Abstract
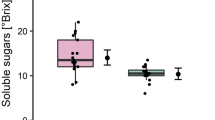
Spontaneous latex exudation is the main symptom of papaya sticky (meleira) disease caused by the Papaya meleira virus (PMeV), a double-stranded RNA (dsRNA) virus. This paper describes different effects of PMeV on papaya latex. Latex samples were subjected to different histochemical tests to evaluate their chemical composition. Additionally, the integrity of the latex particles was assessed by transmission and scanning electron microscopy analysis. Biochemical and micro- and macro-element measurements were performed. PMeV dsRNA extraction was performed to evaluate the interaction of the virus with the latex particles. Sticky diseased latex was positive for alkaloid biosynthesis and showed an accumulation of calcium oxalate crystals. PMeV also increased H2O2 synthesis within sticky diseased laticifers. The protein, sugar and water levels were altered, probably due to chemical changes. The morphology of the latex particles was further altered; PMeV particles seemed to be bound to the latex particles. The alkaloid and H2O2 biosynthesis in the papaya laticifers indicate a papaya defense response against PMeV. However, such efforts failed, as the virus affected the plant latex. The effects described here suggest some advantages of the infection process, including facilitating the movement of the virus within the papaya plant.






Similar content being viewed by others
References
An T, Huang H, Yang Z, Zhang D, Li G, Yao Y, Gao J (2001) Alkaloids from Cynanchum komarovii with inhibitory activity against the Tobacco mosaic virus. Phytochemistry 58:1267–1269
Araújo MMM, Tavares ET, Silva FR, Marinho VLA, Souza Júnior MT (2007) Molecular detection of Papaya meleira virus in the latex of Carica papaya by RT-PCR. J Virol Methods 146:305–310
Azarkan M, Dibiani R, Baulard C, Baeyens-Volant D (2006) Effects of mechanical wounding on Carica papaya cysteine endopeptidases accumulation and activity. Int J Biol Macromol 38:216–224
Borchert R (1986) Calcium acetate induces calcium uptake and formation of calcium-oxalate crystals in isolated leaflets of Gleditsia triacanthos L. Planta 168:571–578
Bouteau F, Lacrotte R, Cornel D, Monestiez M, Bousquet U, Pennarun AM, Rona JP (1991) Electrogenic active proton pump in Hevea brasiliensis laticiferous cells its role in activating sucrose/H+ and glucose/H+ symports at the plasma membrane. Bioelectrochem Bioenerg 26:223–236
Bouteau F, Bousquet U, Pennarun AM, Convert M, Dellis O, Cornel D, Ronal JP (1996) Time dependent K+ currents through plasmalemma of laticifer protoplasts from Hevea brasiliensis. Physiol Plant 98:97–104
Bouteau F, Dellis O, Bousquet U, Rona JP (1999) Evidence of multiple sugar uptake across the plasma membrane of laticifer protoplasts from Hevea. Bioelectrochem Bioenerg 48:135–139
Bravo LM, Hermosilla J, Salas CE (1994) A biochemical comparison between latex from Carica candamarcensis and C. Papaya. Braz J Med Biol Res 27:2831–2842
Calvin M (1987) Fuel oils from euphorbs and other plants. Bot J Linn Soc 94:97–110
Chaykin S, Law J, Philipis AH, Tchen TT, Bloch K (1958) Phosphorylated intermediates in the synthesis of squalene. Proc Natl Acad Sci USA 44:998–1004
Chow KS, Wan KL, Isa MNM, Baharil A, Tan SH, Harikrishna K, Yeang HY (2007) Insights into rubber biosynthesis from transcriptome analysis of Hevea brasiliensis latex. J Exp Bot 58:2429–2440
Da Cunha M, Gomes VM, Xavier-Filho J, Attias M, Souza W, Miguens FC (2000) The laticifer system of Chamaesyce thymifolia: a closed host environment for plant Trypanosomatids. Biocell 24:123–132
Davis MJ, Kramer JB, Ferwerda FH, Brunner BR (1996) Association of a bacterium and not a phytoplasm with papaya bunchy top disease. Phytopathology 86:102–109
El Moussaoui A, Nijs M, Paul C, Wintjens R, Vincentelli J, Azarkan M, Looze Y (2001) Revisiting the enzymes stored in the laticifers of Carica papaya in the context of their possible participation in the plant defense mechanism. Cell Mol Life Sci 58:556–570
Esaú K (1976) Anatomía Vegetal, 3rd edn. Ediciones Omega, Barcelona
Ferrar PH, Walker JRL (1993) o-Diphenol oxidase inhibition—an additional role for oxalic acid in the phytopathogenic arsenal of Sclerotinia sclerotiorum and Sclerotium rolfsii. Physiol Mol Plant Pathol 43:415–422
Gidrol X, Chrestin H, Tan H, Kush A (1994) Hevein, a lectin-like protein from Hevea brasiliensis (rubber tree) is involved in the coagulation of latex. J Biol Chem 269:9278–9283
Guimarães RL, Stotz HU (2004) Oxalate production by Sclerotinia sclerotiorum deregulates guard cells during infection. Plant Physiol 136:3703–3711
Hunter JR (1994) Reconsidering the functions of latex. Trees 9:1–5
Jensen WA (1962) Botanical histochemistry, principles and practice. W.H. Freeman, San Francisco
Johansen DA (1940) Plant microtechnique. McGraw-Hill, New York
Kitajima EW, Rodrigues C, Silveira J, Alves FL, Ventura JA, Aragão FJL, Oliveira LHR (1993) Association of isometric vírus-like particles, restricted to laticifers, with “meleira” (“sticky disease”) of papaya (Carica papaya). Fitopatol Bras 18:118–122
Kraus JE, Arduin M (1997) Manual básico de métodos em morfologia vegetal. EDUR, Seropédica
Kronberg K, Vogel F, Rutten T, Hajirezaei MR, Sonnewald U, Hofius D (2007) The silver lining of a viral agent: increasing seed yield and harvest index in Arabidopsis by ectopic expression of the potato leaf roll virus movement protein. Plant Physiol 145:905–918
Lane BG (1994) Oxalate, germin, and extracellular matrix of higher plants. FASEB J 5:12239–12242
Lane BG (2002) Oxalate, germins, and higher-plant pathogens. IUBMB Life 53:67–75
Lane BG, Dunwell JM, Ray JA, Schmitt MR, Cuming AC (1993) Germin, a marker protein of early plant growth, is an oxalate oxidase. J Biol Chem 268:12239–12242
Lebaudy A, Véry AA, Sentenac H (2007) K+ channel activity in plants: genes, regulations and functions. FEBS Lett 581:2357–2366
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Ma Y, Zhou T, Hong Y, Fan Z, Li H (2008) Decreased level of ferredoxin I in Tobacco mosaic virus-infected tobacco is associated with development of the mosaic symptom. doi:10.1016/j.pmpp.2008.05.004
Miller RO (1997) Nitric-perchloric acid wet digestion in an open vessel. In: Kalra YP (ed) Soil and plant analysis. CRC Press, Edmonton
Moutim V, Silva LG, Lopes MTP, Wilson Fernandes G, Salas CE (1999) Spontaneous processing of peptides during coagulation of latex from Carica papaya. Plant Sci 142:115–121
Nawrot R, Kalinowski A, Gozdzicka-Jozefiak A (2007) Proteomic analysis of Chelidonium majus milky sap using two-dimensional gel electrophoresis and tandem mass spectrometry. Phytochemistry 68:1612–1622
Orozco-Cárdenas ML, Ryan C (1999) Hydrogen peroxide is generated systemically in plant leaves by wounding and system in via the octadecanoid pathway. Proc Natl Acad Sci USA 96:6553–6557
Pickard WF (2008) Laticifers and secretory ducts: two other tube systems in plants. New Phytol 177:877–888
Purvis MJ, Collier DC, Walls D (1964) Laboratory techniques in botany. Butterwoths, London
Radwan DE, Fayez KA, Mahmoud SY, Hamad A, Lu G (2007) Physiological and metabolic changes of Cucurbita pepo leaves in response to zucchini yellow mosaic virus (ZYMV) infection and salicylic acid treatments. Plant Physiol Biochem 45:480–489
Rinne PL, van den Boogaard R, Mensink MG, Kopperud C, Kormelink R, Goldbach R, van der Schoot C (2005) Tobacco plants respond to the constitutive expression of the tospovirus movement protein NS(M) with a heat-reversible sealing of plasmodesmata that impairs development. Plant J 43:688–707
Rodrigues CH, Ventura JA, Marin SLD (1989) Ocorrência e sintomas da meleira do mamoeiro (Carica papaya) no Estado do Espírito Santo. Fitopatol Bras 14:118
Rodrigues SP, Galvão OP, Andrade JS, Ventura JA, Fernandes PMB (2005) Simple molecular method to papaya sticky disease detection from latex and tissues samples. Summa Phytopatol 31:273–275
Sajnani C, Zurita JL, Roncel M, Ortega JM, Baron M, Ducruet JM (2007) Changes in photosynthetic metabolism induced by tobamovirus infection in Nicotiana benthamiana studied in vivo by thermoluminescence. New phytol 175:120–130
Samanani N, Alcantara J, Bourgault R, Zulak KG, Facchini PJ (2006) The role of phloem sieve elements and laticifers in biosynthesis and accumulation of alkaloids in opium. Plant J 47:547–563
Schroeder JI, Hedrich R, Fernandez JM (1984) Potassium-selective single channels in guard cell protoplasts of Vicia faba. Nature 312:361–362
Scott TA Jr, Melvin EH (1953) Determination of dextran with anthrone. Anal Chem 11:1656–1661
Stuart NW (1936) Adaptation of the micro-Kjedahl method for the determination of nitrogen in plant tissues. Plant Physiol 11:173–179
Ventura JA, Costa H, Tatagiba JS (2001) Sintomatologia da meleira do mamoeiro e sua importância para o “roguing”. Fitopatol Bras 26:536
Ventura JA, Costa H, Tatagiba J, da S (2004) Papaya diseases and integrated control. In: Naqvi SAH (ed) Diseases of fruits and vegetables: diagnosis and management. Kluwer, London
Wititsuwannakul R, Pasitkul P, Kanokwiroon K, Wititsuwannakul D (2008a) A role for a Hevea latex lectin-like protein in mediating rubber particle aggregation and latex coagulation. Phytochemistry 69:339–347
Wititsuwannakul R, Rukseree K, Kanokwiroon K, Wititsuwannakul D (2008b) A rubber particle protein specific for Hevea latex lectin binding involved in latex coagulation. Phytochemistry 69:1111–1118
Wititsuwannakul R, Pasitkul P, Jewtragoon P, Wititsuwannakul D (2008c) Hevea latex lectin binding protein in C-serum as an anti-latex coagulating factor and its role in a proposed new model for latex coagulation. Phytochemistry 69:656–662
Yasue T (1969) Histochemical identification of calcium oxalate. Acta Histochem Cytochem 2:83–95
Zambolim EM, Alonso SK, Matsuoka K, Carvalho MG, Zerbini FM (2003) Purification and some properties of Papaya meleira virus, a novel virus infecting papayas in Brazil. Plant Pathol 52:389–394
Acknowledgments
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Financiadora de Estudos e Projetos (FINEP) and Fundação de Apoio à Ciência e Tecnologia do Estado do Espírito Santo (FAPES). We thank Prof. Antônio Alberto R. Fernandes for helpful comments on this manuscript. We also thank João G.Z. Piccin for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Judelson.
Rights and permissions
About this article
Cite this article
Rodrigues, S.P., Da Cunha, M., Ventura, J.A. et al. Effects of the Papaya meleira virus on papaya latex structure and composition. Plant Cell Rep 28, 861–871 (2009). https://doi.org/10.1007/s00299-009-0673-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-009-0673-7