Abstract
Key message
The green - revertible yellow79 mutant resulting from a single-base mutation suggested that the GRY79 gene encoding a putative metallo-β-lactamase-trihelix chimera is involved in chloroplast development at early seedling stage of rice.
Abstract
Functional studies of metallo-β-lactamases and trihelix transcription factors in higher plants remain very sparse. In this study, we isolated the green-revertible yellow79 (gry79) mutant in rice. The mutant developed yellow-green leaves before the three-leaf stage but recovered to normal green at the sixth-leaf stage. Meanwhile, the mutant exhibited reduced level of chlorophylls and arrested development of chloroplasts in the yellow leaves. Genetic analysis suggested that the mutant phenotype was controlled by a single recessive nuclear gene on rice chromosome 2. Map-based cloning revealed that the candidate gene was Os02g33610 encoding a putative metallo-β-lactamase-trihelix chimera. In the gry79 mutant, a single-base mutation occurred in coding region of the gene, resulting in an amino acid change in the encoded protein. Furthermore, the mutant phenotype was rescued by transformation with the wild-type gene. Therefore, we have confirmed that the gry79 mutant phenotype resulted from a single-base mutation in GRY79 (Os02g33610) gene, suggesting that the gene encoding a putative metallo-β-lactamase-trihelix chimera is involved in chloroplast development at early seedling stage of rice. In addition, we considered that the gry79 mutant gene could be applicable as a leaf-color marker gene for efficient identification and elimination of false hybrids in commercial hybrid rice production.







Similar content being viewed by others
References
Ambler RP (1980) The structure of β-lactamases. Philos Trans R Soc Lond B 289:321–331
Archer EK, Bonnett HT (1987) Characterization of a virescent chloroplast mutant of tobacco. Plant Physiol 83:920–925
Arnon DI (1949) Copper enzymes in isolated chloroplasts: polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Callebaut I, Moshous D, Mornon JP, de Villartay JP (2002) Metallo-β-lactamase fold within nucleic acid processing enzymes: the β-CASP family. Nucleic Acids Res 30:3592–3601
Cao LY, Qian Q, Zhu XD, Zheng DL, Min SK, Xiong ZM (1999) Breeding of a photoperiod-sensitive genic male sterile indica rice Zhongzi S with a purple leaf marker and the heterosis of its hybrid rice produced with it. Acta Agron Sin 25:44–49
Deng QY, Yuan LP (1998) Fertility stability of P(T)GMS lines in rice and its identification techniques. Chin J Rice Sci 12:200–206
Dominski Z (2007) Nucleases of the metallo-β-lactamase family and their role in DNA and RNA metabolism. Crit Rev Biochem Mol Biol 42:67–93
Dong FG, Zhu XD, Xiong ZM, Cheng SH, Sun ZX, Min SK (1995) Breeding of a photo-thermoperiod sensitive genie male sterile indica rice with a pale-green-leaf marker. Chin J Rice Sci 9:65–70
Drawz SM, Bonomo RA (2010) Three decades of β-lactamase inhibitors. Clin Microbiol Rev 23:160–201
Emanuelsson O, Nielsen H, Brunak S, Heijne GV (2000) Predicting subcellular localization of protein based on their N-terminal amino acid sequence. J Mol Biol 300:1005–1016
Gao MJ, Lydiate DJ, Li X, Lui H, Gjetvaj B, Hegedus DD, Rozwadowski K (2009) Repression of seed maturation genes by a trihelix transcriptional repressor in Arabidopsis seedlings. Plant Cell 21:54–71
Jung KH, Hur J, Ryu CH, Choi Y, Chung YY, Miyao A, Hirochika H, An G (2003) Characterization of a rice chlorophyll-deficient mutant using the T-DNA gene-trap system. Plant Cell Physiol 44:463–472
Kaplan-Levy RN, Brewer PB, Quon T, Smyth DR (2012) The trihelix family of transcription factors—light, stress and development. Trends Plant Sci 17:163–171
Kimura S, Saotome A, Uchiyama Y, Mori Y, Tahira Y, Sakaguchi K (2005) The expression of the rice (Oryza sativa L.) homologue of Snm1 is induced by DNA damages. Biochem Biophys Res Commun 329:668–672
Kusaba M, Ito H, Morita R, Lida S, Sato Y, Fujimoto M, Kawasaki S, Tanaka R, Hirochika H, Nishimura M, Tanaka A (2007) Rice NON-YELLOW COLORING1 is involved in light-harvesting complex II and grana degradation during leaf senescence. Plant Cell 19:1362–1375
Kusumi K, Sakata C, Nakamura T, Kawasaki S, Yoshimura A, Iba K (2011) A plastid protein NUS1 is essential for build-up of the genetic system for early chloroplast development under cold stress conditions. Plant J 68:1039–1050
Lee S, Kim JH, Yoo ES, Lee CH, Hirochika H, An G (2005) Differential regulation of chlorophyll a oxygenase genes in rice. Plant Mol Biol 57:805–818
Li XR, Moses RE (2003) The β-lactamase motif in Snm1 is required for repair of DNA double- strand breaks caused by interstrand crosslinks in S. cerevisiae. DNA Repair 2:121–129
Li JJ, Pandeya D, Nath K, Zulfugarov IS, Yoo SC, Zhang HT, Yoo JH, Cho SH, Koh HJ, Kim DS, Seo HS, Kang BC, Lee CH, Paek NC (2010) ZEBRA-NECROSIS, a thylakoid- bound protein, is critical for the photoprotection of developing chloroplasts during early leaf development. Plant J 62:713–725
Liao FM, Yuan LP, Yang YS (2001) Sterility purification of the photoperiod-thermo sensitive genic male rice line Pei’ai 64S. Chin J Rice Sci 15:1–6
McCouch SR, Teytelman L, Xu YB, Lobos KB, Clare K, Walton M, Fu BY, Maghirang R, Li ZK, Xing YZ, Zhang QF, Kono I, Yano M, Fjellstrom R, DeClerck G, Schneider D, Cartinhour S, Ware D, Stein L (2002) Development and mapping of 2240 new SSR markers for rice (Oryza sativa L.). DNA Res 9:199–207
McElver J, Tzafrir I, Aux G, Rogers R, Ashby C, Smith K, Thomas C, Schetter A, Zhou Q, Cushman MA, Tossberg J, Nickle T, Levin JZ, Law M, Meinke D, Patton D (2001) Insertional mutagenesis of genes required for seed development in Arabidopsis thaliana. Genetics 159:1751–1763
Molinier J, Stamm ME, Hohn B (2004) SNM-dependent recombinational repair of oxidatively induced DNA damage in Arabidopsis thaliana. EMBO Rep 5:994–999
Normile D (2008) Agricultural research: reinventing rice to feed the world. Science 321:330–333
Riechmann JL, Heard J, Martin G, Reuber L, Jiang CZ, Keddie J, Adam L, Pineda O, Ratcliffe OJ, Samata RR, Creelman R, Pilgrim M, Broun P, Zhang JZ, Ghandehari D, Sherman BK, Yu GL (2000) Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science 290:2105–2110
Sakuraba Y, Rahman ML, Cho SH, Kim YS, Koh HJ, Yoo SC, Peak NC (2013) The rice faded green leaf locus encodes protochlorophyllide oxidoreductase B and is essential for chlorophyll synthesis under high light conditions. Plant J 74:122–133
Sato Y, Morita R, Nishimura M, Yamaguchi H, Kusaba M (2007) Mendel’s green cotyledon gene encodes a positive regulator of the chlorophyll-degrading pathway. Proc Natl Acad Sci USA 104:14169–14174
Sato Y, Morita R, Katsuma S, Nishimura M, Tanaka A, Kusaba M (2009) Two short-chain dehydrogenase/reductases, NON-YELLOW COLORING 1 and NYC1-LIKE, are required for chlorophyll b and light-harvesting complex II degradation during senescence in rice. Plant J 57:120–131
Schmid M, Davison TS, Henz SR, Pape UJ, Demar M, Vingron M, Schölkopf B, Weigel D, Lohmann JU (2005) A gene expression map of Arabidopsis thaliana development. Nat Genet 37:501–506
Shen SQ, Shu QY, Bao JS, Wu DX, Cui HR, Xia YW (2004) Development of a greenable leaf colour mutant Baifeng A and its application in hybrid rice production. Chin J Rice Sci 18:34–38
Song KB, Song ZG (2007) Discovery and preliminary research of the yellowish leaf mutant Annongbiao 810S in rice. Hybrid Rice 22:71–73
Su N, Hu ML, Wu DX, Wu FQ, Fei GL, Lan Y, Chen XL, Shu XL, Zhang X, Guo XP, Cheng ZJ, Lei CL, Qi CK, Jiang L, Wang HY, Wan JM (2012) Disruption of a rice pentatricopeptide repeat protein causes a seedling-specific albino phenotype and its utilization to enhance seed purity in hybrid rice production. Plant Physiol 159:227–238
Sugimoto H, Kusumi K, Noguchi K, Yano M, Yoshimura A, Iba K (2007) The rice nuclear gene, VIRESCENT 2, is essential for chloroplast development and encodes a novel type of guanylate kinase targeted to plastids and mitochondria. Plant J 52:512–527
Sun CH, Fang J, Zhao TL, Xu B, Zhang FT, Liu LC, Tang JY, Zhang GF, Deng XJ, Chen F, Qian Q, Cao XF, Chu CC (2012) The histone methyltransferase SDG724 mediates H3K36me2/3 deposition at MADS50 and RFT1 and promotes flowering in rice. Plant Cell 24:3235–3247
Tzafrir I, Muralla RP, Dickerman A, Berg M, Rogers R, Hutchens S, Sweeney TC, McElver J, Aux G, Patton D, Meinke D (2004) Identification of genes required for embryo development in Arabidopsis. Plant Physiol 135:1206–1220
Wang PR, Gao JX, Wan CM, Zhang FT, Xu ZJ, Huang XQ, Sun XQ, Deng XJ (2010) Divinyl chlorophyll (ide) a can be converted to monovinyl chlorophyll (ide) a by a divinyl reductase in rice. Plant Physiol 153:994–1003
Wu DX, Shu QY, Xia YW (2002) In vitro mutagenesis induced novel thermo/photoperiod-sensitive genic male sterile indica rice with green-revertible xanthan leaf color marker. Euphytica 123:195–202
Wu ZM, Zhang X, He B, Diao LP, Sheng SL, Wang JL, Guo XP, Su N, Wang LF, Jiang L, Wang CM, Zhai HQ, Wan JM (2007) A chlorophyll-deficient rice mutant with impaired chlorophyllide esterification in chlorophyll biosynthesis. Plant Physiol 145:29–40
Yoo SC, Cho SH, Sugimoto H, Li JJ, Kusumi K, Koh HJ, Iba K, Paek NC (2009) Rice virescent3 and stripe1 encoding the large and small subunits of ribonucleotide reductase are required for chloroplast biogenesis during early leaf development. Plant Physiol 150:388–401
Yuan LP (2004) Hybrid rice technology for food security in the world. International conference on sustainable rice systems. FAO, Rome
Zhang HT, Li JJ, Yoo JH, Yoo SC, Cho SH, Koh HJ, Seo HS, Paek NC (2006) Rice Chlorina-1 and Chlorina-9 encode ChlD and ChlI subunits of Mg-chelatase, a key enzyme for chlorophyll synthesis and chloroplast development. Plant Mol Biol 62:325–337
Zhang Y, Su JB, Duan S, Ao Y, Dai JR, Liu J, Wang P, Li YG, Liu B, Feng DR, Wang JF, Wang HB (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7:30
Zhao HJ, Wu DX, Shu QY, Shen SQ, Ma CX (2004) Breeding and characteristics of photo-thermo sensitive genic male sterile rice Yutu S labeled with green-revertible albino leaf marker. Chin J Rice Sci 18:515–521
Acknowledgments
We specially thank Prof. Chengcai Chu from Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, for kindly providing the binary vector pCAMBIA2300. This study was financially supported by the National Natural Science Foundation of China (31071402, 31171533, 31471473, and 91335107).
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by K. Chong.
Electronic supplementary material
Below is the link to the electronic supplementary material.
299_2015_1792_MOESM1_ESM.doc
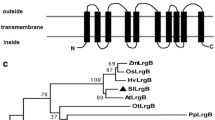
Supplemental Fig. S1 Alignment of the deduced amino acid sequence of GRY79 and its homologs. Identical residues were boxed in black, similar residues (≥75 % identical) were highlighted in gray. The black underline indicates the putative chloroplast-targeting sequence of 36 amino acid residues at its N terminus. The red and blue underlines indicate the putative metallo-β-lactamase domain of 202 amino acid residues at its N terminus and the putative trihelix DNA binding domain of 66 amino acid residues at its C terminus, respectively. The red arrowhead indicates mutational site (from serine (S) to phenylalanine (F) at position 387) of the gry79 mutant. Accession numbers for the respective protein sequences are as Fig. 4. (DOC 263 kb)
Rights and permissions
About this article
Cite this article
Wan, C., Li, C., Ma, X. et al. GRY79 encoding a putative metallo-β-lactamase-trihelix chimera is involved in chloroplast development at early seedling stage of rice. Plant Cell Rep 34, 1353–1363 (2015). https://doi.org/10.1007/s00299-015-1792-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1792-y