Abstract
Key message
Strigolactone has the potential to influence hormone metabolism, in addition to having a role in inhibiting axillary bud elongation, which could be regulated by the expression of phytohormones-related genes.
Abstract
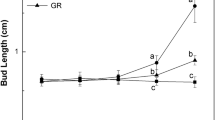
The elongation of axillary buds affects the economic benefits of tobacco. In this study, it was investigated the effect of strigolactone (SL) on the elongation of tobacco axillary buds and its endogenous hormone metabolism and related gene expression by applying the artificial analog of SL, GR24, and an inhibitor of SL synthesis, TIS-108, to the axillary buds. The results showed that the elongation of axillary buds was significantly inhibited by GR24 on day 2 and day 9. Ultra-high-performance liquid-chromatography-mass spectrometry results further showed that SL significantly affected the metabolism of endogenous plant hormones, altering both their levels and the ratios between each endogenous hormone. Particularly, the levels of auxin (IAA), trans-zeatin-riboside (tZR), N6-(∆2-isopentenyl) adenine (iP), gibberellin A4 (GA4), jasmonic acid (JA), and jasmonoyl isoleucine (JA-Ile) were decreased after GR24 treatment on day 9, but the levels of 1-aminocyclopropane-1-carboxylic acid (ACC) and gibberellin A1 (GA1) were significantly increased. Further analysis of endogenous hormonal balance revealed that after the treatment with GR24 on day 9, the ratio of IAA to cytokinin (CTK) was markedly increased, but the ratios of IAA to abscisic acid (ABA), salicylic acid (SA), ACC, JAs, and, GAs were notably decreased. In addition, according to RNA-seq analysis, multiple differentially expressed genes were found, such as GH3.1, AUX/IAA, SUAR20, IPT, CKX1, GA2ox1, ACO3, ERF1, PR1, and HCT, which may play critical roles in the biosynthesis, deactivation, signaling pathway of phytohormones, and the biosynthesis of flavonoids to regulate the elongation of axillary buds in tobacco. This work lays the certain theoretical foundation for the application of SL in regulating the elongation of axillary buds of tobacco.















Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. All data during this study are available from the first author by request.
References
Ahmad P, Abass Ahanger M, Nasser Alyemeni M, Wijaya L, Alam P, Ashraf MA (2018) Mitigation of sodium chloride toxicity in Solanum lycopersicum L. by supplementation of jasmonic acid and nitric oxide. J Plant Interact 13(1):64–72
Balla J, Medveďová Z, Kalousek P, Matiješčuková N, Friml J, Reinöhl V, Procházka S (2016) Auxin flow-mediated competition between axillary buds to restore apical dominance. Sci Rep 6:35955
Barbier FF, Dun EA, Beveridge CA (2017) Apical dominance. Curr Biol 27(17):R864–R865
Barbier FF, Dun EA, Kerr SC, Chabikwa TG, Beveridge CA (2019) An update on the signals controlling shoot branching. Trends Plant Sci 24(3):220–236
Besseau S, Hoffmann L, Geoffroy P, Lapierre C, Pollet B, Legrand M (2007) Flavonoid accumulation in arabidopsis repressed in lignin synthesis affects auxin transport and plant growth. Plant Cell 19(1):148–162
Binenbaum J, Weinstain R, Shani E (2018) Gibberellin localization and transport in plants. Trends Plant Sci 23(5):410–421
Brewer PB, Dun EA, Gui R, Mason MG, Beveridge CA (2015) Strigolactone inhibition of branching independent of polar auxin transport. Plant Physiol 168(4):1820–1829
Carbonnel I, Bennett T, Morffy LY, Stanga JP, Abbas A (2015) SMAX1-LIKE/D53 family members enable distinct MAX2-dependent responses to strigolactone and karrikins in Arabidopsis. Plant Cell 27:3143
Chang S, Chen Y, Jia S, Li Y, Liu K, Lin Z, Wang H, Chu Z, Liu J, Xi C, Zhao H, Han S, Wang Y (2020) Auxin apical dominance governed by the Osasp1-OsTIF1 complex determines distinctive rice caryopses development on different branches. PLoS Genet 16(10):e1009157
Chao WS, Doğramaci M, Horvath DP, Anderson JV, Foley ME (2016) Phytohormone balance and stress-related cellular responses are involved in the transition from bud to shoot growth in leafy spurge. BMC Plant Biol 16:47
Chen XJ, Xia XJ, Guo X, Zhou YH, Shi K, Zhou J, Yu JQ (2016) Apoplastic H2O2 plays a critical role in axillary bud outgrowth by altering auxin and cytokinin homeostasis in tomato plants. New Phytol 211(4):1266–1278
Chen J, Zhang L, Zhu M, Han L, Lv Y, Liu Y, Li P, Jing H, Cai H (2018a) Non-dormant Axillary Bud 1 regulates axillary bud outgrowth in sorghum. J Integr Plant Biol 60(10):938–955
Chen S, Zhou Y, Chen Y, Gu J (2018b) Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics (oxford, England) 34(17):i884–i890
Chen K, Li GJ, Bressan RA, Song CP, Zhu JK, Zhao Y (2020) Abscisic acid dynamics, signaling, and functions in plants. J Integr Plant Biol 62(2):25–54
Cheng X, Ruyter-Spira C, Bouwmeester H (2013) The interaction between strigolactone and other plant hormones in the regulation of plant development. Front Plant Sci 4:199
Dierck R, Dhooghe E, Huylenbroeck JM, Riek JD, Keyser ED, Straeten DV (2016) Response to strigolactone treatment in chrysanthemum axillary buds is influenced by auxin transport inhibition and sucrose availability. Acta Physiol Plant 38:271
Ding N, Qin Q, Wu X, Miller RD, Zaitlin D, Li D, Yang S (2020) Antagonistic regulation of axillary bud outgrowth by the Branched genes in tobacco. Plant Mol Biol 103(1–2):185–196
Duan J, Yu H, Yuan K, Liao Z, Meng X, Jing Y, Liu G, Chu J, Li J (2019) Strigolactone promotes cytokinin degradation through transcriptional activation of Cytokinin oxidase/dehydrogenase 9 in rice. Proc Natl Acad Sci USA 116(28):14319–14324
Dun EA, de Saint GA, Rameau C, Beveridge CA (2012) Antagonistic action of strigolactone and cytokinin in bud outgrowth control. Plant Physiol 158(1):487–498
Faiss M, Zalubìlová J, Strnad M, Schmülling T (1997) Conditional transgenic expression of the ipt gene indicates a function for cytokinins in paracrine signaling in whole tobacco plants. Plant J 12(2):401–415
Feng Lu, Liang X, Zhou Y, Zhang Y, Liu J, Cai M (2020) Functional analysis of Aux/IAAs and SAURs on shoot growth of Lagerstroemia indica through virus-induced gene silencing (VIGS). Forests 11(12):1288
Ferrero M, Pagliarani C, Novák O, Ferrandino A, Cardinale F, Visentin I, Schubert A (2018) Exogenous strigolactone interacts with abscisic acid-mediated accumulation of anthocyanins in grapevine berries. J Exp Bot 69(9):2391–2401
Fujita Y, Yoshida T, Yamaguchi-Shinozaki K (2013) Pivotal role of the AREB/ABF-SnRK2 pathway in ABRE-mediated transcription in response to osmotic stress in plants. Physiol Plant 147(1):15–27
Gao YL, Song ZB, Li WZ, Jiao FC, Wang R, Huang CJ, Li YP, Wang BW (2016) NtBRC1 suppresses axillary branching in tobacco after decapitation. Genet Mol Res 15(4). https://doi.org/10.4238/gmr15049320
Gomez-Roldan V, Fermas S, Brewer PB, Puech-Pagès V, Dun EA, Pillot JP (2008) Strigolactone inhibition of shoot branching. Nature 455(7210):189–194
González-Grandío E, Pajoro A, Franco-Zorrilla JM, Tarancón C, Immink RG, Cubas P (2017) Abscisic acid signaling is controlled by a Branched1/HD-ZIP I cascade in Arabidopsis axillary buds. Proc Natl Acad Sci USA 114(2):E245–E254
Grundmann L, Känel A, Muth J, Beinecke F, Jekat M, Shen Y (2022) Tissue-specific expression of barnase in tobacco delays axillary shoot development after topping. Plant Biotechnol J 20(3):411–413
Hall W, Truchelut GB, Leinweber CL, Herrero FA (1957) Ethylene production by the cotton plant and its effects under experimental and field conditions. Physiol Plant 10(2):306–317
Hayward A, Stirnberg P, Beveridge CA, Leyser O (2009) Interactions between auxin and strigolactone in shoot branching control. Plant Physiol 151(1):400–412
Hedden P, Sponsel VM (2015) A century of gibberellin research. J Plant Growth Regul 34(4):740–760
Hu Q, Zhang S, Huang B (2018) Strigolactone and interaction with auxin regulating root elongation in tall fescue under different temperature regimes. Plant Sci 271:34–39
Ito S, Umehara M, Hanada A, Kitahata N, Hayase H, Yamaguchi S, Asami T (2011) Effects of triazole derivatives on strigolactone levels and growth retardation in rice. PLoS ONE 6(7):e21723
Ito S, Yamagami D, Asami T (2018) Effects of gibberellin and strigolactone on rice tiller bud growth. J Pestic Sci 43(3):220–223
Ivanchenko M, Muday G, Dubrovsky J (2008) Ethylene-auxin interactions regulate lateral root initiation and emergence in Arabidopsis thaliana. Plant J 55(2):335–347
Jiang B, Miao H, Chen S, Zhang SM, Chen FD, Fang WM (2010) The Lateral Suppressor-Like gene, DgLsL, alternated the axillary branching in transgenic chrysanthemum (Chrysanthemum × morifolium) by modulating IAA and GA content. Plant Mol Biol Rep 28:144–151
Jiu ST, Xu Y, Wang JY, Haider MS, Xu JM, Wang L (2022) Molecular mechanisms underlying the action of strigolactone involved in grapevine root development by interacting with other phytohormone signaling. Sci Hortic 293:110709
Kanehisa FM, Tanabe M, Sato Y, Morishima K (2017) KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res 45(D1):D353–D361
Katyayini NU, Rinne PLH, Tarkowská D, Strnad M, van der Schoot C (2020) Dual role of gibberellin in perennial shoot branching: inhibition and activation. Front Plant Sci 11:736
Kerr SC, de Saint Germain A, Dissanayanke IM, Mason MG, Dun EA, Beveridge CA (2020) Hormonal regulation of the BRC1-dependent strigolactone transcriptome involved in shoot branching responses. bioRxiv
King RW, Evans LT, Mander LN, Moritz T, Pharis RP, Twitchin B (2003) Synthesis of gibberellin GA6 and its role in flowering of Lolium temulentum. Phytochemistry 62(1):77–82
Koltai H, Beveridge CA (2013) Strigolactone and the coordinated development of shoot and root//Long-distance systemic signaling and communication in plants. Springer, Berlin, pp 189–204
Kotov AA, Kotova LM, Romanov GA (2021) Signaling network regulating plant branching: recent advances and new challenges. Plant Sci 307:110880
Li ZY, Qiu LH, Yan HF, Zhou HW, Chen RF, Fan YG (2021) Effect of exogenous gibberellin signal on sugarcane tillering and its endogenous hormones. Chinese J Trop Crops 42(10):2942–2951
Li L, Xia T, Li B, Yang H (2022) Hormone and carbohydrate metabolism associated genes play important roles in rhizome bud full-year germination of Cephalostachyum pingbianense. Physiol Plant 174(2):e13674
Liu Y, Xu J, Ding Y, Wang QS, Li GH, Wang SH (2011) Auxin inhibits the outgrowth of tiller buds in rice (Oryza sativa L.) by downregulating OsIPT expression and cytokinin biosynthesis in nodes. Aust J Crop Sci 5:169–174
Liu J, Shi M, Wang J, Zhang B, Li Y, Wang J (2020) Comparative transcriptomic analysis of the development of sepal morphology in tomato (Solanum lycopersicum L.). Int J Mol Sci 21(16):5914
Liu Z, Su J, Luo X, Meng J, Zhang H, Li P (2022a) Nitrogen limits zinc-mediated stimulation of tillering in rice by modifying phytohormone balance under low-temperature stress. Food Energy Secur 11(1):e359. https://doi.org/10.1002/fes3.359
Liu ZL, Meng JR, Sun ZF, Su JK, Luo XY, Song JM (2022b) b) Zinc application after low temperature stress promoted rice tillers recovery: aspects of nutrient absorption and plant hormone regulation. Plant Sci 314:111104
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C (T)) method. Methods 25(4):402-408
Ljung K, Nemhauser JL, Perata P (2015) New mechanistic links between sugar and hormone signalling networks. Curr Opin Plant Biol 25:130–137
Lowe GE, Shepherd M, Rose T (2021) Strigolactone analogue GR24 reduces axillary bud out break and growth in tea tree, Melaleuca alternifolia (Maiden and Betche) Cheel. Adv Hortic Sci 35:399–405
Luan YX, Wang BS, Zhao Q, Ao G, Yu J (2010) Ectopic expression of foxtail millet zip-like gene, SiPf40, in transgenic rice plants causes a pleiotropic phenotype affecting tillering, vascular distribution and root development. Sci China Life Sci 53(12):1450–1458
Luisi A, Lorenzi R, Sorce C (2011) Strigolactone may interact with gibberellin to control apical dominance in pea (Pisum sativum). Plant Growth Regul 65(2):415–419. https://doi.org/10.1007/s10725-011-9603-0
Luo L, Takahashi M, Kameoka H, Qin R, Shiga T, Kanno Y, Seo M, Ito M, Xu G, Kyozuka J (2019) Developmental analysis of the early steps in strigolactone-mediated axillary bud dormancy in rice [J]. Plant J 97(6):1006–1021
Lv J, Chen YQ, Ding AM, Lei B, Yu J, Gao XM (2021) Control of axillary bud growth in tobacco through toxin gene expression system. Sci Rep 11(1):17513
Ma J, Xie L, Zhao Q, Sun Y, Zhang D (2022) Cyclanilide induces lateral bud outgrowth by modulating cytokinin biosynthesis and signalling pathways in apple identified via transcriptome analysis. Int J Mol Sci 23(2):581
Manandhar S, Funnell KA, Woolley DJ, Cooney JM (2018) Interaction between strigolactone and cytokinin on axillary and adventitious bud development in Zantedeschia. J Plant Physiol Pathol 6:1
Mason MG, Ross JJ, Babst BA, Wienclaw BN, Beveridge CA (2014) Sugar demand, not auxin, is the initial regulator of apical dominance. Proc Natl Acad Sci 111(16):6092–6097
Min Z, Li Z, Chen L, Zhang Y, Liu M, Yan X (2021) Transcriptome analysis revealed hormone signaling response of grapevine buds to strigolactone. Sci Hortic 283:109936
Müller D, Waldie T, Miyawaki K, To JP, Melnyk CW, Kieber JJ, Kakimoto T, Leyser O (2015) Cytokinin is required for escape but not release from auxin mediated apical dominance. Plant J 82(5):874–886
Muñoz A, Pillot JP, Cubas P, Rameau C (2021) Methods for phenotyping shoot branching and testing strigolactone bioactivity for shoot branching in Arabidopsis and Pea. Methods in Molecular Biology (Clifton, N.J.) 2309: 115–127.
Negi S, Sukumar P, Liu X, Cohen J, Muday G (2010) Genetic dissection of the role of ethylene in regulating auxin-dependent lateral and adventitious root formation in tomato. Plant J 61(1):3–15
Nguyen TQ, Emery RJN (2017) Is ABA the earliest upstream inhibitor of apical dominance? J Exp Bot 68(5):881–884
Ni J, Gao C, Chen MS, Pan BZ, Ye K, Xu ZF (2015) Gibberellin promotes shoot branching in the perennial woody plant Jatropha curcas. Plant Cell Physiol 56(8):1655–1666
Ni J, Zhao ML, Chen MS, Pan BZ, Tao YB, Xu ZF (2017) Comparative transcriptome analysis of axillary buds in response to the shoot branching regulators gibberellin A3 and 6-benzyladenine in Jatropha curcas. Sci Rep 7(1):11417
Ohashi M, Ishiyama K, Kojima S, Kojima M, Sakakibara H, Yamaya T (2017) Lack of cytosolic glutamine synthetase1; 2 activity reduces nitrogen-dependent biosynthesis of cytokinin required for axillary bud outgrowth in rice seedlings. Plant Cell Physiol 58(4):679–690
Omoarelojie LO, Kulkarni MG, Finnie JF, Van Staden J (2019) Strigolactone and their crosstalk with other phytohormones. Ann Bot 124(5):749–767
Ono E, Hatayama M, Isono Y, Sato T, Watanabe R, Yonekura-Sakakibara K, Fukuchi-Mizutani M, Tanaka Y, Kusumi T, Nishino T, Nakayama T (2006) Localization of a flavonoid biosynthetic polyphenol oxidase in vacuoles. Plant J 45(2):133–143
Parada F, Noriega X, Dantas D, Bressan-Smith R, Pérez FJ (2016) Differences in respiration between dormant and non-dormant buds suggest the involvement of ABA in the development of endodormancy in grapevines. J Plant Physiol 201:71–78
Reddy SK, Holalu SV, Casal JJ, Finlayson SA (2013) Abscisic acid regulates axillary bud outgrowth responses to the ratio of red to far-red light. Plant Physiol 163(2):1047–1058
Rong C, Liu Y, Chang Z, Liu Z, Ding Y, Ding C (2022) Cytokinin oxidase/dehydrogenase family genes exhibit functional divergence and overlap in rice growth and development, especially in control of tillering. J Exp Bot 73(11):3552–3568
Rubio-Moraga A, Ahrazem O, Pérez-Clemente RM, Gómez-Cadenas A, Yoneyama K, López-Ráez JA (2014) Apical dominance in saffron and the involvement of the branching enzymes CCD7 and CCD8 in the control of bud sprouting. BMC Plant Biol 14:171
Ruzicka K, Ljung K, Vanneste S, Podhorská R, Beeckman T, Friml J (2007) Ethylene regulates root growth through effects on auxin biosynthesis and transport-dependent auxin distribution. Plant Cell 19(7):2197–2212
Scaffidi A, Waters MT, Ghisalberti EL, Dxion KW, Flematti GR, Smith SM (2013) Carlactone-independent seedling morphogenesis in Arabidopsis[J]. Plant J 76(1):1–9
Schneider A, Godin C, Boudon F, Demotes-Mainard S, Sakr S, Bertheloot J (2019) Light regulation of axillary bud outgrowth along plant axes: an overview of the roles of sugars and hormone. Front Plant Sci 10:1296
Shen J, Zhang Y, Ge D, Wang Z, Song W, Gu R (2019) CsBRC1 inhibits axillary bud outgrowth by directly repressing the auxin efflux carrier CsPIN3 in cucumber. Proc Natl Acad Sci 116(34):17105–17114
Shi JB, Wang N, Zhou H, Xu QH, Yan GT (2019) The role of gibberellin synthase gene GhGA2ox1 in upland cotton (Gossypium hirsutum L.) responses to drought and salt stress. Biotechnol Appl Biochem 66(3):298–308
Shi J, Zhou H, Liu X, Wang N, Xu Q, Yan G (2021) Correlation analysis of the transcriptome and metabolome reveals the role of the flavonoid biosynthesis pathway in regulating axillary buds in upland cotton (Gossypium hirsutum L.). Planta 254(1):7
Sirhindi G, Mir MA, Abd-Allah EF, Ahmad P, Gucel S (2016) Jasmonic acid modulates the physio-biochemical attributes, antioxidant enzyme activity, and gene expression in glycine max under nickel toxicity. Front Plant Sci 7:591
Stepanova AN, Alonso JM (2009) Ethylene signaling and response: where different regulatory modules mee. Curr Opin Plant Biol 12(5):548–555
Tan M, Li G, Liu X, Cheng F, Ma J, Zhao C (2018a) a) Exogenous application of GA3 inactively regulates axillary bud outgrowth by influencing of branching-inhibitors and bud-regulating hormones in apple (Malus domestica Borkh.). Mol Genet Genom 293:1547–1563
Tan M, Li GF, Qi SY, Liu XJ, Chen XL, Ma JJ, Zhang D, Han MY (2018b) b) Identification and expression analysis of the IPT and CKX gene families during axillary bud outgrowth in apple (Males domestica Borkh.). Gene 651:106–117
Tan M, Li G, Chen X, Xing L, Ma J, Zhang D (2019) Role of cytokinin, strigolactone, and auxin export on outgrowth of axillary buds in apple. Front Plant Sci 10:616
Tenreira T, Lange MJP, Lange T, Bres C, Labadie M, Monfort A (2017) A specific gibberellin 20-oxidase dictates the flowering-runnering decision in diploid strawberry. Plant Cell 29(9):2168–2182
Tischer SV, Wunschel C, Papacek M, Kleigrewe K, Hofmann T, Christmann A (2017) Combinatorial interaction network of abscisic acid receptors and coreceptors from Arabidopsis thalian. Proc Natl Acad Sci 114(38):10280–10285
Vann MC (2017) Soil applied maleic hydrazide does not suppress tobacco axillary bud growth. Crop Forage Turfgrass Manag 3:1–8
Vergara R, Noriega X, Aravena KX, Aravena K (2017) ABA represses the expression of cell cycle genes and may modulate the development of endodormancy in grapevine buds. Front Plant Sci 8:812
Waldie T, Leyser O (2018) Cytokinin targets auxin transport to promote shoot branching. Plant Physiol 177(2):803–818
Wang Y, Yao R (2019) Increased endogenous indole-3-acetic acid: abscisic acid ratio is a reliable marker of Pinus massoniana rejuvenation. Biotech Histochem 94(7):546–553
Wang D, Gao Z, Du P, Xiao W, Tan Q, Chen X (2016) Expression of ABA metabolism-related genes suggests similarities and differences between seed dormancy and bud dormancy of peach (Prunus persica). Front Plant Sci 6:1248
Wang J, Lu K, Nie H, Zeng Q, Wu B, Qian J (2018) Rice nitrate transporter OsNPF7.2 positively regulates tiller number and grain yield. Rice 11(1):12
Wang R, Qian J, Fang Z, Tang J (2020) Transcriptomic and physiological analyses of rice seedlings under different nitrogen supplies provide insight into the regulation involved in axillary bud outgrowth. BMC Plant Biol 20(1):197
Waters MT, Brewer PB, Bussell JD, Smith SM, Beveridge CA (2012a) The Arabidopsis ortholog of rice DWARF27 acts upstream of MAX1 in the control of plant development by strigolactone. Plant Physiol 159(3):1073–1085
Waters MT, Nelson DC, Scaffidi A, Flematti GR, Sun YK, Dixon KW, Smith SM (2012b) Specialisation within the DWARF14 protein family confers distinct responses to karrikins and strigolactones in Arabidopsis. Development (cambridge, England) 139(7):1285–1295
Waters MT, Nelson DC, Scaffidi A, Flematti GR, Sun YK, Dixon KW, Smith SM, Wen C, Zhao Q, Nie J, Liu G, Shen L, Cheng C (2016) Physiological controls of chrysanthemum DgD27 gene expression in regulation of shoot branching. Plant Cell Rep 35(5):1053–1070
Wigchert SCM, Zwanenburg BA (1999) critical account on the inception of Striga seed germination. J Agric Food Chem 47(4):1320–1325
Xia F, Sun T, Yang S, Wang X, Chao J, Li X (2019) Insight into the B3transcription factor superfamily and expression profiling of B3 genes in axillary buds after topping in tobacco (Nicotiana tabacum L.). Genes 10(2):164
Xia X, Dong H, Yin Y, Song X, Gu X, Sang K, Zhou J, Shi K, Zhou Y, Foyer CH, Yu J (2021) Brassinosteroid signaling integrates multiple pathways to release apical dominance in tomato. Proc Natl Acad Sci USA 118(11):e2004384118
Xu J, Zha M, Li Y, Ding Y, Chen L, Ding C (2015) The interaction between nitrogen availability and auxin, cytokinin, and strigolactone in the control of shoot branching in rice (Oryza sativa L.). Plant Cell Rep 34(9):1647–1662
Xu J, Li Q, Yang L, Li X, Wang Z, Zhang Y (2020) Changes in carbohydrate metabolism and endogenous hormone regulation during bulblet initiation and development in Lycoris radiata. BMC Plant Biol 20(1):180
Yang H, Xu F, Liao H, Pan W, Zhang W, Xu B (2021) Transcriptome and metabolite analysis related to branch development in two genotypes of Eucalyptus urophylla. Mol Genet Genom 296:1071–1083
Yao C, Finlayson SA (2015) Abscisic acid is a general negative regulator of Arabidopsis axillary bud growth. Plant Physiol 169(1):611–626
Yoneyama K, Brewer PB (2021) Strigolactone, how are they synthesized to regulate plant growth and development? Curr Opin Plant Biol 63:102072
Yu C, Chen W, Wang Z, Lou H (2021) Comparative proteomic analysis of tomato (Solanum lycopersicum L.) shoots reveals crosstalk between strigolactone and auxin. Genomics 113(5):3163–3173
Yue P, Wang Y, Bu H, Li X, Yuan H, Wang A (2019) Ethylene promotes IAA reduction through PuERFs-activated PuGH31 during fruit ripening in pear (Pyrus ussuriensis). Postharvest Biol Technol 157:110955
Zha M, Imran M, Wang Y, Xu DY, Wang S (2019) Transcriptome analysis revealed the interaction among strigolactone, auxin, and cytokinin in controlling the shoot branching of rice. Plant Cell Rep 38(3):279–293
Zhao B, Wu TT, Ma SS, Jiang DJ, Bie XM, Sui N (2020) TaD27-B gene controls the tiller number in hexaploid wheat. Plant Biotechnol J 18(2):513–525
Zhao Y, Feng Y, Sun P, Wang B, Qiao J, Duan W (2021) Responses of phytohormones, carbon and nitrogen status to the trunk-extension pruning in three-year-old paulownia plantation. J Animal Plant Sci 31(2):450–458
Zhuang W, Gao Z, Wang L, Zhong W, Ni Z, Zhang Z (2013) Comparative proteomic and transcriptomic approaches to address the active role of GA4 in Japanese apricot flower bud dormancy release. J Exp Bot 64(16):4953–4966
Zhuang L, Wang J, Huang B (2017) Drought inhibition of tillering in Festuca arundinacea associated with axillary bud development and strigolactone signaling. Environ Exp Bot 142:15–23
Funding
This study was funded by the National Natural Science Foundation of China (Grant No. 32060420).
Author information
Authors and Affiliations
Contributions
HYT was responsible for experimental implementation, data analysis and manuscript drafting. BXT carried out the experiment and contributed to the reagents / materials / analytical tools; WWF carried out the experiment and participated in data collection; ZYP participated in data collection; YXW and FL participated in data analysis; JTP helped to revise the manuscript; GQL initiated and supervised the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Our authors claimed no competing interests.
Additional information
Communicated by Zsuzsanna Kolbert.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tian, H., Tang, B., Fan, W. et al. The role of strigolactone analog (GR24) in endogenous hormone metabolism and hormone-related gene expression in tobacco axillary buds. Plant Cell Rep 43, 21 (2024). https://doi.org/10.1007/s00299-023-03081-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00299-023-03081-y