Abstract
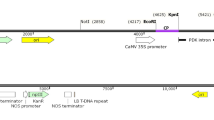
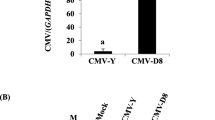
Two transgenic lines, of Nicotiana benthamiana expressing Turnip crinkle virus (TCV)-coat protein (CP) gene with contrasting phenotype, the highest (#3) and the lowest (#18) CP expressers, were selected and challenged with the homologous TCV. The former, the highest expresser, showed nearly five times more CP expression than the latter. Progenies of #3 and #18 lines showed 30 and 100% infection rates, respectively. The infected progenies of #3 line showed mild and delayed symptom with TCV. This is a coat protein-mediated resistance (CP-MR), and its resistance level is directly proportional to CP transgene expression. However, CP-MR of the transgenic plants was specific only for TCV but not for heterologous viruses. Newly growing leaves of those infected progenies of #3 line did not show any visible symptoms at 4-week post-inoculation (wpi) with TCV, suggesting a reversal from infection. This was confirmed by RT-PCR analysis with the disappearance of the target at 4 wpi. This is a case of RNA-mediated resistance, and a threshold level of transgene expression may be needed to achieve the silent state. To confirm the RNA silencing, we infiltrated Agrobacterium carrying TCV-CP into leaves of progenies of #3 and performed RT-PCR analysis. The results indicate that TCV-CP’s suppressor activity against RNA silencing itself can be silenced by the homologous expression of TCV-CP in the transgenic plants. The transgenic plants containing TCV-CP seem to be a model system to study viral protection mediated by a combination of protein and RNA silencing.





Similar content being viewed by others
References
Abel PP, Nelson RS, De B, Hoffmann N, Rogers SG, Fraley RT, Beachy RN (1986) Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 232:738–743
Baulcombe DC (1996) Mechanisms of pathogen-derived resistance to viruses in transgenic plants. Plant Cell 8:1833–1844
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363
Beachy RN (1999) Coat-protein-mediated resistance to tobacco mosaic virus: discovery mechanisms and exploitation. Philos Trans R Soc Lond B 354:659–664
Bendahmane M, Fitchen JH, Zhang G, Beachy RN (1997) Studies of coat protein-mediated resistance to tobamo mosaic tobamovirus: correlation between assembly of mutant coat proteins and resistance. J Virol 71:7942–7950
Brigneti G, Voinnet O, Li WX, Ji LH, Ding SW, Baulcombe DC (1998) Viral pathogenicity determinants are suppressors of transgene silencing in Nicotiana benthamiana. EMBO J 17:6739–6746
Bucher E, Lohuis D, van Poppel PMJA, Greerts-Dimitriadou C, Goldbach R, Prins M (2006) Multilpe virus resistance at a high frequency using a single transgene construct. J Gen Virol 87:3697–3701
Choi CW, Qu F, Ren T, Ye X, Morris TJ (2004) RNA silencing-suppressor function of Turnip crinkle virus coat protein cannot be attributed to its interaction with the Arabidopsis protein TIP. J Gen Virol 85:3415–3420
Clark MF, Lister RM, Bar-Joseph M (1986) ELISA techniques. Methods Enzymol 118:742–766
Clark WG, Fitchen JH, Beachy RN (1995) Studies of coat-protein mediated resistance to TMV using mutant CP. I. The PM2 assembly defective mutant. Virology 208:485–491
Cooley MB, Pathirana S, Wu HJ, Kachroo P, Klessig DF (2000) Members of the Arabidopsis HRT/RPP8 family of resistance genes confer resistance to both viral and oomycete pathogens. Plant Cell 12:663–676
de Hann P, Gielen JJL, Prins M, Wijkamp IG, van Schepen A, Peters D, van Grinsven MQJM, Goldbach R (1992) Characterization of RNA-mediated resistance to tomato spotted wilt virus in transgenic tobacco plants. Bio/Technology 10:1133–1137
Ding SW (2000) RNA silencing. Curr Opin Biotechnol 11:152–156
Ding SW, Li H, Lu R, Li F, Li WX (2004) RNA silencing: a conserved antiviral immunity of plants and animals. Virus Res 102:109–115
Dougherty WG, Lindbo JA, Smith HA, Parks TD, Swaney S, Proebsting WM (1994) RNA-mediated virus resistance in transgenic plants: exploitation of a cellular pathway possibly involved in RNA degradation. Mol Plant Microbe Interact 7:544–552
English JJ, Mueller E, Baulcombe DC (1996) Suppression of virus accumulation in transgenic plants exhibiting silencing of nuclear genes. Plant Cell 8:179–188
Fitchen JH, Beachy RN (1993) Genetically engineered protection against viruses in transgenic plants. Annu Rev Microbiol 4:739–763
Garcia S, Billecocq A, Crance J-M, Prins M, Garin D, Bouloy M (2006) Viral suppressors of RNA interference impair RNA silencing induced by a Semliki Forest virus replicon in tick cells. J Gen Virol 87:1985–1989
Goldbach R, Bucher E, Prins M (2003) Resistance mechanisms to plant viruses: an overview. Virus Res 92:207–212
Goodwin J, Chapman K, Swaney S, Parks TD, Wernsman EA, Dougherty WG (1996) Genetic and biochemical dissection of transgenic RNA-mediated virus resistance. Plant Cell 8:95–105
Hacker DL, Petty ITD, Wei N, Morris TJ (1992) Turnip crinkle virus genes required for RNA replication and virus movement. Virology 186:1–8
Hammond J, Kamo KK (1993) Transgenic coat protein and antisense RNA resistance to bean yellow mosaic potyvirus. Acta Hortic 336:171–178
Hammond J, Lecoq H, Raccah B (1999) Epidemiological risks from mixed virus infections and transgenic plants expressing viral genes. Adv Virus Res 54:189–314
Hefferon KL, Khalilian H, AbouHaidar MG (1997) Expression of the PVYO coat protein (CP) under the control of the PVX CP gene leader sequence: protection under greenhouse and field conditions against PVYO and PVYN infection in three potato cultivars. Theor Appl Genet 94:287–292
Hemmes H, Lakatos L, Goldbach R, Burgyán J, Prins M (2007) The NS3 protein of Rice hoja blanca tenuivirus suppresses RNA silencing in plant and insect hosts by efficiently binding both siRNAs and miRNAs. RNA 13:1079–1089
Hollings M, Stone OM (1972) Turnip crinkle virus. In: AAB descriptions of plant viruses 109. Association of Applied Biologists, Wellesbourne
Hong Y, Levay K, Murphy JF, Klein PG, Shaw JG, Hunt AG (1996) A potyvirus polymerase interacts with the viral coat protein and VPg in yeast cells. Virology 214:159–166
Lawson C, Kaniewski W, Haley L, Rozman R, Newell C, Sanders P, Tumer NE (1990) Engineered resistance to mixed virus infection in a commercial potato cultivar: resistance to potato virus X and potato virus Y in transgenic Russet Burbank. Bio/Technology 8:127–134
Li WZ, Qu F, Morris TJ (1998) Cell-to-cell movement of turnip crinkle virus is controlled by two small reading frames that function in trans. Virology 244:405–416
Li WX, Li H, Lu R, Li F, Dus M, Atkinson P, Brydon EW, Johnson KL, García-Sastre A, Ball LA, Palese P, Ding SW (2004) Interferon antagonist proteins of influenza and vaccinia viruses are suppressors of RNA silencing. Proc Natl Acad Sci USA 101:1350–1355
Lin SS, Henriques R, Wu HW, Niu QW, Yeh SD, Chua NH (2007) Strategy and mechanisms of plant virus resistance. Plant Biotechnol Rep 1:125–134
Lindbo JA, Dougherty WG (1992) Untranslatable transcripts of the tobacco etch virus coat protein gene sequence can interfere with tobacco etch virus replication in transgenic plants and protoplasts. Virology 189:725–753
Lindbo JA, Silva-Rosales L, Proebsting WM, Dougherty WG (1993) Induction of a highly specific antiviral state in transgenic plants: implications for regulation of gene expression and virus resistance. Plant Cell 5:1749–1759
Loesch-Fries LS, Merlo D, Zinnen T, Burhop L, Hill K, Krahn K, Jarvis N, Nelson S, Halk E (1987) Protection against tobacco mosaic virus infection in transgenic plants requires accumulation of coat protein rather than coat protein RNA sequences. EMBO J 6:1845–1851
Lomonossoff GP (1995) Pathogen-derived resistance to plant viruses. Annu Rev Phytopathol 33:323–343
Lu S, Cullen BR (2004) Adenovirus VA1 noncoding RNA can inhibit small interfering RNA and microRNA biogenesis. J Virol 78:12868–12876
Lu B, Stubbs G, Culver JN (1998) Coat protein interactions involved in tobacco mosaic tobamovirus cross-protection. Virology 248:188–198
Malnoe P, Farinelli L, Collet GF, Reust W (1994) Small-scale field tests with transgenic potato, cv. Bintje, to test resistance to primary and secondary infections with potato virus Y. Plant Mol Biol 25:963–975
Mueller E, Gilbert JE, Davenport G, Brigneti G, Baulcombe DC (1995) Homology-dependent resistance: transgenic virus resistance in plants related to homology-dependent gene silencing. Plant J 7:1001–1023
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497
Namba S, Ling K, Gonsalves C, Slightom JL, Gonsalves D (1992) Protection of transgenic plants expressing the coat protein gene of watermelon mosaic virus II or zucchini yellow mosaic virus against six potyviruses. Phytopathology 82:940–946
Nejidat A, Beachy RN (1990) Transgenic tobacco plants expressing a coat protein gene of tobacco mosaic virus are resistant to some other tobamoviruses. Mol Plant Microbe Interact 3:247–251
Pang SZ, Bock JH, Gonsalves C, Slightom JL, Gpnsalves D (1994) Resistance of transgenic Nicotiana benthamiana plants to tomato spotted wilt and impatiens necrotic spot tospoviruses: evidence of involvement of the N protein and N gene RNA in resistance. Phytopathology 84:243–249
Powell PA, Nelson RS, De B, Hoffmann N, Rogers SG, Fraley RT, Beachy RN (1986) Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 232:738–743
Powell PA, Sanders PR, Turner N, Fraley RT, Beachy RN (1990) Protection against tobacco mosaic virus infection in transgenic plants requires accumulation of coat protein rather than coat protein RNA sequences. Virology 175:124–130
Qu F, Ren T, Morris TJ (2003) The coat protein of Turnip crinkle virus suppresses posttranscriptional gene silencing at an early initiation step. J Virol 77:511–522
Ratcliff F, Harrison BD, Baulcombe DC (1997) A similarity between viral defense and gene silencing in plants. Science 276:1558–1560
Ratcliff F, McFarlane SA, Baulcombe DC (1999) Gene silencing without DNA. RNA-mediated cross-protection between viruses. Plant Cell 11:1207–1216
Ravelonandro M, Monsion M, Teycheney PY, Delbos PR, Dunez J (1992) Transgenic tobacco plants that contain the plum pox virus (PPV) coat protein gene. Acta Hortic 309:191–196
Register JC, Beachy RN (1988) Resistance to TMV in transgenic plants results from interference with an early event in infection. Virology 166:524–532
Ren T, Qu F, Morris TJ (2000) HRT gene function requires interaction between a NAC protein and viral capsid protein to confer resistance to Turnip crinkle virus. Plant Cell 12:1917–1925
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sanford JC, Johnston SA (1985) The concept of parasite-derived resistance. Deriving resistance genes from the parasite’s own genome. J Theor Biol 113:395–405
Savenkov EI, Valkonen JPT (2002) Silencing of a viral RNA silencing suppressor in transgenic plants. J Gen Virol 83:2325–2335
Smith HA, Powers H, Swaney S, Brown C, Dougherty WG (1995) Transgenic potato virus Y resistance in potato: evidence for an RNA-mediated cellular response. Phytopathology 85:864–870
Sontheimer EJ (2005) Assembly and function of RNA silencing complexes. Nat Rev Mol Cell Biol 6:127–138
Stark DM, Beachy RN (1989) Protection against potyvirus infection in transgenic plants: evidence for broad spectrum resistance. Bio/Technology 7:1257–1262
Takeda A, Tsukuda M, Mizumoto H, Okamoto K, Kaido M, Mise K, Okuno T (2005) A plant virus suppresses RNA silencing through viral RNA replication. EMBO J 24:3147–3157
Tenllado F, Garcia-Luque I, Serra MT, Diaz-Ruiz JR (1995) Nicotiana benthamiana plants transformed with the 54-kDa region of the pepper mild mottle tobamovirus replicase gene exhibit two types of resistance responses against viral infection. Virology 211:170–183
Tennant PF, Gonsalves C, Ling KS, Fitch M, Manshardt R, Slightom JL, Gonsalves D (1994) Different protection against papaya ringspot virus isolates in coat protein gene transgenic papaya and classically cross-protected papaya. Phytopathology 84:1359–1366
Thomas CL, Leh V, Lederer C, Maule A (2003) Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana. Virology 306:33–41
van der Vlugt RAA, Ruiter RK, Goldbach R (1992) Evidence for sense RNA-mediated resistance to PVYN in tobacco plants transformed with the viral coat protein cistron. Plant Mol Biol 20:631–639
Vance V, Vaucheret H (2001) RNA silencing in plants-defense and counterdefense. Science 292:2277–2280
Voinnet O (2005) Induction and suppression of RNA silencing: insights from viral infections. Nat Rev Genet 6:206–220
Voloudakis AE, Aleman-Verdaguer ME, Padgett HS, Beachy RN (2005) Characterization of resistance in transgenic Nicotiana benthamiana encoding N-terminal deletion and assembly mutants of the Tobacco etch potyvirus coat protein. Arch Virol 150:2567–2582
Wang MB, Metzlaff M (2005) RNA silencing and antiviral defense in plants. Curr Opin Plant Biol 8:216–222
Wilson TM (1993) Strategies to protect crop plants against viruses: pathogen-derived resistance blossoms. Proc Natl Acad Sci USA 90:3134–3141
Worrall D, Elias L, Ashford D, Smallwood M, Sidebottom C, Lillford P, Telford J, Holt C, Bowles D (1998) A carrot leucine-rich-repeat protein that inhibits ice recrystallisation. Science 282:115–117
Acknowledgments
This work was supported by a grant (R05-2003-000-10293-0) from the Basic Research Program of the Korea Research Foundation (KRF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Lakshmanan.
Ayyappan Vasudevan and Tae-Kyun Oh have contributed equally in this study.
Rights and permissions
About this article
Cite this article
Vasudevan, A., Oh, TK., Park, J.S. et al. Characterization of resistance mechanism in transgenic Nicotiana benthamiana containing Turnip crinkle virus coat protein. Plant Cell Rep 27, 1731–1740 (2008). https://doi.org/10.1007/s00299-008-0595-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-008-0595-9