Abstract
Patients with rheumatoid arthritis have higher rates of mental health conditions compared to the general population. It is believed that affective distress and rheumatoid arthritis have a bi-directional relationship. This review will examine the associations between affective distress and rheumatoid arthritis outcomes over time. Several disease outcomes are included covering disease activity, function, and disability to provide a broad picture of the various ways patients are impacted. A quality assessment was also conducted. There were 71 studies included in the review. Three measures (disease activity, disability, and mortality) had enough data to complete meta-analyses of odds ratios or hazard ratios. The outcomes included were disease activity, tender joint count, swollen joints, pain, physician global assessment, patient global assessment, physical disability, acute phase reactants, stiffness, fatigue, work disability, and mortality. Numerous measures were included for most of the outcomes due to the variability across studies of measures used. Patients with affective distress had lower rates of remission according to the DAS-28, greater disability, and higher mortality. All of the outcomes covered had studies with mixed results, but swollen joint count, tender joint count, patient global assessment, and physician global assessment had the strongest evidence that they were associated with mental health longitudinally. The relationships between affective distress and disease outcomes are complex and vary depending on the measures. Overall, the effects fade over time. It is important for clinicians to be aware of the differing manifestations of the relationship between affective distress and rheumatoid arthritis outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a chronic progressive autoimmune inflammatory disease that primarily affects the joints. It is approximately twice as common in females compared to males [1], with a combined prevalence rate of ~ 1% in the adult population [2]. The primary feature of RA is joint inflammation and swelling (synovitis), which typically causes pain and reduced function, which can greatly impact on the patient’s quality of life. Where inflammation is not adequately controlled, persistent inflammation can lead to irreversible joint damage, which can further impact on function and quality of life [3, 4].
Patients with RA have higher rates of affective distress, namely depression and anxiety, than the normal population [5, 6]. Estimates indicate that between one-fifth to one-third of RA patients have comorbid depressive symptoms, indicative of depressive disorder [7,8,9]. Furthermore, patients with affective distress and RA have worse disease outcomes in both the short-term and long-term, even after accounting for disease severity [10, 11].
Affective distress and RA may have a bi-directional relationship [9, 12]. RA is believed to impact depression symptoms through various mechanisms such as arthralgia and disability associated with RA resulting in depression [13] while depression may affect RA symptoms through illness-related cognitions [14, 15]. Recent findings also suggest that depression may at least in part be an extra-articular manifestation of RA-related inflammation [9, 16,17,18]. Mechanisms for affective distress affecting RA outcomes include altered pain perception [19] and shared inflammatory processes [20]. While there has been less research on the biological mechanisms linking anxiety and rheumatoid arthritis, anxiety has also been found to be higher in RA patients [21]. Given the overlap between RA and affective distress causes and processes, they may also negatively affect one another in a downward spiral, leading to both worse affective distress and RA outcomes.
Since the relationship between affective distress and RA seems to be complex and inter-dependent, longitudinal studies are needed to disentangle the temporality and causality of the effects of affective distress on disease outcomes. However, most of the existing literature has been cross-sectional, focusing on associations between the two factors [22, 23]. Furthermore, drawing conclusions across studies has been difficult because numerous different outcome measures of RA have been used, ranging from biological markers to subjective ratings. Similarly, differing measures of depression or anxiety have been used, focusing on different aspects or symptoms.
The aim of this review is to synthesize the extant literature on longitudinal studies of affective distress and clinical outcomes in adults with rheumatoid arthritis, in order to determine if affective distress is associated with worse clinical outcomes over time.
Methods
Identifying publications
The search strategy followed the PICO framework using key terms to conduct searches of the following computerized databases: MEDLINE, PSYCinfo, PSYCarticles, Embase, Cochrane Library, Web of Science, and SCOPUS. Grey literature was identified by searching System for Information on Grey Literature, OpenGrey, and EthOS. Finally, a manual search of reference lists from included studies was conducted. The search strategy included unpublished and published articles or conference abstracts. The timeline included articles from 1998 to 2023.
Keywords were used according to each database’s formats and included the following words or phrases: “depression/depressive symptoms,” “anxiety,” “mood,” “psychological/emotional distress,” “affect/affective disturbance, “rheumatoid arthritis,” “longitudinal,” “prospective,” “cohort,” “retrospective”. An example of the combination of a keywords combination used in the search strategy for outcomes is: “disease activity” OR “clinical outcomes” OR DAS28 OR “DAS 28” OR DAS-28 OR “disease activity score” OR HAQ OR “health assessment questionnaire” OR VAS OR “visual analog scale” OR “pain” OR SJC OR “swollen joint count” OR TJC OR “tender joint count” OR CRP OR “C-reactive protein” OR stiffness OR CDAI OR “clinical disease activity index” OR SDAI OR “simple disease activity index” OR “american college of rheumatology” OR ACR. With Wildcards, the search example becomes: “disease activity” OR “clinical outcomes” OR DAS28 OR “DAS?28” OR “disease activity score” OR HAQ OR “health assessment questionnaire” OR VAS OR “visual analog scale” OR “pain” OR SJC OR “swollen joint count” OR TJC OR “tender joint count” OR CRP OR C?-RP OR “C-reactive protein” OR stiffness OR CDAI OR “clinical disease activity index” OR SDAI OR “simple disease activity index” OR “american college of rheumatology” OR ACR.
Inclusion/exclusion criteria
Studies which met the following criteria were included: (1) Participants were adults 18 years old and over with rheumatoid arthritis and (2) the studies were longitudinal observational studies, including prospective, cohort, and retrospective studies or randomized controlled trials treated as observational studies where depression, depressive symptoms, anxiety, or psychological or affective distress is considered as a prognostic marker, or predictive marker of treatment efficacy. Studies in any language were included where it was possible to translate the paper. There were no date restrictions. Exclusion criteria were: (1) studies with participants under age 18 (2) studies that were not longitudinal or not including the mental health conditions of the inclusion criteria.
Outcomes
The primary outcome of interest was the DAS 28; however, secondary outcomes will include the core criteria outlined by OMERACT (Outcome Measure in Rheumatology): Tender Joint Count (28 count TJC), swollen joints (28 count SJC), pain [Visual Analog Scale (VAS), Arthritis Impact Measurement Scales (AIMS)], physician global assessment [Evaluator Global Assessment (EGA)], patient global assessment [Patient Global Assessment (PGA), VAS], physical disability [Health Assessment Questionnaire (HAQ)], acute phase reactants [C-reactive protein (CRP), Erythrocyte Sedimentation Rate (ESR)] [24]. Radiographs were not included since that measurement is used only for studies of at least one year. OMERACT outcomes that were not part of the core measures, but were included were: Stiffness (Duration prioritized) and fatigue (VAS).
Additional outcomes outside of OMERACT that included were: Disease Activity (Simple Disease Activity Index (SDAI), Clinical Disease Activity Index (CDAI), Work disability/impairment, and Mortality.
Publication screening
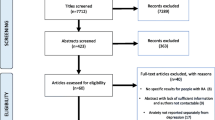
One reviewer (MS) screened the titles/abstracts of all of the studies identified in the search according to the inclusion/exclusion criteria. A second reviewer (MA) independently screened 10% of the full texts that were included. Agreement at the full text stage was achieved in 100% of the papers. A flow chart of the screening process is found in Supplementary Material Fig. S1.
Data extraction
One reviewer (MS) extracted data from the included studies using a pre-designed form for data extraction. Information was extracted about (1) Sample characteristics (e.g. mean age, proportion female) (2) Sample size at each time point, and flowchart of participation (i.e. attrition) (3) Eligibility (4) Study rationale and aims (5) Depression/mental health measures (6) Study design (7) Outcome measures categorised by type (e.g. disease activity, symptom) (8) Types of analysis, including covariates adjusted for (9) Dates of follow up 10) Effect estimate and its precision (i.e. standard error), or sufficient data to calculate precision (e.g. standard deviation) (11) Publication date (12) country of publication and (13) cohort. In cases where the necessary data was not given in a publication, the authors were contacted.
Quality assessment
Risk of bias was assessed using the Quality In Prognosis Studies tool. Grading of Recommendations, Assessment, Development and Evaluation (GRADE) will be used to evaluate the strength of the body of evidence.
Statistical analysis
Meta-analysis of each outcome measure with sufficient data and graphs was conducted in STATA 16.0. Due to there being many different outcomes and methodological heterogeneity, meta-analysis was only done for the outcomes measured dichotomously where possible, then a narrative synthesis was done using an effect size based vote counting approach across all studies. Random-effects models were used to estimate the weighted effect size for each outcome measure. For mortality, the weighted hazard ratio was also calculated. The I2 statistic was also calculated to determine the level of heterogeneity among the effect sizes. Lastly, forest plots were generated to display the weighted effect size.
Results
In total, there were 71 studies included in this review (Supplementary Table 1). The most common outcome measure was the DAS28, with 31 studies reporting results for it (Supplementary Table 2). Disability measures, primarily the HAQ, were also frequently used. The remaining outcomes had fewer studies, but many were still prevalent in the literature. The studies also represented a wide number of countries, though studies from the UK and USA were most common in the sample. A majority of the studies also had large sample sizes, which would have made them better able to detect effects. A quality assessment using the Newcastle-Ottowa Scale is presented in Supplementary Table 3.
Mortality
Meta-analysis of four studies on mortality showed a pooled effect estimate of 0.64 (95% CI: 0.19, 10.8). The pooled hazard ratio estimate was 2.98 (95% CI 1.49–5.97) (see Fig. 1). These indicate that patients with RA who have comorbid depression have higher risk of mortality than non-depressed patients. However, since the I2 statistic indicates high heterogeneity (I2 = 90.3%), these results should be interpreted with caution.
Disease activity
There were 34 studies investigating the relationship between affective distress and disease outcomes over time, including the DAS, CDAI, and SDAI. Due to heterogeneity in the methods used and range of outcomes it was only possible to undertake meta-analysis for DAS remission.
Meta-analysis on the odds ratio for remission using the DAS showed a pooled effect estimate of 0.57 (95% CI 0.51–0.83). The meta-analysis generated a pooled odds ratio of 1.77 (95% CI 1.36–2.29) (Fig. 2). These results indicate that patients with affective distress have a lower chance of reaching remission compared with those without affective distress. These results should be interpreted with caution as the predictors of the odds ratio were mixed between depression and anxiety and the heterogeneity for the analysis is high (I2 = 68.6%).
Across all 34 studies, just over half found a positive effect where affective distress was associated with disease activity, though there were several nuances and nearly half of the studies had an unclear effect. The amount of time for the follow-up ranged from 13 to 104 weeks so it is perhaps not surprising that the findings would be conflicting over such a large range. However, the general trend indicates that the effect fades over time. One study with more frequent follow ups detailed this trend, with the effect only being significant at 13 weeks while the remaining follow ups for just over a year no longer showed an effect [25]. Another factor which influenced the results was the type of affective distress. Distinguishing between depression and anxiety brought mixed results for some studies, though no definitive overall trend was discernable. Similarly, the scales used to measure affective distress seemed to affect the results as well, with some studies using scales such as the SF-36 or EQ5D, which were not originally designed specifically for affective distress. The results of studies with these scales were mixed but mostly indicated that scales which are more specific to mental health were linked with later effects on disease outcomes whereas more general scales were not [11, 26, 27]. Overall, the findings indicate that affective distress indeed affects disease outcomes over time, though the amount of time this effect lasts is undetermined but likely fades over time.
Somatic symptoms
CRP
Three studies examined the effects of affective distress on CRP longitudinally. None of the studies found clear significant associations, even though one of them followed participants for 1 year. The inconclusive results could be due to smaller sample sizes in the studies that did not find significant results, whereas the CORRONA study had 12,445 participants so it had more power to detect differences between groups. Furthermore, the differences in log CRP were small so it is likely that a study would need high power in order to detect significant differences. Distinguishing between depression versus anxiety did not change these inconclusive results since the study which distinguished between them had insignificant findings for both conditions.Thus, while affective distress did not appear to be clearly associated with elevated CRP, it's possible the differences were too small to be detected.
ESR
Of the 8 studies which measured ESR, the relationship with affective distress was unclear for all of them. However, one study which used various measures of affective distress included history of depression rather than only current psychological distress and found that significantly predicted ESR [26]. Another study found an association between psychological distress and ESR at 6 months [28]. These were both the shortest-term studies included. This points towards the possibility that the effects of psychological distress on ESR exist but wane over time. However, the number of studies with similar timepoints for the short vs long-term is small so it is difficult to conclude with much certainty that the effect on ESR diminishes with time. When depression and anxiety were considered separately, the results remained unclear since the findings were insignificant for both conditions. Greater consistency in the measurement of affective distress, its subcomponents, and the time periods measured in future studies could clarify the relationship.
TJC
There were 7 studies found that investigated affective distress and TJC. The scales used and the results were mixed, but 5 of the 7 studies found a significant relationship between affective distress and TJC. The type and severity of affective distress of the studies seemed to define the findings. For example, one study separated affective distress by severity and found those with more severe depression had worse TJC scores [29]. For those studies which measured depression versus anxiety, rather than affective distress more broadly, many more focused on depression. There was only one study which examined anxiety alone and its results were consistent with the depression results [29]. While there is nuance in the severity and type of affective distress in its effect on TJC, the studies overall indicate that affective distress indeed worsens TJC.
SJC
There were 7 studies which investigated the relationship of affective distress on SJC. Similar to TJC, 5 of the 7 studies indicated an effect of affective distress on SJC. This included studies which reported mean scores for depressed vs not depressed patients, but did not report their statistical significance. However, the differences were small (mostly 0.1 or fewer points difference) between the groups so the results also seem to point towards affective distress having less of an effect on SJC compared with other outcomes included in this review. In the three studies that distinguished between anxiety and depression, the direction of the effect was different in one, but it was very small and insignificant [25]. The remainder of studies that distinguished between anxiety and depression did not have any significant differences [30, 31]. The other studies either used only depression or scales such as the SF-36, which measures affective distress more broadly. In terms of timelines, studies lasted a year or more so it is also possible that the effect is short and fades by 1 year so it was not detected by these studies.
EGA
Four studies examined the effect of affective distress on EGA. Most, 3 out of 4, found a significant association between poor mental health and higher EGA scores. Two studies distinguished between severity levels of depression and found greater depression was associated with worse EGA scores [26, 32]. Many of the studies reported only the means without testing for statistical significance so conclusions based on these are limited. However, overall the studies seem to indicate that affective distress is associated with worse EGA scores, with increasing affective distress associated with increasing EGA. The studies either focused on depression alone or affective distress rather than distinguishing between anxiety versus depression, but there was no discernable difference in the results based on these measures. The studies lasted 1–2 years so it appears that the effects of affective distress on EGA may also be longer lasting, compared with other outcomes included in this review.
PGA
There were 2 out of 3 studies which indicated a link between affective distress and PGA. The remaining study reported means over time so the direction of effect was unclear. However, the results across studies showed that the greater the depression scores, the greater the PGA scores. While the variation in statistical methods used between studies makes it difficult to draw any conclusions beyond the single study that tested for an association, they did show increased PGA when affective distress was increased. There was a mix of studies that distinguished between anxiety or depression and those that combined them into affective distress, but the results were not clarified along these lines. Similar to EGA, the studies also lasted 1–2 years, during which time any associations could fade anyway.
Pain
There were 12 studies investigating pain and affective distress. Just under half of the studies, or 5 of the 12, found a relationship between these two factors found a significant relationship. This may be partly due to the outcomes measures being mixed between the VAS, AIMS, and MPQ, though most reported the VAS. Overall, while pain and affective distress were found to be significantly related in nearly half the studies, the results may be mixed due to the relationship being more complex than could be detected in the existing studies, given that pain is one of the most subjective measures included in this systematic review. For example, previous studies have shown the threshold of pain to be lower in patients with depression so the relationship between pain and affective distress may be interdependent so studies aimed at teasing apart these concepts could better explain the mixed findings [33]. Most studies focused on affective distress, but some studied depression alone, while only one studied anxiety alone. There was a significant relationship between anxiety and both pain outcome measures, which aligns overall with the studies that used affective distress or depression [34].
Stiffness
There was only one study that examined psychological distress and stiffness and revealed a complex relationship [35]. Patients were separated into trajectories of affective distress, but all of them showed decreases in early morning stiffness by the end of 3 years. However, the amount of decrease varied based on the level of distress. Those who ended with higher distress had similar stiffness scores by the end of 3 years, regardless of their starting point. Although this does not support the conclusion that earlier psychological distress has long-lasting effects on stiffness, it does strengthen the conclusion that in the immediate or short-term, affective distress is closely related with stiffness.
Fatigue
The results for the effect of affective distress on fatigue were mixed in that half of the studies indicate that depression is significantly associated with fatigue while the remaining studies did not find any significant relationship. This was not entirely clarified by distinguishing between depression, anxiety, or affective distress since there was only one study focused on affective distress while the rest studied depression, but the results of the study for affective distress were insignificant. However, it is possible this is an important distinction for fatigue since it could be more linked with depression than anxiety or affective distress more generally. The studies which were significant looked at fatigue scores within a few months of the depression scores whereas those that were not significant were examining scores a year or more later. These results could indicate that the effect of affective distress on fatigue is significant, but only in the short-term.
Disability
There were 23 studies which examined affective distress and its association with disability long-term. While the majority (13) of the studies did not have conclusive results, several studies did find a significant association between affective distress and later disability, with only two studies finding no relationship. The outcome measures used for disability were the most diverse of all the outcomes included in this review, covering HAQ, AIMS, ADL, GARS, MHAQ, SF-36, and Short-term disability days, so the variety may explain some of the mixed results. The affective distress measures were also diverse but did not yield different results for those which focused on anxiety versus depression versus affective distress. Furthermore, the follow-ups were over the longest time periods, compared with other outcomes, with only two studies including follow-ups at less than a year. Thus, the effects could have been missed or less pronounced due to the longer time between baseline and the follow-ups. While it seems likely that affective distress is related to disability over time, the conclusion is tentative due to the inconsistency of the measures and long time periods.
Meta-analysis of depression on the odds ratio of disability, using the HAQ showed a pooled effect estimate of 1.99 (95% CI 0.54–3.45). The pooled odds ratio was 7.37 (95% CI 1.72–31.64), indicating that patients with depression have a greater risk of disability. The heterogeneity was high (I2 = 77.1%) so these results should also be interpreted with caution (see Fig. 3).
Work disability
There were only 2 studies which examined the effect of emotional distress or depression on presenteeism with conflicting results with one finding a negative effect and the other inconclusive. The study with an effect in the negative direction used anxiety, which may indicate patients try harder to keep up their work to prevent further anxiety so the type of distress could determine the direction. A third study also examined the effect of affective distress on productivity, which could be considered closely related and similarly found an effect in the negative direction, but the measure was psychological distress more generally. There were also a couple additional studies that examined work absence, including absenteeism, sick leave, and leaving the job. One study showed slightly elevated chance of sick leave for those who were depressed while the other study found a higher 3.2 risk ratio for sick leave for anxious patients [36], indicating that the type of emotional distress may also affect outcomes for work disability, similar to other outcomes in this review. The mixed results could also be due to differing scales, different severities of affective distress, or the small number of studies. Distinguishing between anxiety, depression, or affective distress did not change these results since the findings were mixed among these aspects of mental health. Lastly some aspects of work disability, such as presenteeism, could be subjective so patients who are depressed or anxious may have less accuracy in reporting their productivity, which could influence the results.
Discussion
The meta-analyses and narrative review present an overall picture that affective distress impacts RA patients not just in their day-to-day symptoms, but over their lives through work disability and earlier mortality. While the studies were quite mixed in the reported outcomes and measures of affective distress, they seem to indicate that affective distress has an impact, but the specific outcomes which are impacted vary such that not all outcomes are equally affected. These varied results may be helpful for clinicians to understand the different presentations that they may see in clinics.
The differences between the results of the studies also highlighted the subjectivity of many of the measures. For example, TJC was significantly related with affective distress in most studies, whereas SJC, which could be considered more objective, was only found to be linked in two studies. In contrast, the most objective measures, such as CRP and ESR for inflammation, only showed weak evidence for a relationship with affective distress. This pattern is evident overall as well, with more subjective measures showing more associations while more objective measures showing fewer associations. The implications of this highlight the inconsistencies across clinics and measures and potential improvements possible in research by standardization.
However, it may be a more complex situation since the inflammatory markers had opposing results depending on the follow-up timing. It could be that they are affected by affective distress, but only in the short-term. Mood also varies over time, even within the day, so it may be more complex to collect accurate measurements of mood and disease outcomes at the right timepoints [37]. Alternatively, depressive mood may cause patients to report worse symptoms as it could influence their perception of the symptoms. However, it is also possible that the objective ratings are missing something since they may be done by an outside observer or provide only one piece of information.
Additionally, the present study not only used many different outcomes, but also many varied affective distress measures, ranging from history of depression, to anxiety only, to mixed affective distress, to severe depression only. There were multiple scales used. Despite these differing methods, there were sometimes differing results for depression versus anxiety where they could be distinguished, but there was not enough evidence in this review to adequately distinguish between their effects. However, evidence so far shows that they may share some overlapping biological pathways, but not all [38]. Adopting more specific and universal measures could make it easier to draw conclusions due to more consistency or specificity. There are also behavioral and cognitive differences that could distinguish their effects on physical outcomes. Future studies should be designed with awareness of the possible different effects so that they can be accurately captured.
This study had the benefit of a large size of many studies covering many different outcomes. However, there were also limitations, such as the large variance in outcome measures used which prohibited meta-analysis for most measures. The follow-up times also made it difficult to accurately assess the true effects of affective distress since these effects could have been missed due to the follow-ups occurring too soon or too late. Similarly, while the time range of the studies included was large, attitudes towards mental health and treatments for RA could have shifted during that time.
Although there are many differences in the details of the studies, taken as a whole, they appear to confirm that affective distress affects physical symptoms over time. This has the implication of suggesting greater attention is needed to the mental health of RA patients in clinical care. Greater consistency across studies in the future will help further clarify these relationships. These results can be useful for patients and clinicians to better understand the complex and long-lasting relationship between affective distress and physical RA symptoms.
Author | Year | Cohort | Country | Study Design | Sample Size | Weeks Follow up | Mean age | % Female | Measure |
|---|---|---|---|---|---|---|---|---|---|
Zhao [39] | 2015 | UK | Observational | 126 | 52 | Not reported | Not reported | DAS28, HAQ, | |
Matcham [40] | 2015 | UK | Observational | 385 | 52 | 59 | 81 | DAS28, HAQ, ESR, TJC, SJC, PGA | |
van den Hoek [41] | 2016 | Netherlands | Observational | 882 | 156 | 59 | 72 | HAQ, EGA, Mortality | |
Miwa [42] | 2015 | Japan | Intervention | 333 | 26 | Not reported | Not reported | SDAI | |
Brown [43] | 1990 | USA | Observational | 387 | 182 | 53 | 75 | VAS pain | |
Kronisch [44] | 2015 | SERA | UK | Observational | 578 | 52 | 61 | 65 | HAQ |
Corominas [45] | 2014 | Spain | Observational | 120 | 104 | 52 | 87 | DAS28 | |
Gwinnutt [46] | 2019 | RAMS | UK | Intervention | 463 | 52 | Not reported | 68 | Work disability (work leave, sick leave, presenteeism) |
Rathbun [47] | 2016 | CORRONA | USA | Intervention | 1820 | 52 | 58 | 75 | CDAI |
Matcham [31] | 2016 | UK | Observational | 56 | 52 | 54 | 79 | DAS28, ESR, TJC, SJC, PGA | |
Kronisch [48] | 2016 | SERA | UK | Observational | 1140 | 52 | 61 | 65 | HAQ |
Miwa [49] | 2017 | Japan | Retrospective intervention | 232 | 26 | 55 | 71 | HAQ | |
Ang [50] | 2005 | USA | Observational | 1290 | 939 | 57 | 73 | Mortality | |
Parenti [51] | 2016 | USA | Retrospective intervention | 4064 | 26 | Not reported | Not reported | DAS 28, CDAI | |
Cui [52] | 2015 | OBRI | Canada | Observational | 2305 | 26 | Not reported | Not reported | DAS28, SDAI |
Corominas [53] | 2019 | Spain | Intervention | 2305 | 26 | Not reported | Not reported | FACIT-F | |
Michelsen [54] | 2017 | NOR-DMARD | Norway | Intervention | 1326 | 26 | 54 | 75 | DAS28, CDAI, SDAI |
Kleinert [55] | 2016 | Germany | Observational | 764 | 261 | 54 | 80 | Mortality | |
Hider [56] | 2009 | UK | Intervention | 160 | 52 | 56 | 72 | DAS28 | |
Bode [57] | 2012 | USA | Observational | 530 | 255 | 60 | 84 | Mortality | |
Leblanc-Trudeau [58] | 2015 | EUPA | Canada | Intervention | 275 | 182 | 61 | 63 | SDAI |
McFarlane [59] | 1988 | Australia | Observational | 30 | 156 | 53 | 66 | DAS28 | |
Norton [60] | 2011 | ERAS | UK | Observational | 784 | 156 | 57 | 67 | DAS28, HAQ |
Michelsen [61] | 2017 | NOR-DMARD | Norway | Intervention | 1326 | 26 | 54 | 75 | DAS28, CDAI, SDAI |
Kuijper [25] | 2018 | Netherlands | Intervention | 281 | 65 | 53 | 68 | DAS28, ESR, SJC | |
Feldthusen [62] | 2016 | Sweden | Observational | 65 | 52 | 54 | 74 | VAS fatigue | |
Gonzalez-Lopez [36] | 2013 | Mexico | Observational | 123 | 52 | 44 | 73 | Sick leave | |
Tanaka [63] | 2019 | Japan | Intervention | 377 | 104 | Not reported | Not reported | WPAI | |
Doeglas [64] | 2004 | Netherlands | Observational | 264 | 156 | 53 | 65 | GARS | |
Verstappen [65] | 2007 | Netherlands | Intervention | 112 | 52 | 49 | 68 | HAQ | |
Hommel [66] | 1998 | USA | Observational | 42 | 52 | 53 | 81 | MHAQ | |
Fifield [67] | 2001 | USA | Observational | 415 | 417 | 58 | 83 | VAS fatigue | |
Looper [68] | 2011 | McEAR | Canada | Retrospective observational with history of depression | 104 | Medical History | 54 | 61 | HAQ |
Nugaliyadde [69] | 2017 | UK | Intervention | 13 | 51 | 61 | 73 | DAS28 | |
Chung [70] | 2013 | Australia | Observational | 114 | 261 | Not reported | Not reported | DAS28, HAQ | |
England [71] | 2015 | BRAGGS | UK | Intervention | 1847 | 26 | 58 | 76 | DAS28 |
Sergeant [72] | 2015 | RAMS | UK | Intervention | 460 | 26 | 60 | 73 | DAS28 |
Morris [73] | 2011 | UCSF RA panel | USA | Observational | 1115 | 939 | 55 | 80 | HAQ |
Van Den Hoek [74] | 2013 | Netherlands | Observational | 882 | 574 | 59 | 72 | HAQ, SF-36 | |
Bechman [11] | 2018 | OPTTIRA | UK | Intervention | 97 | 52 | 57 | 74 | DAS28 |
McFarlane [75] | 1987 | Australia | Observational | 40 | 156 | 53 | 80 | DAS-28 | |
Odegard [34] | 2007 | EURIDISS | Norway | Observational | 238 | 521 | 52 | 74 | VAS pain |
Vriezekolk [76] | 2010 | Netherlands | Intervention | 73 | 6 | 53 | 72 | AIMS, VAS pain | |
Sergeant [77] | 2016 | RAMS | UK | Intervention | 1050 | 26 | 59 | 70 | DAS28 |
Sergeant [78] | 2018 | RAMS | UK | Intervention | 1656 | 26 | 59 | 67 | DAS28 |
Casalla [79] | 2013 | CONAART | Argentina | Observational | 237 | 52 | 49 | 84 | DAS28 |
Cook [80] | 2016 | NOAR | UK | Observational | 868 | 261 | 56 | 66 | SJC |
Treharne [81] | 2008 | UK | Observational | 189 | 52 | 56 | 74 | VAS fatigue | |
Dobkin [82] | 2013 | Canada | Observational | 248 | 52 | 59 | 63 | MPQ-SF | |
Leggett [83] | 2017 | RAMS | UK | Intervention | 308 | 52 | 52 | 66 | WPS-RA |
Dyball [30] | 2018 | BRAGGSS | UK | Intervention | 2919 | 26 | 57 | 76 | DAS28, CRP, TJC, SJC |
Schieir [84] | 2016 | ERA | Canada | Intervention | 1595 | 52 | 54 | 72 | DAS28 |
Parker [85] | 1992 | USA | Observational | 80 | 26 | 61 | 0 | SJC | |
Norton [60] | 2011 | ERAS | UK | Observational | 784 | 261 | 57 | 67 | HADS, ESR, VAS pain, TJC, SJC, Stiffness |
Matcham [40] | 2015 | CARDERA | UK | Intervention | 467 | 104 | 54 | 68 | HAQ, ESR, SJC |
Iannaccone [86] | 2016 | USA | Observational | 264 | 104 | 57 | 83 | DAS28, MHAQ, CRP | |
Schieir [87] | 2009 | McEAR | Canada | Observational | 320 | 26 | 57 | 69 | MPQ, SJC |
Rathbun [88] | 2013 | CORRONA | USA | Observational | 4250 | 104 | Not reported | Not reported | CDAI, HAQ, CRP, ESR, VAS pain, TJC, SJC, EGA, PGA |
Rathbun [89] | 2015 | CORRONA | USA | Observational | 12,445 | 104 | 58 | 73 | DAS28, CDAI, HAQ, CRP, ESR, TJC, SJC, EGA, PGA |
El Miedany [90] | 2013 | Egypt or UK | Observational | 264 | 156 | 68 | DAS28, TJC, | ||
Uhlig [91] | 2000 | EURIDISS | Norway | Observational | 238 | 261 | 51 | 74 | AIMS |
Li [92] | 2019 | Truven Health Marketscan databse | USA | Observational | 46,700 | 52 | 52 | 78 | Short-term disability |
Matcham [32] | 2014 | CARDERA | UK | Intervention | 467 | 104 | 54 | 68 | DAS28, HAQ, ESR, VAS pain, TJC, SJC, EGA |
Van Den Hoek [93] | 2013 | Netherlands | Observational | 882 | 574 | 59 | Not reported | HAQ, SF-36 | |
Matcham [94] | 2014 | CARDERA | UK | Intervention | 379 | 104 | 54 | 68 | SJC |
Crotty [95] | 1994 | Australia | Observational | 75 | 191 | 42 | 100 | HAQ | |
Overman [28] | 2011 | Netherlands | Intervention | 545 | 26 | 56 | 69 | TJC | |
Matcham [26] | 2018 | BSRBR-RA | UK | Intervention | 18,421 | 52 | 56 | 76 | DAS28, ESR, SJC, EGA |
Euesden [27] | 2017 | CARDERA | UK | Intervention | 520 | 104 | 55 | 69 | DAS28, HAQ, ESR, VAS pain, TJC, SJC, EGA |
Smedstad [96] | 1997 | EURIDISS | Norway | Observational | 238 | 104 | 52 | 73 | HAQ, VAS pain |
Karpouzas [97] | 2017 | UCLA RA cohort | USA | Observational | 156 | 52 | 52 | 89 | HAQ |
Data availability
Data is available from authors upon request.
References
Symmons D, Turner G, Webb R, Asten E, Barrett M, Lunt M et al (2002) The prevalence of rheumatoid arthritis in the United Kingdom: new estimates for a new century. Rheumatology 41:793–800
Alamanos Y, Drosos AA (2005) Epidemiology of adult rheumatoid arthritis. Autoimmun Rev 4:130–136. https://doi.org/10.1016/j.autrev.2004.09.002
McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365:2205–2219. https://doi.org/10.1056/nejmra1004965
Scott DL, Smith C, Kingsley G (2003) Joint damage and disability in rheumatoid arthritis: an updated systematic review. Clin Exp Rheumatol 21:S20–S27
Dickens C, Mcgowan L, Clark-Carter D, Creed F (2002) Depression in rheumatoid arthritis: a systematic review of the literature with meta-analysis
Matcham F, Ali S, Irving K, Hotopf M, Chalder T (2016) Are depression and anxiety associated with disease activity in rheumatoid arthritis? A prospective study. BMC Musculoskelet Disord 17:155. https://doi.org/10.1186/s12891-016-1011-1
Bruce TO (2008) Comorbid depression in rheumatoid arthritis: Pathophysiology and clinical implications. Curr Psychiatry Rep 10:258–264. https://doi.org/10.1007/s11920-008-0042-1
Katz P, Yelin E (1993) Prevalence and correlates of depressive symptoms among persons with rheumatoid arthritis, pp 790–796
Sturgeon JA, Finan PH, Zautra AJ (2016) Affective disturbance in rheumatoid arthritis: psychological and disease-related pathways. Nat Rev Rheumatol 12:532–542. https://doi.org/10.1038/nrrheum.2016.112
Bechman K, Sin FE, Ibrahim F, Norton S, Scott D, Cope A et al (2018) Psychological and functional states predict disease flare following TNF inhibitor tapering in patients with rheumatoid arthritis: a post-hoc analysis of data from the optimisingTNF tapering in RA (OPTTIRA) cohort. Ann Rheum Dis 77:279–280. https://doi.org/10.1136/annrheumdis-2018-eular.3077
Bechman K, Sin FE, Ibrahim F, Norton S, Matcham F, Scott DL et al (2018) Mental health, fatigue and function are associated with increased risk of disease flare following TNF inhibitor tapering in patients with rheumatoid arthritis: an exploratory analysis of data from the Optimizing TNF Tapering in RA (OPTTIRA) trial. RMD Open. https://doi.org/10.1136/rmdopen-2018-000676
Dickens C, Creed F (2001) The burden of depression in patients with rheumatoid arthritis. Rheumatology 40:1327–1330
Margaretten M, Julian L, Katz P, Yelin E (2011) Depression in patients with rheumatoid arthritis: description, causes and mechanisms. Int J Clin Rheumtol 6:617–623. https://doi.org/10.2217/ijr.11.62
Cohen EM, Edwards RR, Bingham CO, Phillips K, Bolster MB, Moreland LW et al (2019) Pain and catastrophizing in patients with rheumatoid arthritis: an observational cohort study. J Clin Rheumatol 25:232–236. https://doi.org/10.1097/RHU.0000000000000834
Brandstetter S, Riedelbeck G, Steinmann M, Loss J, Ehrenstein B, Apfelbacher C (2018) Depression moderates the associations between beliefs about medicines and medication adherence in patients with rheumatoid arthritis: cross-sectional study. J Health Psychol 23:1185–1195. https://doi.org/10.1177/1359105316646440
Nerurkar L, Siebert S, McInnes IB, Cavanagh J (2019) Rheumatoid arthritis and depression: an inflammatory perspective. The Lancet Psychiatry 6:164–173. https://doi.org/10.1016/S2215-0366(18)30255-4
Barnes J, Mondelli V, Pariante CM (2017) Genetic contributions of inflammation to depression, pp 81–98. https://doi.org/10.1038/npp.2016.169.
Davies KA, Cooper E, Voon V, Tibble J, Cercignani M, Harrison NA (2020) Interferon and anti-TNF therapies differentially modulate amygdala reactivity which predicts associated bidirectional changes in depressive symptoms. Mol Psychiatry. https://doi.org/10.1038/s41380-020-0790-9
Ionescu CE, Popescu CC, Agache M, Dinache G, Codreanu C (2022) Depression in rheumatoid arthritis: a narrative review-diagnostic challenges, pathogenic mechanisms and effects. Medicina (Kaunas). https://doi.org/10.3390/medicina58111637
Vallerand IA, Lewinson RT, Frolkis AD, Lowerison MW, Kaplan GG, Swain MG et al (2018) Depression as a risk factor for the development of rheumatoid arthritis: a population-based cohort study. RMD Open 4:e000670. https://doi.org/10.1136/rmdopen-2018-000670
Qiu X-J, Zhang X-L, Cai L-S, Yan C, Yu L, Fan J et al (2019) Rheumatoid arthritis and risk of anxiety: a meta-analysis of cohort studies. Clin Rheumatol 38:2053–2061. https://doi.org/10.1007/s10067-019-04502-8
Godha D, Shi L, Mavronicolas H (2010) Association between tendency towards depression and severity of rheumatoid arthritis from a national representative sample: The medical expenditure panel survey. Curr Med Res Opin 26:1685–1690. https://doi.org/10.1185/03007991003795808
Kojima T, Yabe Y, Kaneko A, Hirano Y, Ishikawa H, Hayashi M et al (2013) Monitoring C-reactive protein levels to predict favourable clinical outcomes from tocilizumab treatment in patients with rheumatoid arthritis. Mod Rheumatol 23:977–985. https://doi.org/10.1007/s10165-012-0782-y
Boers M, Kirwan JR, Tugwell P. The OMERACT Handbook. Unkn Publ http://www.OmeractOrg 2014:2023.
Kuijper TM, Luime JJ, Xiong H, de Jong PHP, van der Lubbe PAHM, van Zeben D et al (2018) Effects of psychosocial factors on monitoring treatment effect in newly diagnosed rheumatoid arthritis patients over time: response data from the tREACH study. Scand J Rheumatol 47:178–184. https://doi.org/10.1080/03009742.2017.1349176
Matcham F, Davies R, Hotopf M, Hyrich KL, Norton S, Steer S et al (2018) The relationship between depression and biologic treatment response in rheumatoid arthritis: An analysis of the British Society for Rheumatology Biologics Register. Rheumatol (United Kingdom) 57:835–843. https://doi.org/10.1093/rheumatology/kex528
Euesden J, Matcham F, Hotopf M, Steer S, Cope AP, Lewis CM et al (2017) The relationship between mental health, disease severity, and genetic risk for depression in early rheumatoid arthritis. Psychosom Med 79:638–645. https://doi.org/10.1097/PSY.0000000000000462
Overman CL, Bossema ER, van Middendorp H, Wijngaards-de Meij L, Verstappen SM, Bulder M et al (2011) The prospective association between psychological distress and disease activity in rheumatoid arthritis: a multilevel regression analysis. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2011-200338
Michelsen B, Fagerli KM, Lie E, Hammer HB, Haugeberg G, Kristianslund EK et al (2016) Do depression and anxiety reduce the chance of remission in rheumatoid arthritis and psoriatic arthritis? Arthritis Rheumatol 68:4122–4124. https://doi.org/10.1002/art.39977
Dyball S, Cordingley L, Isaacs JD, Wilson G, Morgan A, Hyrich K et al (2018) Pre-treatment anxiety and depression levels are correlated with less improvement in selfreported disease activity in rheumatoid arthritis patients treated with biological therapy: results from the Braggss cohort. Rheumatol (United Kingdom). https://doi.org/10.1093/rheumatology/key075.455
Matcham F, Ali S, Irving K, Hotopf M (2016) Are depression and anxiety associated with disease activity in rheumatoid arthritis? A prospective study. BMC Musculoskelet Disord 17:155. https://doi.org/10.1186/s12891-016-1011-1
Matcham F, Norton S, Scott DL, Steer S, Hotopf M. The Longitudinal Impact of Persistent Depression on Physical Health Outcomes in Rheumatoid Arthritis: Table 1. Ann Rheum Dis 2014;73:1197.1–1197. https://doi.org/10.1136/annrheumdis-2014-eular.2878.
Thompson T, Correll CU, Gallop K, Vancampfort D, Stubbs B (2016) Is Pain Perception Altered in People With Depression? A Systematic Review and Meta-Analysis of Experimental Pain Research. J Pain 17:1257–1272. https://doi.org/10.1016/j.jpain.2016.08.007
Odegard S, Finset A, Mowinckel P, Kvien TK, Uhlig T (2007) Pain and psychological health status over a 10-year period in patients with recent onset rheumatoid arthritis. Ann Rheum Dis 66:1195–1201. https://doi.org/10.1136/ard.2006.064287
Norton S, Sacker A, Young A, Done J (2011) Distinct psychological distress trajectories in rheumatoid arthritis: Findings from an inception cohort. J Psychosom Res 71:290–295. https://doi.org/10.1016/j.jpsychores.2011.05.006
Gonzalez-Lopez L, Morales-Romero J, Vazquez-Villegas ML, Villa-Manzano R, Rocha-Muñoz AD, Barragan-Enriquez A et al (2013) Factors influencing sick leave episodes in Mexican workers with rheumatoid arthritis and its impact on working days lost. Rheumatol Int 33:561–569. https://doi.org/10.1007/s00296-012-2395-3
Peeters F, Berkhof J, Delespaul P, Rottenberg J, Nicolson NA (2006) Diurnal mood variation in major depressive disorder. Emotion 6:383–391. https://doi.org/10.1037/1528-3542.6.3.383
Boyer P (2000) Do anxiety and depression have a common pathophysiological mechanism? Acta Psychiatr Scand Suppl 102:24–29. https://doi.org/10.1111/j.0065-1591.2000.acp29-04.x
Zhao S, Chen Y, Chen H (2015) Sociodemographic factors associated with functional disability in outpatients with rheumatoid arthritis in Southwest China. Clin Rheumatol 34:845–851. https://doi.org/10.1007/s10067-015-2896-z
Matcham F, Norton S, Scott DL, Steer S, Hotopf M (2015) Symptoms of depression and anxiety predict treatment response and long-term physical health outcomes in rheumatoid arthritis: secondary analysis of a randomized controlled trial. Rheumatol (United Kingdom) 55:268–278. https://doi.org/10.1093/rheumatology/kev306
van den Hoek J, Boshuizen HC, Roorda LD, Tijhuis GJ, Nurmohamed MT, Dekker J (2016) Association of somatic comorbidities and comorbid depression with mortality in patients with rheumatoid arthritis: a 14-year prospective cohort study. Arthritis Care Res 68:1055–1060. https://doi.org/10.1002/acr.22812
Miwa Y, Takahashi R, Isojima S, Isozaki T, Saito M, Oguro N et al (2015) A study on characteristics of rheumatoid arthritis patients achieving clinical remission after 6 months of treatment with biologic agents. Ann Rheum Dis 74:1003. https://doi.org/10.1136/annrheumdis-2015-eular.1124
Brown GK (1990) A causal analysis of chronic pain and depression. J Abnorm Psychol 99:127–137
Kronisch C, Mclernon D, Dale J, Paterson C, Ralston SH, Reid DM, et al (2015) A Clinical Risk Score to Predict Functional Disability at 1 Year in an Early Rheumatoid Arthritis Inception Cohort. ACR/ARHP Annu Meet 3254:2555–2794
Corominas H, Miguel CA de, Rodríguez-Gόmez M, Fernandez-Cid CM, Maceiras-Pan F (2014) fri0317 act-axis study: impact of Roactemra® (TOCILIZUMAB) on fatigue and related factors in patients with arthritis rheumatoid in daily clinical practice in Spain. Ann Rheum Dis 73:500
Gwinnutt J, Leggett S, Lunt M, Barton A, Hyrich K, Walker-Bone K (2019) Anxiety, occupation and functional ability at the start of treatment and EULAR response are the main predictors of work outcomes in patients with rheumatoid arthritis commencing either methotrexate or biologics. Rheumatol (United Kingdom). https://doi.org/10.1093/rheumatology/kez107.017
Rathbun A, Harrold L, Reed G (2016) A Prospective evaluation of the effects of prevalent depressive symptoms on disease activity in rheumatoid arthritis patients treated with biologic response modifiers. Clin Ther 38:1759-1772.e3. https://doi.org/10.1016/j.clinthera.2016.06.007
Kronisch C, McLernon DJ, Dale J, Paterson C, Ralston SH, Reid DM et al (2016) Brief report: predicting functional disability: one-year results from the scottish early rheumatoid arthritis inception cohort. Arthritis Rheumatol 68:1596–1602. https://doi.org/10.1002/art.39627
Miwa Y, Yajima N, Isozaki T, Takahashi R, Miura Y, Ikari Y et al (2017) A study on characteristics of rheumatoid arthritis patients achieving depression remission with 6 months of biologic agent treatment. Ann Rheum Dis 76:800–801. https://doi.org/10.1136/annrheumdis-2017-eular.1443
Ang DC, Choi H, Kroenke K, Wolfe F (2005) Comorbid depression is an independent risk factor for mortality in patients with rheumatoid arthritis. J Rheumatol 32:1013–1019
Parenti D, Reed G, Kafka S, Ellis L, J.D. Greenberg R, DeHoratius J. THU0130 Comorbidities and Efficacy of anti-TNF Therapies: History of Depression as A Possible Indicator of Lower Response. Ann Rheum Dis 2016;75.
Cui K, Bombardier C, Tomlinson GA. Contributions of Social Determinants of Health on Probability of Remission in Early and Established Rheumatoid Arthritis Patients. ACR/ARHP Annu Meet 2015;45.
Corominas H, Alegre C, Narváez J, Fernández-Cid C, Torrente-Segarra V, Gómez MR et al (2019) Correlation of fatigue with other disease related and psychosocial factors in patients with rheumatoid arthritis treated with tocilizumab: ACT-AXIS study. Medicine (Baltimore) 98:e15947. https://doi.org/10.1097/MD.0000000000015947
Michelsen B, Kristianslund EK, Sexton J, Hammer HB, Fagerli KM, Lie E et al (2017) Do depression and anxiety reduce the likelihood of remission in rheumatoid arthritis and psoriatic arthritis? Data from the prospective multicentre NOR-DMARD study. Ann Rheum Dis 76:1906–1910. https://doi.org/10.1136/annrheumdis-2017-211284
Kleinert S, Marx A, Faller H, Tony H-P, Feuchtenberger M, Kneitz C, et al (2016) FRI0150 depression, inflammation and mortality in patients with rheumatoid arthritis. Nnals Rheum Dis 75:483
Hider SL, Tanveer W, Brownfield A, Mattey DL, Packham JC (2009) Depression in RA patients treated with anti-TNF is common and under-recognized in the rheumatology clinic. Rheumatology 48:1152–1154. https://doi.org/10.1093/rheumatology/kep170
Bode C, Tonner C, Trupin L. Depression predicts mortality in RA. Arthritis Rheum 2012;64:S380. https://doi.org/10.1002/art.37735.
Leblanc-Trudeau C, Dobkin PL, Carrier N, Cossette P, Brum-Fernandes AJd, Liang P et al (2015) Depressive symptoms predict future simple disease activity index scores and simple disease activity index remission in a prospective cohort of patients with early inflammatory polyarthritis. Rheumatol (United Kingdom) 54:2205–2214. https://doi.org/10.1093/rheumatology/kev272
McFarlane AC, Brooks PM (1988) An analysis of the relationship between psychological morbidity and disease activity in rheumatoid arthritis. J Rheumatol 15:926–931
Norton SJ, Young A, Sacker A (2011) Distinct trajectories of psychological distress over 10 years following diagnosis of rheumatoid arthritis are related to subjective appraisal of disease severity and mortality. Rheumatology 50:35–36
Michelsen B, Kristianslund EK, Fageli KM, Lie E, Hammer HB, Haugeberg G, et al (2017) Depression and anxiety reduce the likelihood of achieving remission in patients with rheumatoid arthritis: real life data from the NOR-DMARD Study. Ann Rheum Dis 76:229–230
Feldthusen C, Grimby-Ekman A, Forsblad-D’Elia H, Jacobsson L, Mannerkorpi K (2016) Explanatory factors and predictors of fatigue in persons with rheumatoid arthritis: a longitudinal study. J Rehabil Med 48:469–476. https://doi.org/10.2340/16501977-2090
Tanaka Y, Kameda H, Kazuyoshi S, Kaneko Y, Tanaka E, Yasuda S et al (2019) Factors that affect the response to tocilizumab with regard to work productivity in patients with rheumatoid arthritis: A 2-year follow-up of the first ACTSC study. Ann Rheum Dis 78:1667. https://doi.org/10.1136/annrheumdis-2019-eular.1168
Doeglas DM, Suurmeijer TPBM, Van Den Heuvel WJA, Krol B, Van Rijswijk MH, Van Leeuwen MA et al (2004) Functional ability, social support, and depression in rheumatoid arthritis. Qual Life Res 13:1053–1065. https://doi.org/10.1023/B:QURE.0000031339.04589.63
Verstappen SMM, Jacobs JWG, Huisman A-M, Van Rijthoven AWAM, Sokka T, Bijlsma JWJ (2007) Functional Health Assessment Questionnaire (HAQ) and psychological HAQ are associated with and predicted by different factors in rheumatoid arthritis. J Rheumatol 34:1837–1840
Hommel KA, Chaney JM, Mullins LL, Palmer W, Wees S, Klein H (1998) Relative contributions of attributional style and arthritis helplessness to depression in rheumatoid arthritis: a longitudinal investigation. Int J Rehabil Heal 4:59–67. https://doi.org/10.1023/A:1022999704062
Fifield J, McQuillan J, Tennen H, Sheehan TJ, Reisine S, Hesselbrock V et al (2001) History of affective disorder and the temporal trajectory of fatigue in rheumatoid arthritis. Ann Behav Med 23:34–41. https://doi.org/10.1207/S15324796ABM2301_6
Looper K, Mustafa S, Zelkowitz P, Purden M, Baron M (2011) History of depressive episodes as a risk factor for illness severity in early inflammatory arthritis. J Psychosom Res 71:28–31. https://doi.org/10.1016/j.jpsychores.2010.12.012
Nugaliyadde A, Culfear K, Nandagudi A (2017) High prevalence of depression and its impact on disease activity in patients with rheumatoid arthritis in a district general hospital of the United Kingdom. Rheumatol (United Kingdom). https://doi.org/10.1093/rheumatology/kex062.001
Chung A, Lau W, Perera C, Doraj R, Khoo K, Tymms K (2013) ARA-P18 Impact of mental health on physical outcomes in rheumatoid arthritis patients on BDMARDs. Intern Med J 43:14
England B, Cordingley L, Plant D, Morgan A, Wilson A, Hyrich KL (2015) Investigating psychological predictors of biologic treatment response in patients with severe active rheumatoid arthritis. Arthritis Rheumatol. https://doi.org/10.1002/art.39448
Sergeant JC, Hope HF, Anderson J, Barton A, Hyrich KL (2015) Lifestyle, clinical and psychosocial predictors of good response to methotrexate therapy in the rheumatoid arthritis medication study (RAMS). Ann Rheum Dis 74:783–784. https://doi.org/10.1136/annrheumdis-2015-eular.5305
Morris A, Yelin EH, Panopalis P, Julian L, Katz PP (2011) Long-term patterns of depression and associations with health and function in a panel study of rheumatoid arthritis. J Health Psychol 16:667–677. https://doi.org/10.1177/1359105310386635
van den Hoek J, Roorda LD, Boshuizen HC, van Hees J, Rupp I, Tijhuis GJ et al (2013) Long-term physical functioning and its association with somatic comorbidity and comorbid depression in patients with established rheumatoid arthritis: a longitudinal study. Arthritis Care Res (Hoboken) 65:1157–1165. https://doi.org/10.1002/acr.21950
McFarlane AC, Kalucy RS, Brooks PM (1987) Psychological predictors of disease course in rheumatoid arthritis. J Psychosom Res 31:757–764. https://doi.org/10.1016/0022-3999(87)90024-9
Vriezekolk J, Eijsbouts A, Evers A, Stenger A, Van Den Hoogen F, Van Lankveld W (2010) Poor psychological health status among patients with inflammatory rheumatic diseases and osteoarthritis in multidisciplinary rehabilitation: need for a routine psychological assessment. Disabil Rehabil 32:836–844. https://doi.org/10.3109/09638280903323250
Sergeant JC, Hyrich KL, Anderson J, Kopec-Harding K, Hope HF, Barton A (2016) Prediction of non-response to methotrexate therapy in the rheumatoid arthritis medication study (RAMS). Ann Rheum Dis 75:57–58. https://doi.org/10.1136/annrheumdis-2016-eular.4282
Sergeant JC, Hyrich KL, Anderson J, Kopec-Harding K, Hope HF, Symmons DPM et al (2018) Prediction of primary non-response to methotrexate therapy using demographic, clinical and psychosocial variables: results from the UK Rheumatoid Arthritis Medication Study (RAMS). Arthritis Res Ther. https://doi.org/10.1186/s13075-018-1645-5
Casalla L, del Moral RC, Rillo OL, Benegas M, Pra FD, Ficco HM et al (2013) FRI0073 Predictive factors for unfavorable outcomes in early rheumatoid arthritis: a prospective multicenter cohort study in argentina. Ann Rheum Dis 72:A393–A394
Cook MJ, Diffin J, Scirè CA, Lunt M, MacGregor AJ, Symmons DPM et al (2016) Predictors and outcomes of sustained, intermittent or never achieving remission in patients with recent onset inflammatory polyarthritis: results from the Norfolk Arthritis Register. Rheumatol (United Kingdom) 55:1601–1609. https://doi.org/10.1093/rheumatology/kew210
Treharne GJ, Lyons AC, Hale ED, Goodchild CE, Booth DA, Kitas GD (2008) Predictors of fatigue over 1 year among people with rheumatoid arthritis. Psychol Heal Med 13:494–504. https://doi.org/10.1080/13548500701796931
Dobkin PL, Liu A, Abrahamowicz M, Carrier N, De Brum-Fernandes AJ, Cossette P et al (2013) Predictors of pain for patients with early inflammatory polyarthritis. Arthritis Care Res 65:992–999. https://doi.org/10.1002/acr.21923
Leggett S, Lunt M, Barton A, Hyrich K, Walker-Bone K (2017) Predictors of presenteeism and absenteeism in patients commencing treatment with methotrexate monotherapy or biologic therapy for rheumatoid arthritis. Ann Rheum Dis 76:765. https://doi.org/10.1136/annrheumdis-2017-eular.5077
Schieir O, Bartlett SJ, Hitchon C, Pope JE, Boire G, Haraoui B et al (2016) Prevalence and impact of inflammatory multimorbid conditions on trajectories of disease activity in the first year of follow-up in a multi-center era cohort. Arthritis Rheumatol Conf Am Coll Rheumatol Rheumatol Heal Prof Annu Sci Meet ACR/ARHP 2016 United States Conf Start 20161111 Conf End 68:1911–1912. https://doi.org/10.1002/art.39977
Parker J, Smarr K, Anderson S, Hewett J, Walker S, Bridges A et al (1992) Relationship of changes in helplessness and depression to disease activity in rheumatoid arthritis. J Rheumatol 19:1901–1905
Iannaccone C, Mahmoud TG, Cui J, Weinblatt M, Shadick NA (2016) Symptoms of depression and anxiety predict worse disease activity and functional disability in a cohort of established rheumatoid arthritis patients. Arthritis Rheumatol 86:1–4550
Schieir O, Thombs BD, Hudson M, Taillefer S, Steele R, Berkson L et al (2009) Symptoms of depression predict the trajectory of pain among patients with early inflammatory arthritis: A path analysis approach to assessing change. J Rheumatol 36:231–239. https://doi.org/10.3899/jrheum.080147
Rathbun AM, Reed GW, Harrold LR (2013) The temporal relationship between depression and rheumatoid arthritis disease activity, treatment persistence and response: a systematic review. Rheumatol (United Kingdom) 52:1785–1794. https://doi.org/10.1093/rheumatology/kes356
Rathbun A, Harrold L, Reed G (2015) Temporal effect of depressive symptoms on the longitudinal evolution of rheumatoid arthritis disease activity. Arthritis Care Res (Hoboken) 67:765–775. https://doi.org/10.1002/acr.22515
El Miedany Y, El Gaafary M, El Arousy N, Youssef S (2013) The correlation of self-reported behavioral co-morbidities and disease activity in early inflammatory arthritis patients: a prospective study. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2012-eular.2596
Uhlig T, Smedstad LM, Vaglum P, Moum T, Gérard N, Kvien TK (2000) The course of rheumatoid arthritis and predictors of psychological, physical and radiographic outcome after 5 years of follow-up. Rheumatology 39:732–741. https://doi.org/10.1093/rheumatology/39.7.732
Li N, Chan E, Peterson S (2019) The economic burden of depression among adults with rheumatoid arthritis in the United States. J Med Econ 22:372–378. https://doi.org/10.1080/13696998.2019.1572015
Van Den Hoek J, Roorda LD, Boshuizen H, Dekker J, Van Hees J, Rupp I et al (2013) The influence of somatic and depression comorbidity on physical functioning over eleven years in patients with rheumatoid arthritis. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2012-eular.2573
Matcham F, Norton S, Scott D, Steer S, Hotopf M (2014) The impact of depression on long-term physical health outcomes in rheumatoid arthritis. Rheumatol (United Kingdom) 53:i103–i104. https://doi.org/10.1093/rheumatology/keu103.005
Crotty M, Mcfarlane AC, Brooks PM, Hopper JL, Bieri D, Taylor SJ (1994) The psychosocial and clinical status of younger women with early rheumatoid arthritis: a longitudinal study with frequent measures. Rheumatology 33:754–760. https://doi.org/10.1093/rheumatology/33.8.754
Smedstad LM, Vaglum P, Moum T, Kvien TK (1997) The relationship between psychological distress and traditional clinical variables: a 2 year prospective study of 216 patients with early rheumatoid arthritis. Br J Rheumatol 36:1304–1311
Karpouzas GA, Draper T, Moran R, Hernandez E, Nicassio P, Weisman MH et al (2017) Trends in functional disability and determinants of clinically meaningful change over time in hispanic patients with rheumatoid arthritis in the US. Arthritis Care Res 69:294–298. https://doi.org/10.1002/acr.22924
Author information
Authors and Affiliations
Contributions
Melissa Sweeney and Sam Norton conceptualized and designed the review. The literature review was completed by Melissa Sweeney. Data analysis was completed by Melissa Sweeney with secondary review by Maryam Adas. Writing of the original draft was completed by Melissa Sweeney. Editing was completed by Melissa Sweeney, Andrew Cope, Maryam Adas, and Sam Norton.
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare they have no conflicts of interest.
Ethical approval
This manuscript does not contain clinical studies or patient data. Authors have full control of all primary data and agree to allow the journal to review their data if requested.
Human and animal rights
This review did not involve any human subjects and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sweeney, M., Adas, M.A., Cope, A. et al. Longitudinal effects of affective distress on disease outcomes in rheumatoid arthritis: a meta-analysis and systematic review. Rheumatol Int (2024). https://doi.org/10.1007/s00296-024-05574-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00296-024-05574-9