Abstract
Key message
A platform of gene silencing by amiRNA had been established in fertile transgenic soybean. We demonstrated that knockdown of storage protein shifted the distribution of nitrogen sources in soybean seeds.
Abstract
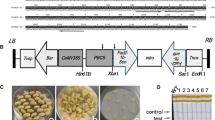
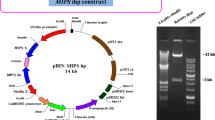
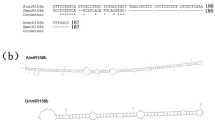
Artificial microRNAs (amiRNAs) were designed using the precursor sequence of the endogenous soybean (Glycine max L. Merrill) miRNA gma-miR159a and expressed in transgenic soybean plants to suppress the biosynthesis of 7S globulin, which is one of the major storage proteins. Seed-specific expression of these amiRNAs (amiR-7S) resulted in a strong suppression of 7S globulin subunit genes and decreased accumulation of the 7S globulin subunits in seeds. Thus, the results demonstrate that a platform for gene silencing by amiRNA was first developed in fertile transgenic soybean plants. There was no difference in nitrogen, carbon, and lipid contents between amiR-7S and control seeds. Four protein fractions were collected from defatted mature seeds on the basis of solubility at different pH to examine the distribution of nitrogen sources and compensatory effects. In the whey and lipophilic fractions, nitrogen content was similar in amiR-7S and control seeds. Nitrogen content was significantly decreased in the major soluble protein fraction and increased in the residual fraction (okara) of the amiR-7S seeds. Amino acid analysis revealed that increased nitrogen compounds in okara were proteins or peptides rather than free amino acids. Our study indicates that the decrease in 7S globulin subunits shifts the distribution of nitrogen sources to okara in transgenic soybean seeds.








Similar content being viewed by others
References
Ai T, Zhang L, Gao Z, Zhu CX, Guo X (2011) Highly efficient virus resistance mediated by artificial microRNAs that target the suppressor of PVX and PVY in plants. Plant Biol 13:304–316. doi:10.1111/j.1438-8677.2010.00374.x
Allen DK, Young JD (2013) Carbon and nitrogen provisions alter the metabolic flux in developing soybean embryos. Plant Physiol 161:1458–1475. doi:10.1104/pp.112.203299
Allen RS, Li J, Stahle MI, Dubroue A, Gubler F, Millar AA (2007) Genetic analysis reveals functional redundancy and the major target genes of the Arabidopsis miR159 family. Proc Natl Acad Sci USA 104:16371–16376. doi:10.1073/pnas.0707653104
Alonso-Peral MM, Li J, Li Y, Allen RS, Schnippenkoetter W, Ohms S, White RG, Millar AA (2010) The microRNA159-regulated GAMYB-like genes inhibit growth and promote programmed cell death in Arabidopsis. Plant Physiol 154:757–771. doi:10.1104/pp.110.160630
Brim CA, Burton JW (1979) Recurrent selection in soybeans. 2. Selection for increased percent protein in seeds. Crop Sci 19:494–498
Cober ER, Voldeng HD (2000) Developing high-protein, high-yield soybean populations and lines. Crop Sci 40:39–42
Diers BW, Keim P, Fehr WR, Shoemaker RC (1992) RFLP analysis of soybean seed protein and oil content. Theor Appl Genet 83:608–612
Dwiyanti MS, Yamada T, Sato M, Abe J, Kitamura K (2011) Genetic variation of gamma-tocopherol methyltransferase gene contributes to elevated alpha-tocopherol content in soybean seeds. BMC Plant Biol. doi:10.1186/1471-2229-11-152
Fahim M, Millar AA, Wood CC, Larkin PJ (2012) Resistance to wheat streak mosaic virus generated by expression of an artificial polycistronic microRNA in wheat. Plant Biotechnol J 10:150–163. doi:10.1111/j.1467-7652.2011.00647.x
Fernandez AI, Viron N, Alhagdow M, Karimi M, Jones M, Amsellem Z, Sicard A, Czerednik A, Angenent G, Grierson D, May S, Seymour G, Eshed Y, Lemaire-Chamley M, Rothan C, Hilson P (2009) Flexible tools for gene expression and silencing in tomato. Plant Physiol 151:1729–1740. doi:10.1104/pp.109.147546
Fischer RL, Goldberg RB (1982) Structure and flanking regions of soybean seed protein genes. Cell 29:651–660. doi:10.1016/0092-8674(82)90181-7
Goto K, Kanazawa A, Kusaba M, Masuta C (2003) A simple and rapid method to detect plant siRNAs using nonradioactive probes. Plant Mol Biol Rep 21:51–58. doi:10.1007/bf02773396
Hajika M, Takahashi M, Sakai SJ, Igita K (1996) A new genotype of 7 S globulin (beta-conglycinin) detected in wild soybean (Glycine soja Sieb et Zucc). Breed Sci 46:385–386
Herman EM, Larkins BA (1999) Protein storage bodies and vacuoles. Plant Cell 11:601–613
Iwabuchi S, Yamauchi F (1987) Electrophoretic analysis of whey proteins present in soybean globulin fractions. J Agric Food Chem 35:205–209. doi:10.1021/jf00074a010
Kinney AJ, Jung R, Herman EM (2001) Cosuppression of the alpha subunits of beta-conglycinin in transgenic soybean seeds induces the formation of endoplasmic reticulum-derived protein bodies. Plant Cell 13:1165–1178. doi:10.1105/tpc.13.5.1165
Kitamura K, Davies CS, Nielsen NC (1984) Inheritance of alleles for cgy1 and gy4 storage protein genes in soybean. Theo Appl Genet 68:253–257. doi:10.1007/bf00266899
Krishnan HB, Natarajan SS, Mahmoud AA, Nelson RL (2007) Identification of glycinin and beta-conglycinin subunits that contribute to the increased protein content of high-protein soybean lines. J Agric Food Chem 55:1839–1845. doi:10.1021/jf062497n
Kwanyuen P, Pantalone VR, Burton JW, Wilson RF (1997) A new approach to genetic alteration of soybean protein composition and quality. J Am Oil Chem Soc 74:983–987. doi:10.1007/s11746-997-0015-2
Li C, Zhang YM (2011) Molecular evolution of glycinin and beta-conglycinin gene families in soybean (Glycine max L. Merr.). Heredity 106:633–641. doi:10.1038/hdy.2010.97
Mansur LM, Orf JH, Chase K, Jarvik T, Cregan PB, Lark KG (1996) Genetic mapping of agronomic traits using recombinant inbred lines of soybean. Crop Sci 36:1327–1336
Maruyama N, Prak K, Motoyama S, Choi SK, Yagasaki K, Ishimoto M, Utsumi S (2004) Structure-physicochemical function relationships of soybean glycinin at subunit levels assessed by using mutant lines. J Agric Food Chem 52:8197–8201. doi:10.1021/jf048786y
Mateos-Aparicio I, Mateos-Peinado C, Ruperez P (2010) High hydrostatic pressure improves the functionality of dietary fiber in okara by-product from soybean. Innov Food Sci Emerg Technol 11:445–450. doi:10.1016/j.ifset.2010.02.003
Melito S, Heuberger AL, Cook D, Diers BW, MacGuidwin AE, Bent AF (2010) A nematode demographics assay in transgenic roots reveals no significant impacts of the Rhg1 locus LRR-Kinase on soybean cyst nematode resistance. BMC Plant Biol. doi:10.1186/1471-2229-10-104
Mori T, Utsumi S, Inaba H, Kitamura K, Harada K (1981) Differences in subunit composition of glycinin among soybean cultivars. J Agric Food Chem 29:20–23. doi:10.1021/jf00103a006
Mori T, Maruyama N, Nishizawa K, Higasa T, Yagasaki K, Ishimoto M, Utsumi S (2004) The composition of newly synthesized proteins in the endoplasmic reticulum determines the transport pathways of soybean seed storage proteins. Plant J 40:238–249. doi:10.1111/j.1365-313X.2004.02204.x
Nakamura T, Utsumi S, Kitamura K, Harada K, Mori T (1984) Cultivar differences in gelling characteristics of soybean glycinin. J Agric Food Chem 32:647–651. doi:10.1021/jf00123a055
Napoli C, Lemieux C, Jorgensen R (1990) Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes in trans. Plant Cell 2:279–289. doi:10.1105/tpc.2.4.279
Narikawa T, Tamura T, Yagasaki K, Terauchi K, Sanmiya K, Ishimaru Y, Abe K, Asakura T (2010) Expression of the stress-related genes for glutathione S-transferase and ascorbate peroxidase in the most-glycinin-deficient soybean cultivar Tousan205 during seed maturation. Biosci Biotechnol Biochem 74:1976–1979. doi:10.1271/bbb.100479
Nielsen NC, Dickinson CD, Cho TJ, Thanh VH, Scallon BJ, Fischer RL, Sims TL, Drews GN, Goldberg RB (1989) Characterization of the glycinin gene family in soybean. Plant Cell 1:313–328. doi:10.1105/tpc.1.3.313
Nishizawa K, Maruyama N, Satoh R, Fuchikami Y, Higasa T, Utsumi S (2003) A C-terminal sequence of soybean beta-conglycinin alpha ‘ subunit acts as a vacuolar sorting determinant in seed cells. Plant J 34:647–659. doi:10.1046/j.1365-313X.2003.01754.x
Nishizawa K, Takagi K, Teraishi M, Kita A, Ishimoto M (2010) Application of somatic embryos to rapid and reliable analysis of soybean seed components by RNA interference-mediated gene silencing. Plant Biotechnol 27:409–420
Niu Q-W, Lin S–S, Reyes JL, Chen K-C, Wu H-W, Yeh S-D, Chua N-H (2006) Expression of artificial microRNAs in transgenic Arabidopsis thaliana confers virus resistance. Nature Biotechnol 24:1420–1428. doi:10.1038/nbt1255
Niwa Y, Hirano T, Yoshimoto K, Shimizu M, Kobayashi H (1999) Non-invasive quantitative detection and applications of non-toxic, S65T-type green fluorescent protein in living plants. Plant J 18:455–463. doi:10.1046/j.1365-313X.1999.00464.x
Ogawa T, Tayama E, Kitamura K, Kaizuma N (1989) Genetic-improvement of seed storage proteins using 3 variant alleles of 7S globulin subunits in soybean (Glycine-max L). Jpn J Breed 39:137–147
Ohtake N, Yamada S, Suzuki M, Takahashi N, Takahashi Y, Chinushi T, Ohyama T (1997) Regulation of accumulation of beta-subunit of beta-conglycinin in soybean seeds by nitrogen. Soil Sci Plant Nutrition 43:247–253
Pathan SM, Tri V, Clark K, Lee J-D, Shannon JG, Roberts CA, Ellersieck MR, Burton JW, Cregan PB (2013) Hyten Genetic mapping and confirmation of quantitative trait loci for seed protein and oil contents and seed weight in soybean. Crop Sci 53:765–774. doi:10.2135/cropsci2012.03.0153
Prak K, Nakatani K, Katsube-Tanaka T, Adachi M, Maruyama N, Utsumi S (2005) Structure-function relationships of soybean proglycinins at subunit levels. J Agric Food Chem 53:3650–3657. doi:10.1021/jf047811x
Redondo-Cuenca A, Villanueva-Suarez MJ, Mateos-Aparicio I (2008) Soybean seeds and its by-product okara as sources of dietary fibre. Measurement by AOAC and Englyst methods. Food Chem 108:1099–1105. doi:10.1016/j.foodchem.2007.11.061
Samoto M, Miyazaki C, Kanamori J, Akasaka T, Kawamura Y (1998) Improvement of the off-flavor of soy protein isolate by removing oil-body associated proteins and polar lipids. Biosci Biotechnol Biochem 62:935–940. doi:10.1271/bbb.62.935
Samoto M, Maebuchi M, Miyazaki C, Kugitani H, Kohno M, Hirotsuka M, Kito M (2007) Abundant proteins associated with lecithin in soy protein isolate. Food Chem 102:317–322. doi:10.1016/j.foodchem.2006.05.054
Sato H, Yamada T, Kita Y, Ishimoto M, Kitamura K (2007) Production of transgenic plants and their early seed set in Japanese soybean variety, Kariyutaka. Plant Biotechnol 5:533–536
Scallon B, Thanh VH, Floener LA, Nielsen NC (1985) Identification and characterization of DNA clones encoding group-II glycinin subunits. Theor Appl Genet 70:510–519. doi:10.1007/bf00305984
Schmidt MA, Herman EM (2008) Proteome rebalancing in soybean seeds can be exploited to enhance foreign protein accumulation. Plant Biotechnol J 6:832–842. doi:10.1111/j.1467-7652.2008.00364.x
Schmidt MA, Barbazuk WB, Sandford M, May G, Song Z, Zhou W, Nikolau BJ, Herman EM (2011) Silencing of soybean seed storage proteins results in a rebalanced protein composition preserving seed protein content without major collateral changes in the metabolome and transcriptome. Plant Physiol 156:330–345. doi:10.1104/pp.111.173807
Sebolt AM, Shoemaker RC, Diers BW (2000) Analysis of a quantitative trait locus allele from wild soybean that increases seed protein concentration in soybean. Crop Sci 40:1438–1444
Shi A, Chen P, Zhang B, Hou A (2010a) Genetic diversity and association analysis of protein and oil content in food-grade soybeans from Asia and the United States. Plant Breed 129:250–256. doi:10.1111/j.1439-0523.2010.01766.x
Shi R, Yang C, Lu S, Sederoff R, Chiang VL (2010b) Specific down-regulation of PAL genes by artificial microRNAs in Populus trichocarpa. Planta 232:1281–1288. doi:10.1007/s00425-010-1253-3
Shibata M, Takayama K, Ujiie A, Yamada T, Abe J, Kitamura K (2008) Genetic relationship between lipid content and linolenic acid concentration in soybean seeds. Breed Sci 58:361–366. doi:10.1270/jsbbs.58.361
Sorgentini DA, Wagner JR (1999) Comparative study of structural characteristics and thermal behavior of whey and isolate soybean proteins. J Food Biochem 23:489–507. doi:10.1111/j.1745-4514.1999.tb00033.x
Takahashi K, Banba H, Kikuchi A, Ito M, Nakamura S (1994) An induced mutant line lacking the alpha-subunit of beta-conglycinin in soybean Glycine-max (L) Merrill. Breed Sci 65-66
Takahashi M, Uematsu Y, Kashiwaba K, Yagasaki K, Hajika M, Matsunaga R, Komatsu K, Ishimoto M (2003) Accumulation of high levels of free amino acids in soybean seeds through integration of mutations conferring seed protein deficiency. Planta 217:577–586. doi:10.1007/s00425-003-1026-3
Tezuka M, Taira H, Igarashi Y, Yagasaki K, Ono T (2000) Properties of tofus and soy milks prepared from soybeans having different subunits of glycinin. J Agric Food Chem 48:1111–1117. doi:10.1021/jf990560l
Thanh VH, Shibasaki K (1976) Major proteins of soybean seeds—straightforward fractionation and their characterization. J Agric Food Chem 24:1117–1121
Thanh VH, Shibasaki K (1978) Major proteins of soybean seeds—reconstitution of beta-conglycinin from its subunits. J Agric Food Chem 26:695–698
Toppino L, Kooiker M, Lindner M, Dreni L, Rotino GL, Kater MM (2011) Reversible male sterility in eggplant (Solanum melongena L.) by artificial microRNA-mediated silencing of general transcription factor genes. Plant Biotechnol J 9:684–692. doi:10.1111/j.1467-7652.2010.00567.x
Tsubokura Y, Hajika M, Kanamori H, Xia Z, Watanabe S, Kaga A, Katayose Y, Ishimoto M, Harada K (2012) The beta-conglycinin deficiency in wild soybean is associated with the tail-to-tail inverted repeat of the alpha-subunit genes. Plant Mol Biol 78:301–309. doi:10.1007/s11103-011-9865-y
Vanderriet WB, Wight AW, Cilliers JJL, Datel JM (1989) Food chemical investigation of tofu and its by-product okara. Food Chem 34:193–202. doi:10.1016/0308-8146(89)90140-4
Wadahama H, Iwasaki K, Matsusaki M, Nishizawa K, Ishimoto M, Arisaka F, Takagi K, Urade R (2012) Accumulation of beta-conglycinin in soybean cotyledon through the formation of disulfide bonds between alpha ‘- and alpha-subunits. Plant Physiol 158:1395–1405. doi:10.1104/pp.111.189621
Warthmann N, Chen H, Ossowski S, Weigel D, Herve P (2008) Highly specific gene silencing by artificial miRNAs in Rice. Plos One. doi:10.1371/journal.pone.0001829
Yamada T, Ohashi Y, Ohshima M, Inui H, Shiota N, Ohkawa H, Ohkawa Y (2002) Inducible cross-tolerance to herbicides in transgenic potato plants with the rat CYP1A1 gene. Theor Appl Genet 104:308–314. doi:10.1007/s001220100736
Yamada T, Watanabe S, Arai M, Harada K, Kitamura K (2010) Cotyledonary node pre-wounding with a micro-brush increased frequency of Agrobacterium-mediated transformation in soybean. Plant Biotechnol 27:217–220
Zhang B, Pan X, Stellwag EJ (2008) Identification of soybean microRNAs and their targets. Planta 229:161–182. doi:10.1007/s00425-008-0818-x
Zhang B, Chen P, Florez-Palacios SL, Shi A, Hou A, Ishibashi T (2010) Seed quality attributes of food-grade soybeans from the US and Asia. Euphytica 173:387–396. doi:10.1007/s10681-010-0126-y
Acknowledgments
We thank Y. Kitsui and M. Suzuki (Hokkaido University), and E. Okuda (Kyoto University) for technical assistance. This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (23580002) and by CREST of the Japan Science and Technology Agency (JAT).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Kinya Toriyama.
T. Yamada and Y. Mori contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamada, T., Mori, Y., Yasue, K. et al. Knockdown of the 7S globulin subunits shifts distribution of nitrogen sources to the residual protein fraction in transgenic soybean seeds. Plant Cell Rep 33, 1963–1976 (2014). https://doi.org/10.1007/s00299-014-1671-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-014-1671-y