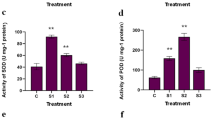
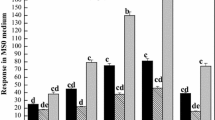
Abstract
Key message
Gibberellic acid elicited synthesis of many phenols from different classes and enhanced production of sesquiterpenoids, polyterpenoids, steroids and monoterpenoids compared to control and 6-benzylaminopurine.
Abstract
Little is known about the effects of 6-benzylaminopurine (BA) and gibberellic acid (GA3) on the synthesis of secondary metabolites in species of Lamiaceae. In this study, for the first time, the profile of secondary metabolites in plantlets of Cunila menthoides was characterized, using UPLC-ESI-Qq-oaTOF-MS. Ninety metabolites were identified, including polyphenols and terpenes. BA down-regulated most of the identified molecules in relation to GA3 and MS0 (control). The results showed that GA3 elicited synthesis of many phenols from different classes, and seemed to play a major role in the shikimate pathway in relation to BA. GA3 enhanced production of sesquiterpenoids, polyterpenoids, steroids and monoterpenoids compared to MS0 and BA, and also seemed to positively influence the MEP/DOXP and MVA pathways. These data show the most comprehensive metabolomic profile of Cunila menthoides to date, and the effects of BA and GA3 on the synthesis of secondary metabolites, modulating quantitative aspects of metabolism in Lamiaceae.





Similar content being viewed by others
Abbreviations
- UPLC–MSE :
-
Ultra performance liquid chromatography—mass spectrometry with independent MSE mode data
- HPLC:
-
High-performance liquid chromatography
- PGRs:
-
Plant growth regulators
- BA:
-
6-benzylaminopurine
- GA3:
-
Gibberellic acid
- TDZ:
-
Thidiazuron
- IAA:
-
Indole acetic acid
- MS0:
-
Sterile MS medium with no growth regulators
- UPLC-ESI-Qq-oaTOF-MS:
-
Ultra-performance liquid chromatography—electrospray ionization—quadrupole time-of-flight mass spectrometry
- PCA:
-
Principal components analysis
- HE:
-
Hydrophilic extract
- LE:
-
Lipophilic extract
- CV:
-
Coefficient of variation
- RT:
-
Retention time
- m/z :
-
Mass to charge
- RA:
-
Rosmarinic acid
- MVA:
-
Mevalonic acid
- DOXP/MEP:
-
2C-methyl-D-erythritol-4-phosphate
- GPP:
-
Geranyl diphosphate
- GGPP:
-
Geranylgeranyl diphosphate
- GGPPS:
-
Geranylgeranyl diphosphate synthases
- FPP:
-
(E,E)-farnesyl diphosphate
References
Agostini G (2008) Filogenia e diversidade genética do gênero Cunila D. Royen ex L, (Lamiaceae). Universidade Federal do Rio Grande do Sul
Agostini G, Agostini F, Bertolazzi M et al (2010) Variation of the chemical composition of essential oils in Brazilian populations of Cunila menthoides Benth. (Lamiaceae). Biochem Syst Ecol 38:906–910. https://doi.org/10.1016/j.bse.2010.09.011
Akula R, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6:1720–1731. https://doi.org/10.4161/psb.6.11.17613
Ali M, Abbasi BH (2015) Elicitation of antioxidant secondary metabolites with jasmonates and gibberellic acid in cell suspension cultures of Artemisia absinthium L. Plant Cell Tiss Organ Cult 120:1099–1106. https://doi.org/10.1007/s11240-014-0666-2
Antoniolli A, Fontana AR, Piccoli P, Bottini R (2015) Characterization of polyphenols and evaluation of antioxidant capacity in grape pomace of the cv. Malbec Food Chem 178:172–178. https://doi.org/10.1016/j.foodchem.2015.01.082
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Bordignon SADL., Montanha JA, Schenkel EP (2003) Flavones and flavanones from South American Cunila species (Lamiaceae). Biochem Syst Ecol 31:785–788. https://doi.org/10.1016/S0305-1978(03)00003-6
Boufridi A, Quinn RJ (2016) Turning metabolomics into drug discovery. J Braz Chem Soc 27:1334–1338. https://doi.org/10.5935/0103-5053.20160083
Cai S-Q, Wang R, Yang X et al (2006) Antiviral flavonoid-type C-glycosides from the flowers of Trollius chinensis. Chem Biodivers 3:343–348. https://doi.org/10.1002/cbdv.200690037
Céliz G, Rodriguez J, Soria F, Daz M (2015) Synthesis of hesperetin 7-O-glucoside from flavonoids extracted from Citrus waste using both free and immobilized α-l-rhamnosidases. Biocatal Agric Biotechnol 4:335–341. https://doi.org/10.1016/j.bcab.2015.06.005
Chen DQ, Chen H, Chen L et al (2016) Metabolomic application in toxicity evaluation and toxicological biomarker identification of natural product. Chem Biol Interact 252:114–130. https://doi.org/10.1016/j.cbi.2016.03.028
Cordoba E, Salmi M, Leon P (2009) Unravelling the regulatory mechanisms that modulate the MEP pathway in higher plants. J Exp Bot 60:2933–2943. https://doi.org/10.1093/jxb/erp190
Cramer CN, Brown JM, Tomczyk N et al (2017) Electron transfer dissociation of all ions at all times, MSETD, in a quadrupole time-of-flight (Q-ToF) mass spectrometer. J Am Soc Mass Spectrom 28:384–388. https://doi.org/10.1007/s13361-016-1538-2
Cuperlovic-Culf M, Culf AS (2016) Applied metabolomics in drug discovery. Expert Opin Drug Discov 11:759–770. https://doi.org/10.1080/17460441.2016.1195365
Delgado G, Hernández J, Pereda-Miranda R (1989) Triterpenoid acids from Cunila lythrifolia. Phytochemistry 28:1483–1485. https://doi.org/10.1016/S0031-9422(00)97769-0
Erkan N, Ayranci G, Ayranci E (2008) Antioxidant activities of rosemary (Rosmarinus officinalis L.) extract, blackseed (Nigella sativa L.) essential oil, carnosic acid, rosmarinic acid and sesamol. Food Chem 110:76–82. https://doi.org/10.1016/j.foodchem.2008.01.058
Francoise B, Hossein S, Halimeh H, Zahra NF (2007) Growth optimization of Zataria multiflora Boiss. Tissue cultures and rosmarinic acid production improvement. Pak J Biol Sci 10:3395–3399. https://doi.org/10.3923/pjbs.2007.3395.3399
Goyal S, Arora J, Ramawat KG (2014) Biotechnological approaches to medicinal plants of Aravalli Hills: Conservation and scientific validation of biological activities. 203–245. https://doi.org/10.1007/978-3-319-09381-9_11
Hazzoumi Z, Moustakime Y, Amrani Joutei K (2014) Effect of gibberellic acid (GA), indole acetic acid (IAA) and benzylaminopurine (BAP) on the synthesis of essential oils and the isomerization of methyl chavicol and trans-anethole in Ocimum gratissimum L. Springerplus 3:1–7. https://doi.org/10.1186/2193-1801-3-321
He Y, Dong X, Jia X et al (2015) Qualitative and quantitative analysis on chemical constituents from Curculigo orchioides using ultra high performance liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry. J Pharm Biomed Anal 102:236–245. https://doi.org/10.1016/j.jpba.2014.09.024
Hippolyte I, Marin B, Baccou JC, Jonard R (1992) Growth and rosmarinic acid production in cell suspension cultures of Salvia officinalis L. Plant Cell Rep 11:. https://doi.org/10.1007/BF00232160
Hong G-J, Xue X-Y, Mao Y-B et al (2012) Arabidopsis MYC2 interacts with DELLA proteins in regulating sesquiterpene synthase gene expression. Plant Cell 24:2635–2648. https://doi.org/10.1105/tpc.112.098749
Inthima P, Nakano M, Otani M et al (2017) Overexpression of the gibberellin 20-oxidase gene from Torenia fournieri resulted in modified trichome formation and terpenoid metabolities of Artemisia annua L. Plant Cell, Tissue Organ Cult 129:223–236. https://doi.org/10.1007/s11240-017-1171-1
Kodja H, Noirot M, Khoyratty SS et al (2015) Biochemical characterization of embryogenic calli of Vanilla planifolia in response to two years of thidiazuron treatment. Plant Physiol Biochem 96:337–344. https://doi.org/10.1016/j.plaphy.2015.08.017
Lyseng-Williamson KA, Perry CM (2003) Micronised purified flavonoid fraction: a review of its use in chronic venous insufficiency, venous ulcers and haemorrhoids. Drugs 63:71–100
Macedo AF, Barbosa NC, Esquibel MA et al (1999) Pharmacological and phytochemical studies of callus culture extracts from Alternanthera brasiliana. Pharmazie 54:776—777
Mahmoud SS, Croteau RB (2002) Strategies for transgenic manipulation of monoterpene biosynthesis in plants. Trends Plant Sci 7:366–373. https://doi.org/10.1016/S1360-1385(02)02303-8
Manole-Paunescu A (2014) Biotechnology for endangered plant conservation. 181–202. https://doi.org/10.1007/978-3-319-09381-9_10
Medrado HH, Dos Santos EO, Ribeiro EMO et al (2017) Rosmarinic and cinnamic acid derivatives of in vitro tissue culture of Plectranthus ornatus: Overproduction and correlation with antioxidant activities. J Braz Chem Soc 28:505–511. https://doi.org/10.21577/0103-5053.20160300
Meira PR, David JP, Erika EM et al (2017) Abiotic factors influencing podophyllotoxin and yatein overproduction in Leptohyptis macrostachys cultivated in vitro. Phytochem Lett 22:287–292. https://doi.org/10.1016/j.phytol.2017.10.016
Misra MC, Imlitemsu (2005) Drug treatment of haemorrhoids. Drugs 65:1481–1491
Mota J, Leite S, Kato AC MJ, et al (2011) Isoswertisin flavones and other constituents from Peperomia obtusifolia. Nat Prod Res 25:1–7. https://doi.org/10.1080/14786410903244954
Mukherjee PK, Bahadur S, Harwansh RK et al (2017) Paradigm shift in natural product research: traditional medicine inspired approaches. Phytochem Rev. https://doi.org/10.1007/s11101-016-9489-6
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Murcia G, Fontana A, Pontin M et al (2017) ABA and GA3 regulate the synthesis of primary and secondary metabolites related to alleviation from biotic and abiotic stresses in grapevine. Phytochemistry 135:34–52. https://doi.org/10.1016/j.phytochem.2016.12.007
Murthy HN, Lee E (2014) Production of secondary metabolites from cell and organ cultures: strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tiss Organ Cult 118:1–16. https://doi.org/10.1007/s11240-014-0467-7
Ng TLM, Karim R, Tan YS et al (2016) Amino acid and secondary metabolite production in embryogenic and non-embryogenic callus of fingerroot ginger (Boesenbergia rotunda). PLoS One 11:e0156714. https://doi.org/10.1371/journal.pone.0156714
Oliveira JPS, Koblitz MGB, Ferreira MSL et al (2016) Cunila menthoides Benth (Lamiaceae) beyond menthone, isomenthone and pulegone: a metabolomic study by UPLC-QTOF-MSE. In: 1st Ibero-american Conference on Mass Spectrometry. Rio de Janeiro, pp 206–207
Oliveira JPS, Hakimi O, Murgu M et al (2018) Tissue culture and metabolome investigation of a wild endangered medicinal plant using high definition mass spectrometry. Plant Cell Tissue Organ Cult. https://doi.org/10.1007/s11240-018-1408-7
Patel K, Patel J, Patel M et al (2010) Introduction to hyphenated techniques and their applications in pharmacy. Pharm Methods 1:2. https://doi.org/10.4103/2229-4708.72222
Perelli S, Moubayidin L, Sabatini S (2010) The molecular basis of cytokinin function. Curr Opin Plant Biol 13:21–26. https://doi.org/10.1016/j.pbi.2009.09.018
Perveen S, El-Shafae AM, Al-Taweel A et al (2011) Antioxidant and urease inhibitory C -glycosylflavonoids from Celtis africana. J Asian Nat Prod Res 13:799–804. https://doi.org/10.1080/10286020.2011.593171
Petersen M, Simmonds MSJ (2003) Rosmarinic acid. Phytochemistry 62:121–125. https://doi.org/10.1016/S0031-9422(02)00513-7
Prins CL, Vieira IJC, Freitas SP (2010) Growth regulators and essential oil production. Brazilian J Plant Physiol 22:91–102. https://doi.org/10.1590/S1677-04202010000200003
Qin Y, Liang Y, Ren D et al (2015) Separation of phenolic acids and flavonoids from Trollius chinensis Bunge by high speed counter-current chromatography. J Chromatogr B 1001:82–89. https://doi.org/10.1016/j.jchromb.2015.07.051
Rainville PD, Stumpf CL, Shockcor JP et al (2007) Novel application of reversed-phase UPLC-oaTOF-MS for lipid analysis in complex biological mixtures: A new tool for lipidomics. J Proteome Res 6:552–558. https://doi.org/10.1021/pr060611b
Ribeiro DM, Araújo WL, Fernie AR et al (2012) Translatome and metabolome effects triggered by gibberellins during rosette growth in Arabidopsis. J Exp Bot 63:2769–2786. https://doi.org/10.1093/jxb/err463
Sahu R, Dewanjee S, Gangopadhyay M (2013) Bioproduction and optimization of rosmarinic acid production in Solenostemon scutellarioides through media manipulation and conservation of high yielding clone via encapsulation. Nat Prod Commun 8:1275–1278
Saint-Lary L, Roy C, Paris J-P et al (2016) Metabolomics as a tool for the authentication of rose extracts used in flavour and fragrance area. Metabolomics 12:49. https://doi.org/10.1007/s11306-016-0963-3
Sangwan NS, Farooqi AHA, Shabih F, Sangwan RS (2001) Regulation of essential oil production in plants. Plant Growth Regul 34:3–21. https://doi.org/10.1023/A:1013386921596
Savaris M, Sabedot-Bordin SM, Mendes CE et al (2014) Evaluation of extracts and essential oils from “poejo”, Cunila angustifolia (Lamiales: Lamiaceae) leaves to control adults of maize weevil, Sitophilus zeamais (Coleoptera: Curculionidae). Int J Entomol Res 2:23–28
Schmiderer C, Grausgruber-Gröger S, Grassi P et al (2010) Influence of gibberellin and daminozide on the expression of terpene synthases and on monoterpenes in common sage (Salvia officinalis). J Plant Physiol 167:779–786. https://doi.org/10.1016/j.jplph.2009.12.009
Schweiger R, Heise AM, Persicke M, Müller C (2014) Interactions between the jasmonic and salicylic acid pathway modulate the plant metabolome and affect herbivores of different feeding types. Plant Cell Environ 37:1574–1585. https://doi.org/10.1111/pce.12257
Servili M, Montedoro G (2002) Contribution of phenolic compounds to virgin olive oil quality. Eur J Lipid Sci Technol 104:602–613. https://doi.org/10.1002/1438-9312(200210)104:9/10<602::AID-EJLT602>3.0.CO;2-X
Sharafzadeh S, Zare M (2011) Influence of growth regulators on growth and secondary metabolites of some medicinal plants from Lamiaceae family. 5:2296–2302
Shilpa K, Varun K, Lakshmi BS (2010) An alternate method of natural drug production: elciting secondary metabolite production using plant cell culture. J Plant Sci 5:222–247. https://doi.org/10.3923/jps.2010.222.247
Simões CMO, Mentz LA, Schenkel EP et al (1998) Plantas da medicina popular no Rio Grande do Sul. Editora da Universidade Federeal do Rio Grande do Sul, Rio Grande do Sul
Soriano IR, Asenstorfer RE, Schmidt O, Riley IT (2004) Inducible flavone in Oats (Avena sativa) is a novel defense against plant-parasitic nematodes. Phytopathology 94:1207–1214. https://doi.org/10.1094/PHYTO.2004.94.11.1207
Souza GHMF., Guest PC, Martins-de-Souza D (2017) LC-MSE, multiplex MS/MS, ion mobility, and label-free quantitation in clinical proteomics. 57–73
Stefano M, Papini A, Brighigna L (2008) A new quantitative classification of ecological types in the bromeliad genus Tillandsia (Bromeliaceae) based on trichomes. Rev Biol Trop 56:191–203
Szkopi A, Danuta P (2005) Farnesyl diphosphate synthase; regulation of product. Acta Biochim Pol 52:45–55
Thorn C, Whirl-Carrillo M, Klein T, Altman R (2007) Pathway-based Approaches to Pharmacogenomics. Curr Pharmacogenomics 5:79–86. https://doi.org/10.2174/157016007780077167
Trindade PF, Rosa CC, de S, Macedo AF (2015) Micropropagação da planta Cunila menthoides em diferentes tipos de hormônio. 14a Jornada de Iniciação Científica (JIC). Universidade Federal do Estado do Rio de Janeiro - UNIRIO, Rio de Janeiro, pp 88–90
Wang X, Yang B, Sun H, Zhang A (2012) Pattern recognition approaches and computational systems tools for ultra performance liquid chromatography-mass spectrometry-based comprehensive metabolomic profiling and pathways analysis of biological data sets. Anal Chem 84:428–439. https://doi.org/10.1021/ac202828r
Xi X, Kwok L-Y, Wang Y et al (2017) Ultra-performance liquid chromatography-quadrupole-time of flight mass spectrometry MSE -based untargeted milk metabolomics in dairy cows with subclinical or clinical mastitis. J Dairy Sci 100:4884–4896. https://doi.org/10.3168/jds.2016-11939
Yan H, Yoo M-J, Koh J et al (2014) Molecular Reprogramming of Arabidopsis in Response to Perturbation of Jasmonate Signaling. J Proteome Res 13:5751–5766. https://doi.org/10.1021/pr500739v
Zhao Y-Y, Lin R-C (2014) UPLC–MSE application in disease biomarker discovery: The discoveries in proteomics to metabolomics. Chem Biol Interact 215:7–16. https://doi.org/10.1016/j.cbi.2014.02.014
Zuk M, Kulma A, Dymińska L et al (2011) Flavonoid engineering of flax potentiate its biotechnological application. BMC Biotechnol 11:10. https://doi.org/10.1186/1472-6750-11-10
Acknowledgements
The authors are grateful to Prof. Dr. Agostini from the Federal University of Rio Grande do Sul (UFRGS), Porto Alegre, for his support in collecting and identifying the plant material; to the Universidade Federal do Estado do Rio de Janeiro (UNIRIO) for a scholarship; to Prof. Dr. Suellen G. Moreira from the Federal Institute of Rio de Janeiro (IFRJ), Rio de Janeiro, Brazil, for helping to develop the UPLC separation methodology; to Prof. Dr. André Ferreira from the Oswaldo Cruz Fundation (FIOCRUZ) and to the Waters Corporation for logistical and technical support, respectively.
Funding
This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Financiadora de Estudos e Projetos (FINEP), Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) and Universidade Federal do Estado do Rio de Janeiro (Unirio).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Attila Feher.
Electronic supplementary material
Below is the link to the electronic supplementary material.
299_2018_2303_MOESM1_ESM.pdf
Supplementary Table 1 and 2. List of identified compounds in HE and LE respectively, with average abundance by treatment, isotope distribution, maximum fold change, mass error, molecular formula, retention time, mass/charge and score of C. menthoides (PDF 1201 KB)

Rights and permissions
About this article
Cite this article
Oliveira, J.P.S., Koblitz, M.G.B., Ferreira, M.S.L. et al. Comparative metabolomic responses to gibberellic acid and 6-benzylaminopurine in Cunila menthoides Benth. (Lamiaceae): a contribution to understand the metabolic pathways. Plant Cell Rep 37, 1173–1185 (2018). https://doi.org/10.1007/s00299-018-2303-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-018-2303-8