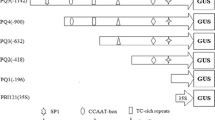
Abstract
Arabidopsis harbors two α and two β genes of pyrophosphate:fructose-6-phosphate 1-phosphotransferase (PFP). The spatial expression patterns of the two AtPFPα genes were analyzed using transgenic plants containing a promoter::ß-glucuronidase (GUS) fusion construct. Whereas the AtPFPα1 promoter was found to be ubiquitously active in all tissues, the AtPFPα2 promoter is preferentially expressed in specific heterotrophic regions of the Arabidopsis plant such as the trichomes of leaves, cotyledon veins, roots, and the stamen and gynoecium of the flowers. Serial deletion analysis of the AtPFPα2 promoter identified a key regulatory element from nucleotides −194 to −175, CGAAAAAGGTAAGGGTATAT, which we have termed PFPα2 and which is essential for AtPFPα2 gene expression. Using a GUS fusion construct driven by this 20-bp sequence in conjunction with a −46 CaMV35S minimal promoter, we also demonstrate that PFPα2 is sufficient for normal AtPFPα2 expression. Hence, this element can not only be used to isolate essential DNA-binding protein(s) that control the expression of the carbon metabolic enzyme AtPFPα2, but has also the potential to be utilized in the production of useful compounds in a specific organ such as the leaf trichomes.






Similar content being viewed by others
References
Alves AM, Meijer WG, Vrijbloed JW, Dijkhuizen L (1996) Characterization and phylogeny of the pfp gene of Amycolatopsis methanolica encoding PPi-dependent phosphofructokinase. J Bacteriol 178:149–155
Benfey PN, Chua NH (1990) The cauliflower mosaic virus-35s promoter - combinatorial regulation of transcription in plants. Science 250:959–966
Black CC, Mustardy L, Sung SS, Kormanik PP, Xu D-P, Paz N (1987) Regulation and roles for alternative pathways of hexose metabolism in plants. Physiol Plant 69:387–394
Buchel AS, Brederode FT, Bol JF, Linthorst HJ (1999) Mutation of GT-1 binding sites in the Pr-1A promoter influences the level of inducible gene expression in vivo. Plant Mol Biol 40:387–396
Carlisle SM, Blakeley SD, Hemmingsen SM, Trevanion SJ, Hiyoshi T, Kruger NJ, Dennis DT (1990) Pyrophosphate-dependent phosphofructokinase. Conservation of protein sequence between the α- and β-subunits and with the ATP-dependent phosphofructokinase. J Biol Chem 265:18366–18371
Claassen PAM, Budde MA, de Ruyter HJ, van Calker MH, van Es A (1991) Potential role of pyrophosphate:fructose-6-phosphate phosphotransferase in carbohydrate metabolism of cold stored tubers of Solanum tuberosum cv Bintje. Plant Physiol 95:1243–1249
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Dennis DT, Greyson MF (1987) Fructose-6-phosphate metabolism in plants. Physiol Plant 69:395–404
Duke SO, Canel C, Rimando AM, Tellez MR, Duke MV, Paul RM (2000) Current and potential exploitation of plant glandular trichome productivity. In: Hallahan DL, Gray JC (eds) Advances in botanical research, vol 31. Academic Press, San Diego, pp 121–151
Eisner T, Eisner M, Hoebeke ER (1998) When defense backfires: detrimental effect of a plant’s protective trichomes on an insect beneficial to the plant. Proc Natl Acad Sci USA 95:4410–4414
Fahrendorf T, Holtum JA, Mukherjee U, Latzko E (1987) Fructose-2,6-bisphosphate, carbohydrate partitioning and crassulacean acid metabolism. Plant Physiol 84:182–187
Fuse T, Sasaki T, Yano M (2001) Ti-plasmid vectors useful for functional analysis of rice genes. Plant Biotechnol 18:219–222
Gutiérrez-Alcal G, Calo L, Gros F, Caissard JC, Gotor C, Romero LC (2005) A versatile promoter for the expression of proteins in glandular and non-glandular trichomes from a variety of plants. J Exp Bot 56:2487–2494
Hajirezaei M, Sonnewald U, Viola R, Carlisle S, Dennis D, Stitt M (1994) Transgenic potato plants with strongly decreased expression of pyrophosphate:fructose-6-phosphate phosphotransferase show no visible phenotype and only minor changes in metabolic fluxes in their tubers. Planta 192:16–30
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Jeon J-S, Chung Y-Y, Lee S, Yi G-H, Oh B-G, An G (1999) Isolation and characterization of an anther-specific gene, RA8, from rice (Oryza sativa L.). Plant Mol Biol 39:35–44
Kim HJ, Triplett BA (2001) Cotton fiber growth in planta and in vitro. Models for plant cell elongation and cell wall biogenesis. Plant Physiol 127:1361–1366
Kruger NJ, Dennis DT (1987) Molecular properties of pyrophosphate:fructose-6-phosphate phosphotransferase from potato tuber. Arch Biochem Biophys 256:273–279
Larkin JC, Oppenheimer DG, Pollock S, Marks MD (1993) Arabidopsis GLABROUS1 gene requires downstream sequences for function. Plant Cell 5:1739–1748
Liu HC, Creech RG, Jenkins JN, Ma DP (2000) Cloning and promoter analysis of the cotton lipid transfer protein gene Ltp3. Biochim Biophys Acta 1487:106–111
Mena M, Cejudo FJ, Isabel-Lamoneda I, Carbonero P (2002) A role for the DOF transcription factor BPBF in the regulation of gibberellin-responsive genes in barley aleurone. Plant Physiol 130:111–119
Mertens E (1990) Occurrence of pyrophosphate:fructose 6-phosphate 1-phosphotransferase in Giardia lamblia trophozoites. Mol Biochem Parasitol 40:147–149
Morita A, Umemura T, Kuroyanagi M, Futsuhara Y, Perata P, Yamaguchi J (1998) Functional dissection of a sugar-repressed α-amylase gene (RAmy1 A) promoter in rice embryos. FEBS Lett 423:81–85
Nielsen TH, Rung JH, Villadsen D (2004) Fructose-2,6-bisphosphate: a traffic signal in plant metabolism. Trends Plant Sci 9:556–563
Nielsen TH, Stitt M (2001) Tobacco transformants with strongly decreased expression of pyrophosphate:fructose-6-phosphate expression in the base of their young growing leaves contain much higher levels of fructose-2,6-bisphosphate but no major changes in fluxes. Planta 214:106–116
Park HC, Kim ML, Kang YH, Jeon JM, Yoo JH, Kim MC, Park CY, Jeong JC, Moon BC, Lee JH, Yoon HW, Lee S-H, Chung WS, Lim CO, Lee SY, Hong JC, Cho MJ (2004) Pathogen- and NaCl-induced expression of the SCaM-4 promoter is mediated in part by a GT-1 box that interacts with a GT-1-like transcription factor. Plant Physiol 135:2150–2161
Podesta FE, Plaxton WC (2003) Fluorescence study of ligand binding to potato tuber pyrophosphate-dependent phosphofructokinase: evidence for competitive binding between fructose-1,6-bisphosphate and fructose-2,6-bisphosphate. Arch Biochem Biophys 414:101–107
Spilatro SR, Anderson JM (1989) Cabohydrate metabolism and activity of pyrophosphate:fructose-6-phosphate phosphotransferase in photosynthetic soyabean (Glycine max, Merr.) suspension cells. Plant Physiol 88:862–888
Stitt M (1990) Fructose-2,6-bisphosphate as a regulatory molecule in plants. Annu Rev Plant Physiol Plant Biol 41:153–158
Theodorou ME, Cornel FA, Duff SM, Plaxton WC (1992) Phosphate starvation-inducible synthesis of the α-subunit of the pyrophosphate-dependent phosphofructokinase in black mustard suspension cells. J Biol Chem 267:21901–21905
Todd JF, Blakeley SD, Dennis DT (1995) Structure of the genes encoding the α- and β-subunits of castor pyrophosphate-dependent phosphofructokinase. Gene 152:181–186
Traw MB, Bergelson J (2003) Interactive effects of jasmonic acid, salicylic acid, and gibberellin on induction of trichomes in Arabidopsis. Plant Physiol 133:1367–1375
Wang E, Gan S, Wagner GJ (2002) Isolation and characterization of the CYP71D16 trichome-specific promoter from Nicotiana tabacum L. J Exp Bot 53:1891–1897
Wang YH, Shi JN (1999) Evidence that fructose 1,6-bisphosphate specifically protects the α-subunit of pyrophosphate-dependent 6-phosphofructo-1-phosphotransferase against proteolytic degradation. FEBS Lett 459:448–452
Xu DP, Sung SJ, Black CC. (1989) Sucrose metabolism in Lima black beans. Plant Physiol 89:1100–1116
Yanagisawa S (2000) Dof1 and Dof2 transcription factors are associated with expression of multiple genes involved in carbon metabolism in maize. Plant J 21:281–288
Yanagisawa S, Schmidt RJ (1999) Diversity and similarity among recognition sequences of Dof transcription factors. Plant J 17:209–214
Zhou DX (1999) Regulatory mechanism of plant gene transcription by GT-elements and GT-factors. Trends Plant Sci 4:210–214
Acknowledgments
This work was supported, in part, by grants from the SRC program of MOST/KOSEF(R11-2000-081) through the Plant Metabolism Research Center, from the Crop Functional Genomics Center (CFGC) of the 21st Century Frontier Research Program (CG1422, CG1111), from the Biogreen 21 program, Rural Development Administration, and from the BK21 program, Ministry of Education.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. T. Kim
Rights and permissions
About this article
Cite this article
Lim, HM., Cho, JI., Lee, S. et al. Identification of a 20-bp regulatory element of the Arabidopsis pyrophosphate:fructose-6-phosphate 1-phosphotransferase α2 gene that is essential for expression. Plant Cell Rep 26, 683–692 (2007). https://doi.org/10.1007/s00299-006-0272-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-006-0272-9