Abstract
A procedure has been established for Agrobacterium tumefaciens-mediated genetic transformation of Hevea brasiliensis embryogenic friable calli. Precultivation of tissues on a CaCl2-free maintenance medium dramatically enhanced the transient activity of the reporter gene, gusA encoding β-glucuronidase (GUS). The increase was first noticed in highly active cells (undifferentiated or/and embryogenic), in tissues precultured for 2–8 weeks. Beyond 8 weeks of preculture, GUS activity increased again, but this time in tissues consisting of differentiated cells accumulating polyphenols. Out of five Agrobacterium strains cocultivated with CaCl2-free precultured tissues, only inoculation with EHA105pC2301 led to high transient GUS activity. Paromomycin proved more effective than kanamycin for the selection of transformed cells, as it inhibits the growth of non-transformed cells more radically. Five paromomycin-resistant callus lines were established. The presence of gusA and neomycin phosphotransferase (nptII) genes in the plant genome was confirmed by DNA amplification, and by Southern hybridization. These results confirmed that A. tumefaciens is an effective system for mediating stable transformation of rubber tree calli with a low copy number of transgenes. Transgenic callus lines constitute a useful tool for studying genes of interest on a cellular level and for regenerating transgenic rubber trees.


Similar content being viewed by others
Abbreviations
- DM:
-
Decontamination medium
- FM :
-
Fresh matter
- GUS :
-
β-Glucuronidase
- LSD:
-
Least significant difference
- MM :
-
Maintenance medium
- NBB :
-
Naphthol blue-black stain
- PAS :
-
Periodic acid-Schiff stain
- PCR:
-
Polymerase chain reaction
- RGR :
-
Relative growth rate
- SDS :
-
Sodium dodecyl sulphate
- TCL :
-
Transgenic callus line
References
Arokiaraj P, Jones H, Cheong KF, Coomber S, Charlwood BV (1994) Gene Insertion into Hevea brasiliensis. Plant Cell Rep 13:425–31
Arokiaraj P, Jones H, Jaafar H, Coomber S, Charlwood BV (1996) Agrobacterium-mediated transformation of Hevea anther calli and their regeneration into plantlets. J Nat Rubber Res 11(2):77–86
Arokiaraj P, Yeang HY, Cheong KF, Hamzah S, Jones H, Coomber S, Charlwood BV (1998) CaMV 35 S promoter directs β-glucuronidase expression in the laticiferous system of transgenic Hevea brasiliensis (rubber tree). Plant Cell Rep 17:621–625
Azhakanandam K, McCabe MS, Power JB, Lowe KC, Cocking EC, Davey MR (2000) T-DNA transfer, integration, expression and inheritance in rice: effects of plant genotype and agrobacterium super-virulence. J Plant Physiol 157:429–439
Bassie L, Noury M, Wisniewski J-P, Topson L, Christou P, Capell T (2000) Transgenic cell lines as a useful tool to study the biochemistry of down-regulation of an endogenous rice gene using a heterologous diamine-oxidase cDNA. Plant Physiol Biochem 38:729–737
Bhatnagar P, Glasheen BM, Bains SK, Long SL, Moinocha R, Walter C, Minocha SC (2001) Transgenic manipulation of the metabolism of polyamines in poplar cells. Plant Physiol 125:2139–2153
Bzrozowska J, Hanover P , Chezeau R (1974) Free amino acids of Hevea brasiliensis latex. Experientia 30:894–896
Carron MP (2001) L'embryogenèse somatique de l'hévéa. In: IRRDB meeting, Montpellier, France. CIRAD
Carron M-P, Dea GB, Tison J, Leconte A, Keli J (1997) Croissance en champ de clones d'Hevea brasiliensis produits par culture in vitro. Plant Rech Dév 4:264–273
Carron MP, Etienne H, Lardet L, Campagna S, Perrin Y, Leconte A, Chaine C (1995) Somatic embryogenesis in rubber (Hevea brasiliensis Müll. Arg.). In: Jain SM, Gupta PK, Newton RJ (eds) Somatic embryogenesis in woody plants. Kluwer, Dordrecht, pp 117–136
Carron M-P, Lardet L, Dea GB (1998) La micropropagation de l'Hévéa par embryogenèse somatique. Plant Rech Dév 5:187–92
Cervera M, Pina JA, Juarez J, Navarro L, Pena L (1998) Agrobacterium-mediated transformation of citrange: factors affecting transformation and regeneration. Plant Cell Rep 18:271–278
Chair H, Kuhapituktum R, Attathom T, Pannetier C, Attathom S (1997) Agrobacterium mediated transformation of Sri Sumrong 60, a Thai cotton variety. Kasetsart J (Nat Sci) 33:149–156
Cheng M, Fry JE, Pang SZ, Zhou HP, Hironaka CM, Duncan DR, Conner TW, Wan YC (1997) Genetic transformation of wheat mediated by Agrobacterium tumefaciens. Plant Physiol 115:971–980
Chye ML, Cheung KY (1995) Beta-1,3-glucanase is highly-expressed in laticifers of Hevea brasiliensis. Plant Mol Biol 29:397–402
D'Auzac J, Jacob JL, Chrestin H (eds) (1989) Physiology of rubber tree latex. CRC Press, Boca Raton, Fla., USA
Deblaere R, Bytebier B, De Greve H, Deboeck F, Schell J, Van Montagu M, Leemans J (1985) Efficient octopine Ti plasmid-derived vectors for Agrobacterium-mediated gene transfer to plants. Nucleic Acids Res 13:4777–4788
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version 2. Plant Mol Biol Rep 1:19–22
Edwards K, Johnstone C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA for PCR analysis. Nucleic Acids Res 19:1349
Eklund L, Eliasson L (1990) Effects of calcium ion concentration on cell wall synthesis. J Exp Bot 41:863–867
Etienne H, Berger A, Carron MP (1991a) Water status of callus from Hevea brasiliensis during induction of somatic embryogenesis. Physiol Plant 82:213–218
Etienne H, Montoro P, Carron M-P (1991b) Incidence des paramètres hydriques sur le développement des cals d'Hevea brasiliensis en culture in vitro. Ann Sci For 48:253–265
Fisher D (1968) Protein staining of ribboned open sections for light microscopy. Histochemie 16:92–96
Foolad MR, Arulsekar S, Rodriguez RL (1995) Applications of polymerase chain reaction (PCR) to plant genome analysis. In: Gamborg OL,Phillips GC (eds) Plant cell, tissue and organ culture. Fundamental methods. Springer, New York Berlin Heidelberg, pp 281–298
Gidrol X, Chrestin H, Tan HL, Kush A (1994) Hevein, a lectin-like protein from Hevea brasiliensis (rubber tree) is involved in the coagulation of latex. J Biol Chem 269:9278–9283
Hajdukiewicz P, Svap Z, Maliga P (1994) The small versatile pPZP family of Agrobacterium binary vectors for plant transformation. Plant Mol Biol 25:989–994
Hoekema A, Hirsch PR, Hooykaas PJJ, Schilperoort RA (1983) A binary plant vector strategy based on separation of vir- and T-region of the Agrobacterium tumefaciens Ti plasmid. Nature 303:179–80
Hood EE, Gelvin SB, Melchers LS, Hoekema A (1993) New Agrobacterium helper plasmids for gene transfer to plants. Transgenic Res 2:208–218
Jacob JL, Prévôts JC, Lacote R, Gohet E, Clément A, Gallois R, Joet T, Pujade-Renaud V, D'Auzac J (1998) The biological mechanisms controlling Hevea brasiliensis rubber yield. Plantation, research, development 5:5–17
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive gene fusion marker in higher plants. EMBO J 6:3901–3907
Katen A de, Jacobsen H-J (1995) Cell competence for Agrobacterium -mediated DNA transfer in Pisum sativum L. Transgenic Res 4:184–191
Koncz C, Schell J (1986) The promoter of Tl-DNA gene 5 controls the tissue-specific expression of chimeric genes carried by a novel type of Agrobacterium binary vector. Mol Breed 204:383–396
Le QV, Bogusz D, Gherbi H, Lappartient A, Dohoux E, Franche C (1996) Agrobacterium tumefaciens gene transfer to Caduarina glauca,a tropical nitrogen-fixing tree. Plant Sci 118:57–69
Lee YW, Jin S, Sim WS, Nester EW (1996) The sensing of plant signal molecules by Agrobacterium: genetic evidence for direct recognition of phenolic inducers by the VirA Protein. Gene 179:83–88
Levée V, Lelu M-A, Jouanin L, Cornu D, Pilate G (1997) Agrobacterium tumefaciens-mediated transformation of hybrid larch (Larix kaempferi×L. Decidua) and transgenic plant regeneration. Plant Cell Rep 16:680–685
Machado LDR, Deandrade GM, Cid LPB, Penchel RM, Brasileiro ACM (1997) Agrobacterium strain specificity and shooty tumour formation in Eucalypt (Eucalyptus grandis×E. urophylla). Plant Cell Rep 16:299–303
Mohri T, Mukai Y, Shinohara K (1997) Agrobacterium tumefaciens-mediated transformation of Japanese white birch (Betula platyphylla var. Japonica). Plant Sci 127:53–60
Montoro P, Etienne H, Carron M-P, Nougarède A (1992) Effect of cytokinins on the induction of embryogenesis and the quality of somatic embryos in Hevea brasiliensis Müll. Arg. C R Acad Sci 315:567–574
Montoro P, H. E, Michaux-Ferriere N, Carron MP (1993) Callus friability and somatic embryogenesis in Hevea brasiliensis. Plant Cell Tissue Organ Cult 33:331–338
Montoro P, Pujade-Renaud V, Teinseree N (1997) Strategy to study functionality of putative promotor from Hevea brasiliensis: Attempts of Agrobacterium tumefaciens-mediated gene transfer in various tissues. In: Proceedings of the French-Thai Seminar on the Biochemical and Molecular Tools for Exploitation of Diagnostic and Rubber Tree Improvement, Bangkok, Thailand, 20 October 1997–22 October 1997
Montoro P, Teinseree N, Rattana W, Kongsawadworakul P, Michaux-Ferriere N (2000) Effect of exogenous calcium on Agrobacterium tumefaciens-mediated gene transfer in Hevea brasiliensis (rubber tree) friable calli. Plant Cell Rep 19:851–855
Rattana W, Teinseree N, Tadakittisarn S, Pujade-Renaud V, Monkolsook Y, Montoro P (2001) Characterization of factors involved in tissue growth recovery and stability of GUS activity in rubber tree (Hevea brasiliensis) friable calli transformed by Agrobacterium tumefaciens. Thai J Agric Sci 34(3–4)
Rossignol M, Lamant D, Salsac L, Heller R (1977) Calcium fixation by the roots of calcicole and calcifuge plants: the importance of membrane systems and their lipid composition. In: Thellier M, Monnier A, Demarty M, Dainty J (eds) Transmembrane ionic exchange in plants. CNRS, Paris, p 483
Sainte-Beuve J (2001) Filière hévéa: conjoncture et bilan de la recherche. Plant Rech Dév 1:59–60
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Sanders D, Brownlee C, Harper JF (1999) Communicating with calcium. Plant Cell 11:691–706
Sangwan RS, Bourgeois Y, Brown S, Vasseur G, Sangwan-Norreel B (1992) Characterization of competent cells and early events of Agrobacterium-mediated genetic transformation in Arabidopsis thaliana. Planta 188:439–456
Siler DJ, Cornish K (1995) Measurement of protein in natural rubber latex. Anal Biochem 229:278–281
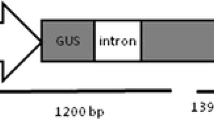
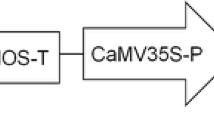
Vancanneyt G, Schimdt R, O'Connor-Sanchez A, Wilmitzer L, Rocha-Sosa M (1990) Construction of an intron-containing marker gene: splicing of the intron in transgenic plants and its use in monitoring early events in Agrobacterium-mediated plant transformation. Mol Gen Genet 220:245–250
Verdeil J-L, Hocher V, Huet C, Grosdemange F, Escoute J, Ferrière N, Nicole M (2001) Ultrastructural changes in coconut calli associated with the acquisition of embryogenic competence. Ann Bot 88:9-18
Yeang H-Y, Arokiaraj P, Jaafar H, Hamzah S, Arif SMA, Jones H (1998) Rubber latex as an expression system for high-value proteins. In: Shewry PR, Napier JA, Davis PJ (eds) Engineering crop plants for industrial end uses: proceedings of the symposium of the Industrial and Biotechnology Group of the Biochemical Society. Portland, London, pp 55–64
Acknowledgements
This work was supported by Franco-Thai co-operation between CIRAD, DTEC, Department of Agriculture/Rubber Research Institute of Thailand, Kasetsart University and the French Embassy in Bangkok. We would like to thank Associate Professor Vichai Haruthaithanasan, Director of KAPI-KU, and Associate Professor Klanarong Sriroth, Head of CSTR unit, for hosting the Rubber Tree Genetic Engineering Research Programme at KAPI. We also thank Dr. Marc-Philippe Carron and Dr. Ludovic Lardet (CIRAD, France) for providing friable callus lines, Ms. Kanikar Therawattanasuk (RRIT, Thailand) for providing rubber tree fruits, Dr. Richard Jefferson (CAMBIA, Australia) for providing the pCAMBIA2301 vector and the EHA105 Agrobacterium tumefaciens strain. Finally, the authors are grateful to Mrs. Dolores Triaire for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Debergh
Rights and permissions
About this article
Cite this article
Montoro, P., Rattana, W., Pujade-Renaud, V. et al. Production of Hevea brasiliensis transgenic embryogenic callus lines by Agrobacterium tumefaciens: roles of calcium. Plant Cell Rep 21, 1095–1102 (2003). https://doi.org/10.1007/s00299-003-0632-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-003-0632-7