H-MRS for the evaluation of hepatic steatosis compared with liver biopsy: a meta-analysis. Eur Radiol 21, 87–97 (2011). https://doi.org/10.1007/s00330-010-1905-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-010-1905-5