Abstract
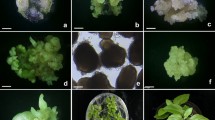
Embryogenic cell suspension cultures of garlic (Allium sativum L.) were initiated in liquid medium from friable embryogenic tissue. The optimal parameters for culture maintenance were: (1) an initial cell density of 1–4% (v/v); (2) medium renewal every 14 days and subculturing every 28 days; (3) a low 2,4-dichlorophenoxyacetic acid concentration (0.1–0.3 mg/l). Cultures regenerated during a 14-month period. The cell suspension cultures differentiated embryos following transfer to a semi-solid embryo induction medium, with histological studies confirming and characterising the embryogenic nature of the process. Forty percent of these embryos converted into plantlets, which produced micro bulbs in vitro. The composition of the sulphur compounds of the micro bulbs obtained from cell suspension embryo-derived plantlets differed slightly from those produced by in vitro shoot proliferation-derived plantlets, but after two cycles of multiplication in the field these differences had disappeared.






Similar content being viewed by others
Abbreviations
- BAP:
-
6-Benzylaminopurine
- CS:
-
Cell suspension-derived plantlet
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- IAA:
-
Indole-3-acetic acid
- NAA:
-
α-Naphthaleneacetic acid
- SCV:
-
Sedimented cell volume
- SP:
-
In vitro shoot proliferation-derived plantlet
References
Al Zahim MA, Ford Lloyd BV, Newbury HJ (1999) Detection of somaclonal variation in garlic (Allium sativum L.) using RAPD and cytological analysis. Plant Cell Rep 18:473–477
Ammirato P (1983) The regulation of somatic embryo development in plant cell cultures: Suspension culture techniques and hormone requirements. Biotechnology 1:68–74
Arnault I, Christidès JP, Mandon N, Kahane R, Haffner T, Auger J (2003) High performance ion pair chromatography method for simultaneous analysis of alliin, deoxyalliin, allicin and dipeptide precursors in garlic products using multiple mass spectrometry and UV. J Chromatogr A 991:69–75
Ayabe M (2001) A novel and efficient tissue culture method - “stem-disc dome culture” - for producing virus-free garlic (Allium sativum L.). Plant Cell Rep 20:503–507
Barandiaran X, Martin N, Rodriguez Conde MF, Pietro Ad, Martin J (1999) An efficient method for callus culture and shoot regeneration of garlic (Allium sativum L.). HortScience 34:348-349
Barrueto Cid L, Illg R, Piedrabuena A (1994) Regeneration of garlic plants (Allium sativum L., cv “chonan”) via cell culture in liquid medium. In Vitro Cell Dev Biol-Plant 30:150–155
Chovelon V, Leroux JP, Dore C (1990) Sélection sanitaire de l’ail et de l’échalote: culture de méristèmes et régénération de variétés. In: Doré C (ed) Cinquantenaire de la culture in vitro. Colloques de l’INRA 51:142–150
Chu C, Wang C, Sun C, Hsu C, Yin K, Chu C, Bi F (1975) Establishment of an efficient medium for anther culture of rice through comparative experiments of the nitrogen source. Sci Sin 118:659–668
Cote F, Domergue R, Monmarson S, Schwendiman J, Teisson C, Escalan J (1996) Embryogenic cell suspension from the male flower of Musa AAA cv. Grand nain. Physiol Plant 97:285–290
Dolores LM, Patena LF, Barg E, Green SK (2002) Detection of Allium viruses in garlic and shallots using serological and molecular assay. Philos J Crop Sci 27:18
Etoh T, Simon PW (2002) Diversity, fertility and seed production of garlic. In: Rabinowitch HD (ed) Allium crop science: recent advances. CABI Pub, Wallingford, pp 101–117
Fereol L, Chovelon V, Causse S, Michaux-Ferriere N, Kahane R (2002) Evidence of a somatic embryogenesis process for plant regeneration in garlic (Allium sativum L.). Plant Cell Rep 21:197–203
Gamborg O, Miller R, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root. Cells 50:151–158
Haque MS, Wada T, Hattori K (1997) High frequency shoot regeneration and plantlet formation from root tip of garlic. Plant Cell Tissue Organ Cult 50:83–89
Hari V (1980) Effect of cell density changes and conditioned media on carrot cell embryogenesis. Z Pflanzenphysiol 96:227–231
Heller R (1953) Recherches sur la nutrition minerale des tissus végétaux cultivés in vitro. Ann Sci Natl Biol Veg 14:1–223
Kahane R, Rancillac M, Schweisguth B (1992) Bulbing in vitro in Allium species. Allium Improve Newsl 2:18–20
Kamenetsky R, Rabinowitch HD (2001) Floral development in bolting garlic. Sex Plant Rep 13:235–241
Kim EK, Hahn EJ, Murthy HN, Paek KY (2003) High frequency of shoot multiplication and bulblet formation of garlic in liquid cultures.. Plant Cell Tissue Organ Cult 73:231–236
Lot H, Delécolle B, Bocardo G, Marzachi C, Milne R (1994) Partial characterization of retrovirus-like particles associated with garlic dwarf disease. Plant Pathol 43:537–546
Messiaen C, Cohat J, Leroux J, Pichon M, Beyries A (1993) Les Allium alimentaires reproduits par voie végétative. INRA, Paris
Murashige T, Skoog F (1962) A revised medium for rapid growth and biosassay with tobacco tissue culture. Physiol Plant 15:473–497
Myers J, Simon PW (1998) Continuous callus production and regeneration of garlic (Allium sativum L.) using root segments from shoot tip-derived plants. Plant Cell Rep 17:726–730
Nagakubo T, Nagasawa A, Ohkawa H (1993) Micropropagation of garlic through in vitro bulblet formation. Plant Cell Tissue Organ Cult 32:175–183
Nagasawa A, Finer J (1988) Development of morphogenic suspension cultures of garlic (Allium sativum L.). Plant Cell Tissue Organ Cult 15:183–187
Novak FJ (1990) Allium tissue culture. In: Rabinovitch HD, Brewster JL (eds) Onions and allied crops, vol. 2. CRC Press, Boca Raton, pp 233–250
Phillips G, Luteyn K (1983) Effects of picloram and other auxins on onion tissue cultures. J Am Soc Hortic Sci 108:948-953
Robledo Paz A, Villalobos Arambula VM, Jofre Garfias AE (2000) Efficient plant regeneration of garlic (Allium sativum L.) by root- tip culture. In Vitro Cell Dev Biol–Plant 36:416–419
SAS Institute (1982) SAS user’s guide: statistics. SAS Institute, Cary, N.C.
Senula A, Keller ERJ, Leseman DE, Cassells AC, Doyle BM, Curry RF (2000) Elimination of viruses through meristem culture and thermotherapy for the establishment of an in vitro collection of garlic (Allium sativum). Acta Hortic 530:121–128
Xue HM, Araki H, Shi L, Yakuwa T (1991) Somatic embryogenesis and plant regeneration in basal plate and receptacle derived-callus cultures of garlic (Allium sativum L.). J Jpn Soc Hortic Sci 60:627–634
Yasuda H, Nakajima M, Masuda H, Ohwada T (2000) Direct formation of heart-shaped embryos from differentiated single carrot cells in culture. Plant Sci 152:1–6
Zheng SJ, Henken B, Krens FA, Kik C (2003) The development of an efficient cultivar-independent plant regeneration system from callus derived from both apical and non-apical root segments of garlic (Allium sativum L.). In Vitro Cell Dev Biol-Plant 39:288–292
Acknowledgements
This work was supported by a grant from the EU for the Garlic & Health research program of the 5th PCRDT, Quality of Life (QLK1-1999-00498). We thank the CIRAD-FLHOR Biometrics Department (Xavier Perrier, Cécile Dubois, Jean Pierre Jacquemoud) for the statistical analyses
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Beckert
Rights and permissions
About this article
Cite this article
Fereol, L., Chovelon, V., Causse, S. et al. Establishment of embryogenic cell suspension cultures of garlic (Allium sativum L.), plant regeneration and biochemical analyses. Plant Cell Rep 24, 319–325 (2005). https://doi.org/10.1007/s00299-005-0937-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-005-0937-9