Abstract
As the climate warms, boreal fish species are expected to expand into the Arctic domain. Though water temperature is an important factor driving expansion of aquatic species, other variables may play a critical role in restricting those movements. Continuous darkness during the Arctic polar night has been suggested to impair foraging in visually searching boreal fish and may thus limit their northward expansion. We discuss feeding and diet composition of co-existing polar cod (Boreogadus saida), Atlantic cod (Gadus morhua), and haddock (Melanogrammus aeglefinus) during the polar night. The data presented cover January sampling during five consecutive years (2012–2016) in Svalbard fjords (78–80°N). Across species, more than 70% of individuals were feeding. Few stomachs were well filled, suggesting an overall moderate foraging activity or success. The endemic polar cod had the highest frequency of empty stomachs (40%), while Atlantic cod (25% empty) and haddock (21% empty) had fed most extensively. Diet composition indicated opportunistic feeding, with extensive diet overlap of the most abundant prey species (krill, Thysanoessa spp.) during years of highly abundant krill (2014–2015) but reverting to more distinct diets in a year (2016) with lower krill abundance. For all three species, presence of prey items in advanced degrees of digestion indicated feeding activity prior to each annual trawl sampling. The stomach fullness and diet overlap suggest that coexisting and similar sized gadoids feed on the same available prey resources during polar night.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent studies in the waters around Svalbard have documented the northward expansion of invertebrates and fish species (Berge et al. 2005; Fossheim et al. 2015). Boreal fish species are expanding their distribution range (Renaud et al. 2012; Wiedmann et al. 2014a, b) and endemic Arctic species have retreated northwards (Fossheim et al. 2015; Drost et al. 2016). Boreal zooplankton have also shifted their distributions northwards (Buchholz et al. 2012; Kraft et al. 2013; Møller and Nielsen 2020), and changes in zooplankton communities may affect the structure and energy flow of Arctic marine food webs (Falk-Petersen et al. 2007; Drinkwater et al. 2010; Renaud et al. 2015). These community changes at different levels of the food web raise questions as to how recent immigrants will respond to the new environmental conditions they encounter and how new community compositions will affect ecosystem functioning.
Reduced Arctic sea-ice cover is a consequence of a warmer climate (Serreze and Barry 2011). Less sea ice in the Arctic allows more light penetration into the sea and, potentially, higher primary production in open waters (Assmy et al. 2017). Although temperature is often described as the most important factor determining species distribution in Arctic seas (Drinkwater et al. 2010), the light environment at high latitudes has also been proposed to limit species distributions and define northern range limits (Kaartvedt 2008; Varpe et al. 2015; Ljungström et al. 2021). For predators relying on vision to locate prey, light availability mediated by ice cover may influence prey encounter and, thereby, access to food (Langbehn and Varpe 2017), with consequences for the prey community (Langbehn et al. 2023). Fish and seabirds are examples of visually searching predators (Vinyard and O'Brien 1976; Aksnes and Giske 1993; Utne-Palm 2005; Strod et al. 2008) for which light availability in winter influences spatial and temporal distribution as well as feeding (Johansen et al. 2001; Aksnes et al. 2004; Svenning et al. 2007; Varpe and Fiksen 2010).
Despite the paradigm in marine ecology suggesting that the polar night is a period of low biological activity due to the absence of sunlight, feeding by native ‘visually predaceous’ fish and seabirds has been documented in the polar night (Lønne et al. 2015; Berge et al. 2015a, 2015b). The relative foraging abilities and visual acuities of boreal fish species and their Arctic counterparts during the polar night, however, have not been compared. Polar cod (Boreogadus saida) is an Arctic gadoid fish with circumpolar distribution. It is a key species in the Arctic marine food web, and is preyed upon by seabirds, fish such as Atlantic cod (Gadus morhua), and marine mammals (Gjøsæter 2009). The boreal Atlantic cod and haddock (Melanogrammus aeglefinus) occupy sub-Arctic to temperate regions of the north Atlantic, where some amount of daylight is present year-round (Olsen et al. 2010; Bogstad et al. 2015). These species are regularly found at water depths well below significant light penetration, and feeding during winter in fjords at 70ºN has been documented in Atlantic cod (e.g. Falk-Petersen and Hopkins 1981; Klemetsen 1982; Dos Santos and Falk-Petersen 1989). Continuous darkness during the high Arctic polar night, however, represents a new environment for these species. In a warmer Arctic, the polar night may thus be an obstacle for boreal fish species expanding northwards.
Feeding of haddock and Atlantic cod have been extensively studied in the Barents Sea (Jiang and Jørgensen 1996; Bogstad et al. 2015; Eriksen et al. 2021). Both species are considered generalists, with a shift from invertebrates towards fish prey with increasing body size (Kanapathippillai et al. 1994; Jiang and Jørgensen 1996; Dalpadado and Bogstad 2004). They feed on a variety of food including Calanus copepods, while Themisto (amphipods) and small pelagic fish are preferred food for adult polar cod (Orlova et al. 2009; Cusa et al. 2019). Feeding by polar cod, haddock, and Atlantic cod during the early autumn in the waters around Svalbard has been investigated by Renaud et al. (2012), and a limited diet overlap was registered. Few other studies have published data on dietary composition of coexisting Atlantic cod, polar cod, and haddock from the polar night (Geoffroy and Priou 2020).
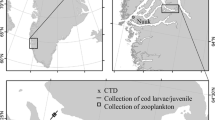
The current warming of the western Svalbard fjords has proceeded rapidly. In particular, Kongsfjorden (Fig. 1), has recently experienced a warmer ocean climate. There, temperatures for much of the year are now well within the physiological temperature ranges for Atlantic cod (www.fishbase.se accessed 9 May 2023), haddock (https://climefish.eu/haddock/ accessed 9 May 2023), and polar cod (Schurmann and Christiansen 1994) for much of the year (Fig. 2). Studies of coexistence and interactions among polar cod, Atlantic cod, and haddock, may serve as a model for more general investigations of food-web interactions in fish communities in warmer, less ice-covered Arctic seas. In the present study, we focus on dietary overlap among gadoids in six Svalbard fjords. We investigate whether similar sized polar cod, Atlantic cod, and haddock differ in terms of feeding intensity, measured as stomach fullness. We compare the dietary composition (ingested prey abundance) of the three species and investigate how polar night diets (weight of ingested prey) of the three species differ during three years of sampling (2014–2016).
Temperature plots from moored observatories between 4 October 2012 and 1 September 2013 in Rijpfjord (upper panel) and Kongsfjord (lower panel). Vertical blue square indicates timing of the January trawl sampling (adapted from Nahrgang et al. 2014 with permission)
Materials and methods
Research area and sampling
From early November to the end of January, the polar night engulfs Svalbard (77–81°N).Footnote 1 The samples for our study were collected in the period 9–24 January 2012–2016 (Table 1). This period corresponds to the mid-winter irradiation minimum when the sun has been continuously below the horizon for approximately two months.
The water masses of the research area (Fig. 1) are dominated by the northward flowing West Spitsbergen Current (WSC), bringing in warm Atlantic water to the area (Walczowski et al. 2012; Pavlov et al. 2013). The shelf and fjord areas of west Spitsbergen are also influenced by the South Cape Current, carrying cold water of Arctic origin from the east of Svalbard (Svendsen et al. 2002). The distribution of Atlantic and Arctic water masses varies, but the general dominance of warm Atlantic water results in reduced ice cover along west and north Spitsbergen (Falk-Petersen et al. 2015; Pavlova et al. 2019). Except for a small area of newly formed ice in the inner part of Isfjord (Billefjord) in 2014, we did not register any sea ice in any of the trawling areas (Fig. 1) during the cruises in 2012–2016.
During the five surveys, 50 trawl hauls (Table 1) were carried out from RV Helmer Hanssen (UiT, The Arctic University of Norway). Demersal trawl hauls (n = 26) were conducted with a 60 m wide aperture Campelen 1800 bottom trawl with a 20 mm cod end mesh size. Pelagic hauls (n = 24) were performed using a Harstad pelagic trawl with a mouth opening of 20 × 20 m (Eriksen et al. 2012) and eight millimetres cod-end mesh size. Each trawl haul lasted approximately 15 min with a towing speed of 3 knots. The depth of pelagic trawl hauls was selected based on the depth of maximum pelagic backscatter on the hull-mounted 38 kHz transducer of the EK60, typically in the 90–130 m depth range.
Samples were collected from six fjords divided across four fjord systems (Fig. 1). Smeerenburgfjord was only visited once, while Rijpfjord, Kongsfjord (including Krossfjord), and Isfjord (including Billefjord) were visited during four of the five surveys (Table 1). The ambient bottom-water temperatures varied from below −1 °C in Rijpfjord in 2012, to 3.9 °C in Kongsfjord in 2016.
Located at 80°20′N on Nordaustlandet, Rijpfjord is the northernmost fjord of our study (Fig. 1). Rijpfjord is an Arctic fjord with limited influence of advected Atlantic water and has sub-zero water temperatures most of the year (Fig. 2). The fjord is often ice-covered from late autumn to July but had open water during our January cruises. Rijpfjord is 40 km long, and up to 12 km wide. The maximum water depth is 276 m.
Smeerenburgfjord on the northwestern Spitsbergen (79°65′N; 11°20′E) (Fig. 1) has three connections to the open sea. The maximum water depth is 220 m, and the water masses in this fjord are influenced by advected warm Atlantic water.
Kongsfjord (79°N; 12°E) is a 20 km long and four to ten km wide fjord located on the west coast of Spitsbergen. Kongsfjord shares a west-facing entrance with Krossfjord (Fig. 1). Kongsfjord has no pronounced sill westward of the trawling areas, allowing unlimited connection to the shelf. Maximum water depth is 400 m. Kongsfjorden is strongly influenced by Atlantic Water from the WSC, providing higher water temperatures compared to the Arctic Rijpfjord (Fig. 2). Twenty-two of the 50 trawl hauls in this study (Table 1) were taken in Kongsfjord/Krossfjord, making it our most investigated fjord.
Isfjord is 170 km long, 24 km at its widest (Fig. 1), and up to 455 m deep. It is located on the west side of Spitsbergen and oriented in a southwest (78°7′N) northeast (78°27′N) direction. The fjord is linked directly to the shelf and slope area along West Spitsbergen as it has no sill, thus permitting inflow of Atlantic Water from the WSC (Nilsen et al. 2008; Forwick and Vorren 2009).
Billefjord (78°40′N) is a silled-fjord at the innermost (eastern) end of the Isfjord system (Fig. 1). A sill extending up to 50 m depth and separating a basin of 180 m depth, limits the water exchange. Billefjord is thus largely unaffected by inflowing Atlantic water, and the bottom water temperature is < −1 °C year-round (Daase 2016). In Billefjord, sea ice normally forms in December-January and lasts until June (Daase 2016). However, except for the 2014 cruise, no sea ice was recorded during any of the January cruises.
Sample processing and analyses
Fish catches in each trawl were sorted to species (or lowest possible taxon) and counted onboard. For large catches of schooling fish, such as polar cod, herring (Clupea harengus), and juvenile redfish (Sebastes sp.), subsamples were collected and counted. Invertebrates were mainly recorded qualitatively, except in 2014 when a one litre sample of the extremely abundant krill (Thysanoessa spp.) collected in Kongsfjord was counted and weighed. A catch per unit effort (standard trawl haul of 15 min at 3 knots) was calculated for the gadoids.
It was not possible to measure all individual fish, but for the polar cod, Atlantic cod, and haddock included in fish stomach analyses, total length (TL) and weight were recorded. The stomachs were recovered by cutting the oesophagus and the intestine behind the pyloric sphincter. The entire stomach was preserved in 70% ethanol, or frozen at −20 °C. An estimate of fullness, judged on a scale from 0 (presumably empty) to 5 (distended) (Haram and Jones 1971), was assigned to each stomach prior to preservation. In the laboratory, the opening of the stomachs frequently revealed that stomachs judged as category 1, only contained water or non-measurable amounts of food material. We therefore have joined the categories 0 and 1 into one joint category, labelled "nearly empty", while the remaining categories of Haram and Jones (1971) were applied.
In the laboratory, stomachs were opened under a 10× magnifying lamp and a binocular, and identifiable stomach content was sorted to lowest possible taxon. For stomachs collected in 2012 and 2013, only presence/absence of categories of prey organisms was recorded. For the stomachs from 2014, 2015, and 2016, both counts and wet weight of each prey taxon was registered. Non-identifiable, highly digested stomach contents was recorded and weighed. On-board, the stomachs collected in 2014–2016 were each given a unique letter and number code, so when processing the stomachs in the laboratory, we did not know which species they belonged to. This was done to avoid presumptions on stomach contents based on knowing which fish species each stomach belonged to. Dietary overlap for 2014–2016 was calculated on wet-weight basis, using Schoeners index (Schoener 1970),
where pxi and pyi are the proportions of food item i in species x and y, and n is the total number of prey items. For the 2014–2016 material, frequency of occurrence of 12 categories of prey organisms was calculated, based on presence/absence, for fish of two size categories: < 25 cm TL including all three species, and > 25 cm TL, comprising only Atlantic cod and haddock. Quantitative (wet weight) diet composition was calculated for the three gadoids from Kongsfjord 2014–2016 per species and size group.
Diet composition of gadoids for 2014–2016 in Kongsfjord was compared for fish < 25 cm TL using a correspondence analysis (CA). The calculations were performed using the open-source program R, version 3.2.5 (R core Team 2016), and the correspondence analysis and plot of dietary overlap was prepared according to an R package from Nenadić and Greenacre (2007). Ellipses were drawn by hand.
Results
Fish catches
In all trawl hauls (demersal and pelagic) the three gadoids made up 53% of the total catch of 58,001 fish (Table 2): polar cod 40%, Atlantic cod 11%, and haddock 2%. At least twenty-five different fish species were registered in the catches (Table 2). Polar cod and Atlantic cod were caught in all fjords for all five sampling years, while no haddock were recorded in Rijpfjord in 2013 and Isfjord in 2015.
Each survey visited different fjords and applied varying combinations of demersal and pelagic trawls (Table 1). Catch per 15 min standard trawl haul for the gadoids is presented in Table 3.
The highest catch per standard trawl haul of polar cod was recorded in Isfjord 2016 (Table 3). The pelagic schooling redfish were mostly < 10 cm TL and became more frequently caught throughout the sampling years. The highest numbers were recorded in 2016 in all fjords (Tables 2 and 3), and the largest catch was taken in Rijpfjord (Tables 2 and 3). The number of juvenile redfish increased markedly from 2012 to 2016 in all fjords (Table 2). The group "shannies" (Stichaeidae) includes several pelagic species of similar body shape and appearance and were found in all fjords all years. Like the Sebastes sp., the catches of shannies increased from 2012 to 2016 in all the fjords.
Herring were all < 10 cm TL, and were mostly caught in Isfjord and Kongsfjord, and only sporadically captured in Rijpfjord. In 2012, 38 herring stomachs were also investigated and they were found to be empty. Capelin (Mallotus villosus) was encountered in nearly all fjords each year (except Isfjord 2012 and 2015). Skates, rays, and sculpins were also regularly found, but in limited numbers (Table 2). In Kongsfjord, one saithe (Pollachius virens), measuring 29.5 cm TL was caught in 2015.
Gadoid catches: size distribution and habitat
Only the gadoids included in the stomach-sampling programme were measured and weighed. Stomach fullness was recorded for a total of 1742 individual gadoids: 763 polar cod, 692 Atlantic cod, and 287 haddock (Fig. 3). Most of the polar cod and haddock caught were < 25 cm TL, while approx. 50% of the Atlantic cod were > 25 cm TL. The largest polar cod in our material was 27.3 cm TL (an individual caught in Kongsfjord 2014). For the diet comparison study (2014–2016 catches), our largest polar cod was 24.7 cm TL. The largest Atlantic cod was 90.0 cm TL, and the largest haddock was 60.0 cm TL (Fig. 3).
Length distribution (three cm intervals) of a Polar cod (n = 763), b Atlantic cod (n = 692), and c haddock (n = 287) included in the polar night stomach sampling programme in Svalbard fjords, 2012–2016. Colour code indicates year of sampling. No polar cod > 30 cm were recorded. Right hand histogram is fish > 30 cm
The use of both demersal and pelagic trawls provides a broad selection of fish from each habitat and fjord. Numbers of each fish species taken in different habitats during the 2014–2016 surveys are presented in Table 4. Most of the polar cod in the stomach analysis programme were taken in demersal trawls (Table 4).
The fish catches varied considerably among fjords, years, and habitats. Yet, the same species co-occurred in all the fjords, but in varying relative abundances. Polar cod, Atlantic cod, and haddock were found in all fjords and all years and were also abundant in subsequent years (Geoffroy and Priou 2020).
Invertebrate catches
Krill (mainly Thysanoessa spp.), amphipods (mainly Themisto spp.), and juvenile northern shrimp (Pandalus borealis) were found in the pelagic trawls. Adult P. borealis and several invertebrate macrofauna taxa were only captured in the demersal trawls. In Kongsfjord in 2014, we took a 1L subsample of the pelagic krill, weighed it, and then counted the number of individuals in the sample. We calculated a weight and number for the entire catch by applying this conversion factor. No quantitative invertebrate catch recordings were made during the other cruises.
Feeding activity and stomach fullness
We found food in the stomachs of most of the fish. However, 40% of the stomachs of the polar cod were classified as "almost empty (< 5% full)" compared to 25 and 22% in the Atlantic cod and haddock (Table 5). Well-filled stomachs (category 4 and 5) were rare in all species (Table 5). During January, no direct sunlight reached any of our investigated fjords, and light conditions were therefore considered equal among all sampling locations (habitats > 90 m water depth).
Dietary composition
Prey organisms from more than sixty taxa were recorded in the 1742 stomachs (Online resource 1 Table 1 and 2). The prey items are grouped in 12 prey categories, and presence/absence of each category in the three predatory species from all fjords 2014–2016 are presented in Table 6. Of the total 244 polar cod stomachs examined, 39.3% contained the prey category "Copepods", while only one haddock had eaten copepods (Table 6). The prey category "Euphausiidae and Mysida" is the most frequently occurring prey for all three predator species for fish < 25 cm, and for Atlantic cod > 25 cm (Table 6). This prey category consisted of an estimated 95% krill of the species Thysanoessa inermis, while the remaining 5% was a mixture of several other krill and mysid species.
Of the large haddock, 63% had eaten polychaetes and haddock had the largest frequency of amphipods in their stomachs (54%, Table 6). The large Atlantic cod had the highest frequency of fish and caridean shrimp in their stomachs. In general, a broad range of prey items at greater than 20% frequency (both pelagic and benthic/hyperbenthic) were found in the polar night diet of all three predator species.
The diet of all three species of gadoids small gadoids (< 25 cm) in Kongsfjord during 2014–2016 was composed of a high proportion of krill (Euphausiids and Mysids) in 2014 and 2015 compared to 2016 (Fig. 4).
All three gadoids fed on fish, and Atlantic cod fed mainly on the other two gadoids as well as some cannibalism. Some prey items were largely confined to a particular predator species, such as copepods for polar cod (2014) and polychaetes for haddock and Atlantic cod (2016).
In all three years (2014–2016), the diet of polar cod and Atlantic cod < 25 cm had more than 50% overlap (Table 7). The overlap in diet was also high for polar cod and haddock < 25 cm in 2014 and 2015 when the most important prey item for both species was krill (Fig. 5). A diet shift was noted in fish < 25 cm in all three species over the three years (Table 8). Small haddock showed a marked dietary overlap between 2014 and 2015, followed by a limited overlap in 2016, with the reduction of krill in the diet and a shift to fish prey for polar cod and to polychaete prey for haddock (Table 8). Overall, Atlantic cod had a larger diet overlap with the polar cod than with haddock. The large Atlantic cod and haddock (> 25 cm) had a lower diet overlap, however the number of large haddock stomachs was limited (Fig. 5).
We noted a change in diet composition over time in Kongsfjord for fish < 25 cm, the size interval for which we have catches of all three species. In 2014 and 2015 all three gadoids fed on abundant mysids and euphausiids, while in 2016, a year of low euphausiid abundance, the three species had different diets. Haddock fed on benthic organisms including bivalves and polychaetes, polar cod on small pelagic crustaceans, and the Atlantic cod was preying on fish (Fig. 6).
Correspondence analysis (CA) of the diet of fish < 25 cm in Kongsfjord, January 2014–2016. 95% confidence ellipses (of the mean) for each species by year are shown: Polar cod (BS, brown), Atlantic cod (GM, green), and haddock (MA, purple). Each dot represents one fish. Prey categories on wet weight. Prey categories abbreviations (ref Table 6). Cop: Copepods, NonGadPel: Non gadoid pelagic fish, Amph: Amphipods, Gad: Gadoids, EuphMys: Euphausids and mysids, Unid Fish: Unidentified fish, Carid: Caridean shrimps, Gastro: Gastropods, Other Crust: Other crustaceans, Polych: Polychaeta, Biv: Bivalves, Unid: Unidentified
We saw a particularly strong overlap in predation on krill in 2014 and 2015. Atlantic cod was the predator with the highest occurrence of fish, including gadoid fish, in the stomachs. For Atlantic cod and haddock > 25 cm, the largest difference observed was the absence of fish prey in haddock, while the large cod had a particularly broad diet which included fish.
The prey items are the "contribution coordinates", the longer the arrow the more important for the ordination axes. The ellipses are the 95% confidence regions for the mean of each species-year group. All the 2016 means and ellipses are to the right, the important prey items pointing to the right, all the 2014 and 2015 means and ellipses are to the left and strongly dominated by the Euphausiids and Mysids prey category in 2014 and 2015, followed by a marked change to more predator species-specific diets in 2016.
Discussion
Across species, more than 70% of individuals were feeding, but few stomachs were well filled, suggesting an overall moderate foraging activity or success. Despite expectations to the contrary (Varpe and Fiksen 2010; Ljungström et al. 2021). we found limited evidence that the low-light conditions in the polar night in six high Arctic fjords inhibited feeding by the predatory gadoids studied. In fact, stomach fullness of Atlantic cod and haddock was equal to or above that of the resident polar cod. During January, no direct sunlight reaches any of our investigated fjords, and light conditions are therefore considered equal among our sampling locations (habitats > 90 m water depth).
Fish communities in the polar night
In 2016, high numbers of polar cod and juvenile redfish were recorded. Redfish were also abundant in winter 2017 (Geoffroy and Priou 2020). The observed increase in catches of juvenile redfish is consistent with observations of strong 2014 and 2015 year-classes in the Barents Sea at large (Eriksen et al. 2016). The gadoids made up 53% of the total numeric catches. Polar cod was the numerically most abundant gadoid in our catches. The saithe, which is a gadoid fish with its main distribution area south of approx. 73°N (Olsen et al. 2010), was recorded once in Kongsfjord in 2015. This recording is, together with one observation from outside the Smeerenburg fjord (Wienerroither et al. 2011), the northernmost documented records of this species, and is considered a further indication of the ongoing northwards expansion of boreal gadoids.
The pelagic trawl hauls were carried out at 90–130 m water depth. The maximum depth of the fjords varies from 180 to 455 m. This means that shifts in foraging habitat will require a vertical swimming distance of 90–350 m. For gadoids > 25 cm TL, this seems to be well within size dependent swimming abilities (Pálsson and Thorsteinsson 2003). Hence, in contrast to the deep basins of the Arctic Ocean, both pelagic and demersal habitats are available as feeding areas for the large piscivorous fish in our study area.
Feeding intensity during the polar night
We found no evidence to suggest that boreal gadoids were less efficient in feeding during the polar night than the native polar cod. Initially we hypothesized that boreal gadoids expanding into the Arctic would have difficulties detecting prey in the dark, and thereby would be disadvantaged compared to their Arctic counterpart, the polar cod. This hypothesis, is primarily based on visual search models for pelagic species (e.g. Varpe and Fiksen 2010), which may not be applicable to demersal and epibenthopelagic gadoids such as Atlantic cod and haddock (Mecklenburg et al. 2018, Jönsson et al. 2014). Geoffroy and Priou (2020) examined the stomach contents of boreal species during the polar night in Svalbard between 2016 and 2018 and demonstrated that, while gadoids all had less than 10% of empty stomachs, capelin (M. villosus), herring (C. harengus), and beaked redfish (Sebastes mentella) all had > 70% of empty stomachs. This suggests that some boreal species may in fact exhibit lower feeding during polar night. For these species, a combination of factors including seasonal food availability, timing of life- and annual-cycle events, and low irradiance may explain the high proportion of empty stomachs of non-gadoid pelagic fishes during the polar night.
Both boreal gadoids are known to consume considerable benthic prey (Planque et al. 2014). Whereas relative visual acuities of the three species of gadoids are not known, both Atlantic cod and haddock exhibit ontogenic behavioural and physiological changes whereby they become more sensitive to low levels of light as they get older (Trippel and Neil 2003; Vollset et al. 2011; Valen et al. 2014). Ultimately, they are more responsive and feed better at very low light levels than at higher intensities (Huse 1994; Meager et al. 2010).
It is unclear why polar cod in this study had lower gut fullness than its confamilials, but this may be a phenomenon linked to winter physiology or to the availability of their preferred prey (Cusa et al. 2019). First, at temperatures near zero degrees, gut evacuation is prolonged in polar cod, and feeding does not take place on a daily basis (Sæther et al. 1999). Second, polar cod spawn in winter. The smaller Atlantic cod (< 25 cm) were non-maturing juveniles with no developing gonads, while polar cod down to approx. 10 cm TL were observed having developing gonads during our winter surveys. The gonads occupy considerable space in the body cavity and may contribute to a lower fulness index for polar cod compared to the other two species (Table 7 and Nahrgang et al. 2014).
It is also important to note that whereas vision in many fish species is a key element of their ability to locate and capture prey, it is but one of a variety of senses employed. Foraging efficiency is related to spatial scale of the combination of a predator's sensory abilities (LaScala-Gruenewald et al. 2019), including motion detection (via the lateral line), and chemosensation ('taste', via chemoreceptors and other sensory structures). For example, both G. morhua and M. aeglefinus have barbels, that can help detect prey in the dark at some spatial scales. At larger scales, just choosing appropriate feeding habitats/micro-habitats using cues such as hydrographic parameters or distance from the seafloor can situate the predator in an area that should increase its chances of foraging success.
Prey selectivity and potential for competition
Distribution of top predators such as large Atlantic cod is mainly governed by the availability of prey, within the physiological limits set by the abiotic environment. Prey organisms, on the other hand, may seek shelter in habitats difficult to access by predators. For example, polar cod seeks shelter from pinniped predators in crevices and cracks in the sea ice (Gradinger and Bluhm 2004) or at depth (Benoit et al. 2010), and gammarid amphipods such as Gammarus wilkitskii essentially live within the sea ice. No sea ice was observed during the January cruises so hiding in the ice was thus not an option. The foraging situation in Svalbard fjords was more comparable to the one recorded during winter at 70°N in mainland Norway where cod was either found feeding on pelagic aggregations of krill at specific depths, or on single target prey close to the bottom (Falk-Petersen and Hopkins 1981; Dos Santos and Falk-Petersen 1989).
Our catch data reflect the availability of the various prey categories to the predators. Gadoids are presumably selecting individual prey items, compared to, e.g. herring and mackerel (Scomber scombrus), applying a filtering feeding strategy in dense schools of prey (Skaret et al. 2015; Bachiller et al. 2016). Further, we found that krill, at least in 2014, was the most important prey in all species. In a study in fjords at 70°N in northern Norway, Falk-Petersen and Hopkins (1981) and Dos Santos and Falk-Petersen (1989) found that Atlantic cod was feeding on pelagic aggregations of krill during mid-winter. In their study, the Atlantic cod seemed to apply filter feeding, while in fjords with no defined krill layers, the Atlantic cod picked single target preys. They further noted that Atlantic cod filter-feeding on krill layers had a stomach fullness of 60–100%. Their studies suggest that it is prey availability which is the most important factor for feeding strategy and success during the polar night, and not the light conditions.
The diet of the three gadoids in Kongsfjord varied considerably from 2014 to 2016. High krill abundance in 2014 and 2015 led to considerable overlap in diets of the gadoids < 25 cm during these years. There is no indication, however, that krill abundance was limiting, and though it is unlikely, it remains unclear whether dietary overlap during such a period of high krill abundance indicates competition. All three species showed a marked change in major type of prey. In 2016, haddock had changed from a diet focussed on krill to a more diverse diet dominated by amphipods and polychaetes, and polar cod and Atlantic cod had switched to diets containing more fish. With the high densities of krill reported both from our own 2014 trawl sampling, and from the upper 30 m of the water column, reported by Grenvald et al. (2016), it seems likely that all three gadoids were actively targeting pelagic krill as their main prey. The abundance of krill in these fjords increased in the period from 2012 and peaked in 2014 and 2015 (Grenvald et al. 2016), but has decreased in recent years and is now on a low level (authors’ own observations). The increase in the number of krill in Kongsfjord can be explained partly by the large influx of Atlantic water in 2014 and 2015 (Tverberg et al. 2019). The importance of krill in fish diets from these fjords has, however, decreased since 2015 and was particularly low in 2022 (Stig Falk-Petersen and Maxime Geoffroy personal observations).
Despite differences in orientation of the mouth, where polar cod has a mouth pointing upwards for pelagic feeding and mouths of the other two species point slightly downwards for demersal feeding, the three gadoids are similar in bodily design and appearance (online resource 2). This means that a given prey item of a specific size is most likely available to any of the three predator species of the same body size, regardless of how the prey is detected. Renaud et al. (2012) found no strong interspecific diet overlap among coexisting juvenile haddock, juvenile Atlantic cod (< 10 cm TL), and polar cod (< 13 cm TL) around Svalbard during the light part of the year. Here, the interspecific overlap in fish < 25 cm TL was highest among polar cod and Atlantic cod. Haddock and Atlantic cod had higher dietary overlap than haddock had with polar cod. Atlantic cod is thus a potential competitor of polar cod during the first years of life, and the larger individuals (> 25 cm TL) are predators feeding on both haddock and polar cod, as well as on other Atlantic cod. We found gadoids in one third of the stomachs of the larger cod. The importance of diet composition during polar night, as a contribution to the cod's total annual food intake, has to be evaluated in combination with the degree of stomach fullness. The latter was generally low, and a varied diet consumed in small amounts during the polar night may not be the major factor determining success or failure in the competition among the three species.
Conclusion
For all three gadoid species, food was found in more than 70% of the stomachs, but very few stomachs were well filled, suggesting a continuous but moderate feeding activity during the polar night. For all three species, prey items in advanced degrees of digestion were found in the stomachs, indicating feeding activity prior to each sampling. The two boreal species do not feed less than their Arctic counterpart, the polar cod.
The diet composition indicated opportunistic feeding, including some benthic feeding, with extensive diet overlap of the abundant prey species (krill) among all three gadoid predators in the two "krill years" in this study. In 2016 when krill populations had decreased, fish reverted to other prey, and exhibited lower dietary overlap.
Teleost prey were frequently found in the stomach of larger (> 25 cm) Atlantic cod, and in smaller specimens in 2015. Overlap in distribution and food preferences indicate a larger potential for competition between young Atlantic cod and polar cod, and less competition between haddock and polar cod. Atlantic cod was also an important predator of polar cod, whereas haddock had a more varied diet that included less fish. Polar night feeding success in Atlantic cod and haddock may contribute to increased interspecific competition with the polar cod, and perhaps predation upon polar cod, in a warmer Arctic.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information (online resource 1 and 2). Should any of the raw data files underlying Figs. 3, 4 or 5 be needed they are available from the corresponding author upon reasonable request.
Notes
Civil Polar night, implying continuous absence of sunlight, occurs when the sun is more than six degrees below the horizon. At 80°N this condition lasts from Nov 11 to Jan 30.
References
Aksnes DL, Giske J (1993) A theoretical model of aquatic visual feeding. Ecol Model 67:233–250
Aksnes DL, Nejstgaard J, Sædberg E, Sørnes T (2004) Optical control of fish and zooplankton populations. Limnol Oceanogr 49:233–238
Assmy P, Fernández-Méndez M et al (2017) Leads in Arctic pack ice enable early phytoplankton blooms below snow-covered sea ice. Sci Rep 7:40850. https://doi.org/10.1038/srep40850
Bachiller E, Skaret G, Nøttestad L, Slotte A (2016) Feeding ecology of northeast Atlantic mackerel Norwegian Spring-Spawning Herring and Blue Whiting in the Norwegian Sea. PLoS One. https://doi.org/10.1371/journal.pone.0149238
Benoit D, Simard Y, Gagné J, Geoffroy M, Fortier L (2010) From polar night to midnight sun: photoperiod, seal predation, and the diel vertical migrations of polar cod (Boreogadus saida) under landfast ice in the Arctic Ocean. Polar Biol 33:1505–1520. https://doi.org/10.1007/s00300-010-0840-x
Berge J, Nilsen F, Gulliksen B, Slagstad D (2005) Ocean temperature oscillations enable reappearance of blue mussels (Mytilus edulis) in Svalbard after a 1000 year absence. Mar Ecol Prog Ser 303:167–175. https://doi.org/10.3354/meps303167
Berge J, Daase M, Renaud PE, Ambrose WG Jr, Darnis G et al (2015a) Unexpected levels of biological activity during the polar night offer new perspectives on a warming Arctic. Curr Biol 25:1–7. https://doi.org/10.1016/j.cub.2015.08.024
Berge J, Renaud PE, Darnis G, Cottier F, Last K et al (2015b) In the dark; a review of ecosystem processes during the Arctic polar night. Prog Oceanogr 139:258–271. https://doi.org/10.1016/j.pocean.2015.08.005
Bogstad B, Gjøsæter H, Haug T, Lindstrøm U (2015) A review of the battle for food in the Barents Sea; cod vs. marine mammals. Front Ecol Evol 3:1–17. https://doi.org/10.3389/fevo.2015.00029
Buchholz F, Werner T, Buchholz C (2012) First observation of krill spawning in the high Arctic Kongsfjorden, west Spitsbergen. Polar Biol 35:1273–1279. https://doi.org/10.1007/s00300-012-1186-3
Cusa M, Berge J, Varpe Ø (2019) Seasonal shifts in feeding patterns: Individual and population realized specialization in a high Arctic fish. Ecol Evol 9:11112–11121. https://doi.org/10.1002/ece3.5615
Daase M (2016) Cruise Report Polar Night Cruise 2016. University of Tromsø
Dalpadado P, Bogstad B (2004) Diet of juvenile cod (age 0–2) in the Barents Sea in relation to food availability and cod growth. Polar Biol 27:140–154. https://doi.org/10.1007/s00300-003-0561-5
Dos Santos J, Falk-Petersen S (1989) Feeding ecology of cod (Gadus morhua L.) in Balsfjorden and Ullsfjorden, Northern Norway 1982–83. J Cons Int Explor Mer 45:190–199
Drinkwater KF, Beaugrand G, Kaeriyama M, Kim S, Ottersen G, Perry RI, Pörtner H-O, Polovina JJ, Takasuka A (2010) On the processes linking climate to ecosystem changes. J Mar Syst 79:374–388. https://doi.org/10.1016/j.jmarsys.2008.12.014
Drost HE, Lo M, Carmack EC, Farrell AP (2016) Acclimation potential of Arctic cod (Boreogadus saida) from the rapidly warming Arctic Ocean. J Exp Biol 219:3114–3125. https://doi.org/10.1242/jeb.140194
Eriksen E, Prozorkevich D, Trofimov A, Howell D (2012) Biomass of scyphozoan jellyfish, and its spatial association with 0-group fish in the Barents Sea. PLoS One 73:e33050. https://doi.org/10.1371/journal.pone.0033050
Eriksen E, Skjoldal HR, Dolgov AV, Strand E, Keulder-Stenevik F et al (2021) Diet and trophic structure of fishes in the Barents Sea: seasonal and spatial variations. Prog Oceanogr. https://doi.org/10.1016/j.pocean.2021.102663
Eriksen E, Prokhorova T, Prozorkevich D (2016) Chapter 5.1 Fish recruitment: fish distribution and abundance/biomass indices. In: Prozorkevich D, Sunnanå K (eds) Survey report from the joint Norwegian/Russian ecosystem survey in the Barents Sea and adjacent waters, August-October 2015. IMR/PINRO Joint Report series, No. 1/2016
Falk-Petersen S, Hopkins CCE (1981) Zooplankton Sound Scattering Layers in North Norwegian fjords: Interactions between fish and krill shoals in a winter situation in Ullsfjorden and Øksfjorden. Kieler Meeresforsch Sonderh 5:191–201
Falk-Petersen S, Timofeev S, Pavlov V, Sargent JR (2007) Climate variability and possible effects on arctic food chains: the role of Calanus. In: Ørbæk JB, Tombre T, Kallenborn R, Hegseth E, Falk-Petersen S, Hoel AH (eds) Arctic alpine ecosystems and people in a changing environment. Springer, Berlin, pp 147–166
Falk-Petersen S, Pavlov V, Berge J, Cottier F, Kovacs KM, Lydersen C (2015) At the rainbow’s end: high productivity fuelled by winter upwelling along an Arctic shelf. Polar Biol 38:5–11. https://doi.org/10.1007/s00300-014-1482-1
Forwick M, Vorren TO (2009) Late Weichselian and Holocene sedimentary environments and ice rafting in Isfjorden, Spitsbergen. Palaeo Geogr Palaeo Climatol Palaeo Ecol 280:258–274. https://doi.org/10.1016/j.palaeo.2009.06.026
Fossheim M, Primicerio R, Johannesen E, Ingvaldsen RB, Aschan MM, Dolgov AV (2015) Recent warming leads to a rapid borealization of fish communities in the Arctic. Nat Clim Change 5:673–677. https://doi.org/10.1038/NCLIMATE2647
Geoffroy M, Priou P (2020) Fish ecology during the polar night. Adv Polar Ecol 4:181–217. https://doi.org/10.1007/978-3-030-33208-2_7
Gjøsæter H (2009) Commercial fisheries (Fish, seafood marine mammals). In: Sakshaug E, Johnsen G, Kovacs KM (eds) Ecosystem Barents Sea. Tapir Academic Press, Trondheim, pp 373–414
Gradinger R, Bluhm B (2004) In-situ observations on the distribution and behaviour of amphipods and Arctic cod (Boreogadus saida) under the sea ice of the High Arctic Canada Basin. Polar Biol 27:595–603. https://doi.org/10.1007/s00300-004-0630-4
Grenvald JC, Callesen TA, Daase M, Hobbs L, Darnis G, Renaud PE, Cottier F, Nielsen TG, Berge J (2016) Plankton community composition and vertical migration during polar night in Kongsfjorden. Polar Biol 39:1879–1895. https://doi.org/10.1007/s00300-016-2015-x
Haram OJ, Jones JW (1971) Some observations on the food of the gwyniard Coregonus clupeoides pennantii Valenciennes of Llyn Tegid (Lake Bala) North Wales. J Fish Biol 3:287–295
Huse I (1994) Feeding at different illumination levels in larvae of three marine teleost species: cod, Gadus morhua L., plaice, Pleuronectes platessa L., and turbot, Scophthalmus maximus (L.). Aquac Res 25:687–695. https://doi.org/10.1111/j.1365-2109.1994.tb00733.x
Jiang W, Jørgensen T (1996) The diet of haddock (Melanogrammus aeglefinus L.) in the Barents Sea during the period 1984–1991. ICES J Mar Sci 53:11–21. https://doi.org/10.1006/jmsc.1996.0002
Johansen R, Barrett RT, Pedersen T (2001) Foraging strategies of Great Cormorants Phalacrocorax carbo wintering north of the Arctic circle. Bird Study 48:59–67. https://doi.org/10.1080/00063650109461203
Jönsson M, Varpe Ø, Kozlowski T, Berge J, Kröger R (2014) Differences in lens optical plasticity in two gadoid fishes meeting in the Arctic. J Comp Physiol 200:949–957. https://doi.org/10.1007/s00359-014-0941-z
Kaartvedt S (2008) Photoperiod may constrain the effect of global warming in arctic marine systems. J Plankton Res 30:1203–1206. https://doi.org/10.1093/plankt/fbn075
Kanapathippillai P, Berg E, Dos Santos J, Gulliksen B, Pedersen T (1994) The food consumption of cod, Gadus morhua L., in a high-latitude enhancement area. Aquac Fish Manage 25(suppl 1):65–76
Klemetsen A (1982) Food and feeding habits of cod from the Balsfjord, northern Norway during a one year period. J Cons Int Explor Mer 40:101–111
Kraft A, Nöthig EM, Bauerfeind E, Wildish DJ, Pohle GW et al (2013) First evidence of reproductive success in a southern invader indicates possible community shifts among Arctic zooplankton. Mar Ecol Prog Ser 493:291–296. https://doi.org/10.3354/meps10507
Langbehn TJ, Varpe Ø (2017) Sea-ice loss boosts visual search: Fish foraging and changing pelagic interactions in polar oceans. Global Change Biol 23:5318–5330. https://doi.org/10.1111/gcb.13797
Langbehn TJ, Aarflot JM, Freer JJ, Varpe Ø (2023) Visual predation risk and spatial distributions of large Arctic copepods along gradients of sea ice and bottom depth. Limnol Oceanogr. https://doi.org/10.1002/lno.12354
LaScala-Gruenewald DE, Mehta RS, Liu Y, Denny MW (2019) Sensory perception plays a larger role in foraging efficiency than heavy-tailed movement strategies. Ecol Mod 404:69–82. https://doi.org/10.1016/j.ecolmodel.2019.02.015
Ljungström G, Langbehn TJ, Jørgensen C (2021) Light and energetics at seasonal extremes limit poleward range shifts. Nat Clim Change 11:530–536. https://doi.org/10.1038/s41558-021-01045-2
Lønne OJ, Falk-Petersen S, Berge J (2015) Introduction to the special issue on polar night studies conducted onboard RV Helmer Hanssen in the Svalbard area. Polar Biol 38:1–3. https://doi.org/10.1007/s00300-014-1616-5
Meager JJ, Moberg O, Strand E, Utne-Palm AC (2010) Effects of light intensity on visual prey detection by juvenile Atlantic cod (Gadus morhua L.). Mar Freshw Behav Physiol 43:99–108. https://doi.org/10.1080/10236241003798910
Møller EF, Nielsen TG (2020) Borealization of Arctic zooplankton - smaller and less fat zooplankton species in Disko Bay, Western Greenland. Limnol Oceanogr 65:1175–1188. https://doi.org/10.1002/lno.11380
Nahrgang J, Varpe Ø, Korshunova E, Murzina S, Hallanger IG, Vieweg I, Berge J (2014) Gender specific reproductive strategies of an Arctic key species (Boreogadus saida) and implications of climate change. PLoS One 9(5):e98452. https://doi.org/10.1371/journal.pone.0098452
Nenadić O, Greenacre M (2007) Correspondence analysis in R, with two- and three-dimensional graphics: the ca package. J Stat Softw. https://doi.org/10.18637/jss.v020.i03
Nilsen F, Cottier F, Skogseth R, Mattsson S (2008) Fjord–shelf exchanges controlled by ice and brine production: the interannual variation of Atlantic Water in Isfjorden, Svalbard. Cont Shelf Res 28:1838–1853. https://doi.org/10.1016/j.csr.2008.04.015
Olsen E, Aanes S, Mehl S, Holst JC, Aglen A, Gjøsæter H (2010) Cod, haddock, saithe, herring, and capelin in the Barents Sea and adjacent waters: a review of the biological value of the area. ICES J Mar Sci 67:87–101. https://doi.org/10.1093/icesjms/fsp229
Orlova E, Dolgov AV, Rudneva GB, Oganin IA, Konstantinova LK (2009) Trophic relations of capelin Mallotus villosus and polar cod Boreogadus saida in the Barents Sea as a factor of impact on the ecosystem. Deep-Sea Res II 56:2054–2067. https://doi.org/10.1016/j.dsr2.2008.11.016
Pálsson ÓK, Thorsteinsson V (2003) Migration patterns, ambient temperature, and growth of Icelandic cod (Gadus morhua): evidence from storage tag data. Can J Fish Aquat Sci 60:1409–1423. https://doi.org/10.1139/f03-117
Pavlov AK, Tverberg V, Ivanov BV, Nilsen F, Falk-Petersen S, Granskog MA (2013) Warming of Atlantic Water in two west Spitsbergen fjords over the last century (1912–2009). Polar Res 32:11206. https://doi.org/10.3402/polar.v32i0.11206
Pavlova O, Gerland S, Hop H (2019) Changes in sea-ice extent and thickness in Kongsfjorden, Svalbard (2003–2016). In: Hop H, Wiencke C (eds) The ecosystem of Kongsfjorden, Svalbard. Springer, Cham, pp 105–136
Planque B, Primicerio R, Michalsen K, Aschan M, Certain G et al (2014) Who eats whom in the Barents Sea: a food web topology from plankton to whales: ecological archives E095–124. Ecology 95:1430–1430. https://doi.org/10.1890/13-1062.1
R Core Team (2016) R: A Language and Environment for Statistical Computing. Version 3.2.5. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Renaud PE, Berge J, Varpe Ø, Lønne OJ, Nahrgang J et al (2012) Is the poleward expansion by Atlantic cod and haddock threatening native polar cod, Boreogadus saida? Polar Biol 35:401–412. https://doi.org/10.1007/s00300-011-1085-z
Renaud P, Løkken TS, Jørgensen LL, Berge J, Johnson BJ (2015) Macroalgal detritus and food-web subsidies along an Arctic fjord depth-gradient. Front Mar Sci 2:31. https://doi.org/10.3389/fmars.2015.00031
Sæther B-S, Christiansen JS, Jobling M (1999) Gastrointestinal evacuation of particulate matter in polar cod Boreogadus saida. Mar Ecol Prog Ser 188:201–205. https://doi.org/10.3354/meps188201
Schoener TW (1970) Non-synchronous spatial overlap of lizards in patchy habitats. Ecology 60:703–710
Schurmann H, Christiansen JS (1994) Behavioural thermoregulation and swimming activity of two arctic teleosts (subfamily gadinae) - the polar cod (Boreogadus saida) and the navaga (Eleginus navaga). J Thermal Biol 19:207–212. https://doi.org/10.1016/0306-4565(94)90032-9
Serreze M, Barry RG (2011) Processes and impacts of Arctic amplification: a research synthesis. Global Planet Change 77:85–96. https://doi.org/10.1016/j.gloplacha.2011.03.004
Skaret G, Bachiller E, Langøy H, Stenevik EK (2015) Mackerel predation on herring larvae during summer feeding in the Norwegian Sea. ICES J Mar Sci 72:2313–2321. https://doi.org/10.1093/icesjms/fsv087
Strod T, Izhaki I, Arad Z, Katzir G (2008) Prey detection by great cormorant (Phalacrocorax carbo sinensis) in clear and in turbid water. J Exp Biol 211:866–872. https://doi.org/10.1242/jeb.014324
Svendsen H, Beszczynska-Møller A, Hagen JO, Lefauconnier B, Tverberg V, Gerland S, Ørbæk JB et al (2002) The physical environment of Kongsfjorden-Krossfjorden, an Arctic fjord system in Svalbard. Polar Res 21:133–166. https://doi.org/10.3402/polar.v21i1.6479
Svenning MA, Klemetsen A, Olsen T (2007) Habitat and food choice of Arctic charr in Linnevatn on Spitsbergen, Svalbard: the first year –round investigation in a High Arctic lake. Ecol Freshw Fish 16:70–77. https://doi.org/10.1111/j.1600-0633.2006.00183.x
Trippel EA, Neil SRE (2003) Effects of photoperiod and light intensity growth and activity of juvenile haddock (Melanogrammus aeglefinus). Aquaculture 217:633–645. https://doi.org/10.1016/S0044-8486(02)00198-9
Tverberg V, Skogseth R, Cottier F, Sundfjord A, Walczowski W et al (2019) The Kongsfjorden transect: seasonal and inter-annual variability in hydrography. In: Hop H, Wiencke C (eds) The ecosystem of Kongsfjorden, Svalbard. Advances in Polar Ecology, 2. Springer, Cham
Utne-Palm AC (2005) The effect of prey mobility, prey contrast, turbidity and spectral composition on the reaction distance of Gobiusculus flavescens to its planktonic prey. J Fish Biol 54:1244–1258. https://doi.org/10.1111/j.1095-8649.1999.tb02052.x
Valen R, Edvardsen RB, Søviknes AM, Drivenes Ø, Helvik JV (2014) Molecular evidence that only two opsin subfamilies, the blue light- (SWS2) and green light-sensitive (RH2), Drive Color Vision in Atlantic Cod (Gadus Morhua). PLoS One. https://doi.org/10.1371/journal.pone.0115436
Varpe Ø, Fiksen Ø (2010) Seasonal plankton-fish interactions: light regime, prey phenology and herring foraging. Ecology 91:311–318. https://doi.org/10.1890/08-1817.1
Varpe Ø, Daase M, Kristiansen T (2015) A fish-eye view on the new Arctic lightscape. ICES J Mar Sci 72:2532–2538. https://doi.org/10.1093/icesjms/fsv129
Vinyard GL, O’Brien WD (1976) Effects of light and turbidity on reactive distance of bluegill (Lepomis macrochirus). J Fish Res Board Can 33:2845–2849
Vollset KW, Folkvord A, Browman H (2011) Foraging behaviour of larval cod (Gadus morhua) at low light intensities. Mar Biol 158:1125–1133. https://doi.org/10.1007/s00227-011-1635-5
Walczowski W, Piechura J, Goszczko I, Wieczorek P (2012) Changes in Atlantic water properties: an important factor in the European Arctic marine climate. ICES J Mar Sci 69:864–869. https://doi.org/10.1093/icesjms/fss068
Wiedmann MA, Primicerio R, Dolgov A, Ottesen CAM, Aschan M (2014a) Life history variation in Barents Sea fish: implications for sensitivity to fishing in a changing environment. Ecol Evol 4:3596–3611. https://doi.org/10.1002/ece3.1203
Wiedmann MA, Aschan M, Certain G, Dolgov A, Greenacre M, Johannesen E, Planque B, Primicerio R (2014b) Functional diversity of the Barents Sea fish community. Mar Ecol Prog Ser 495:205–218. https://doi.org/10.3354/meps10558
Wienerroither R, Johannesen E, Dolgov A, Byrkjedal I, Bjelland O, Drevetnyak K, Eriksen KB, Høines Å, Langhelle G, Langøy H, Prokhorova T, Prozorkevich D, Wenneck T (2011) Atlas of the Barents Sea Fishes. IMR/PINRO Joint Report Series 1-2011
Acknowledgements
The trawl hauls were all performed on-board the research vessel Helmer Hanssen of UiT, The Arctic University of Norway. The crew and officers on board Helmer Hansen are gratefully acknowledged for skilled handling of equipment and catches. This project was made possible by a grant from the Norwegian Foreign Department to the A-lex project (ref 12/00900), by three grants from the Norwegian Research Council: Arctic ABC (project no 244319), the Marine Night project (NRC 226417) and Deep Impact (project no 300333). E. Stübner is gratefully acknowledged for assistance in analyses of stomach contents, and J. Nahrgang and C. N. Webster are acknowledged for assistance during one or more cruises. M. Greenacre, H. Andrade, and C. Emblow are greatly acknowledged for assistance with statistical analyses and in preparing tables and figures. Akvaplan-niva provided additional financial support.
Author information
Authors and Affiliations
Contributions
LHL: concept and idea, fieldwork, stomach contents analyses, manuscript preparation. MC: concept and idea, fieldwork, stomach contents analyses, data analyses, manuscript preparation. SEN: fieldwork, stomach content analyses, manuscript preparation. JB, PER: concept and idea, manuscript preparation. SFP: manuscript preparation. ØV: concept and idea, fieldwork, manuscript preparation. MG: fieldwork, manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Compliance with ethical standards
All applicable international, national, and institutional guidelines for the care and use of animals were followed. Trawling in the territorial waters of Svalbard was approved by the Norwegian Directorate of Fisheries (permission no 12/17919). The trawling did not yield any bycatch of mammals or birds. The authors declare that they have no conflicts of interest performing this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Larsen, LH., Cusa, M., Eglund-Newby, S. et al. Diets of gadoid fish in Arctic waters of Svalbard fjords during the polar night. Polar Biol 46, 783–799 (2023). https://doi.org/10.1007/s00300-023-03167-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-023-03167-z