Abstract
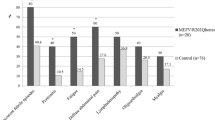
Many studies have shown that oxidative stress levels increase in patients with Familial Mediterranean Fever (FMF). Thiols are a class of compounds that include a sulfhydryl group (–SH) and can react with free oxygen radicals to protect tissues. We aimed to investigate thiol–disulphide homeostatic status in FMF patients and examined the effect of different mutations in the MEFV gene on the thiol–disulphide balance. We investigated thiol–disulphide parameters in patients with FMF and healthy controls. To determine the differential effect of MEFV gene mutations on thiol–disulphide balance, subjects were divided into five groups based on homozygous or compound heterozygous exon 10 and nonexon 10 mutations. Tests of thiol–disulphide homeostasis were conducted using the automated spectrophotometric method. Patients with FMF had significantly lower native thiol [433.8 µmol/l (243.3–536.4) vs. 484.1 µmol/L (340.2–612.3), p < 0.001], total thiol levels [459.7 µmol/L (281.3–575.4) vs. 529.9 µmol/L (363–669.5), p < 0.001], and disulphide levels [14.0 µmol/l (2.7–33.3) vs. 24.4 µmol/l (7.2–36.6), p < 0.001] compared to the control group. Moreover, disulphide/native thiol (3.4 ± 1.7 vs. 4.7 ± 1.3, p < 0.001) and disulphide/total thiol (3.1 ± 1.4 vs. 4.3 ± 1.0 p < 0.001) were also detected lower in the FMF group compared to the control group. But the native thiol/total thiol ratios (93.6 ± 2.9 vs. 91.3 ± 2.1, p < 0.001) were higher in the FMF group. There was no significant difference between the native thiol, total thiol, and disulphide levels of individuals with nonexon 10 homozygous or compound heterozygous (Group 1), nonexon 10–exon 10 compound heterozygous (Group 2), exon 10 homozygous or compound heterozygous (Group 3), and heterozygous (Group 4) mutations. However, these parameters significantly differed from those of the healthy control group. Since no differences were found in our study between thiol and disulfide levels of Groups 1, 2 and 3, we believe that this rate cannot be shown as an indicator of oxidative damage in different mutations of FMFs. To the best of our knowledge, this study is the first study that demonstrates the effect of different FMF mutations on the thiol–disulphide balance.
Similar content being viewed by others
References
Dundar M, Kiraz A, Balta B, Emirogullari EF, Zararsiz G, Yurci A, Aslan D, Baskol M (2013) The role of TNF-alpha and PAI-1 gene polymorphisms in familial mediterranean fever. Mod Rheumatol 23(1):140–145. https://doi.org/10.1007/s10165-012-0687-9
French FMFC. (1997) A candidate gene for familial mediterranean fever. Nat Genet 17(1):25–31. https://doi.org/10.1038/ng0997-25
Dogan HO, Omma A, Turhan T, Bogdaycioglu N, Karaaslan Y, Yavuz H, Demirpence O, Aydin H, Bakir S (2016) Decreased chitotriosidase activity and levels in familial mediterranean fever. J Korean Med Sci 31(12):1902–1906. https://doi.org/10.3346/jkms.2016.31.12.1902
Frijhoff J, Winyard PG, Zarkovic N, Davies SS, Stocker R, Cheng D, Knight AR, Taylor EL, Oettrich J, Ruskovska T, Gasparovic AC, Cuadrado A, Weber D, Poulsen HE, Grune T, Schmidt HH, Ghezzi P (2015) Clinical relevance of biomarkers of oxidative stress. Antioxid Redox Signal 23(14):1144–1170. https://doi.org/10.1089/ars.2015.6317
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48(6):749–762. https://doi.org/10.1016/j.freeradbiomed.2009.12.022
Kirlin WG, Cai J, Thompson SA, Diaz D, Kavanagh TJ, Jones DP (1999) Glutathione redox potential in response to differentiation and enzyme inducers. Free Radic Biol Med 27(11–12):1208–1218
Nkabyo YS, Ziegler TR, Gu LH, Watson WH, Jones DP (2002) Glutathione and thioredoxin redox during differentiation in human colon epithelial (Caco-2) cells. Am J Physiol Gastrointest Liver Physiol 283(6):G1352–G1359. https://doi.org/10.1152/ajpgi.00183.2002
Tetik S, Ahmad S, Alturfan AA, Fresko I, Disbudak M, Sahin Y, Aksoy H, Yardimci KT (2010) Determination of oxidant stress in plasma of rheumatoid arthritis and primary osteoarthritis patients. Indian J Biochem Biophys 47(6):353–358
Go YM, Jones DP (2011) Cysteine/cystine redox signaling in cardiovascular disease. Free Radic Biol Med 50(4):495–509. https://doi.org/10.1016/j.freeradbiomed.2010.11.029
Prabhu A, Sarcar B, Kahali S, Yuan Z, Johnson JJ, Adam KP, Kensicki E, Chinnaiyan P (2014) Cysteine catabolism: a novel metabolic pathway contributing to glioblastoma growth. Cancer Res 74(3):787–796. https://doi.org/10.1158/0008-5472.CAN-13-1423
Smeyne M, Smeyne RJ (2013) Glutathione metabolism and Parkinson’s disease. Free Radic Biol Med 62:13–25. https://doi.org/10.1016/j.freeradbiomed.2013.05.001
Sbrana E, Paladini A, Bramanti E, Spinetti MC, Raspi G (2004) Quantitation of reduced glutathione and cysteine in human immunodeficiency virus-infected patients. Electrophoresis 25(10–11):1522–1529. https://doi.org/10.1002/elps.200305848
Dundar M, Emirogullari EF, Kiraz A, Taheri S, Baskol M (2011) Common familial mediterranean fever gene mutations in a Turkish cohort. Mol Biol Rep 38(8):5065–5069. https://doi.org/10.1007/s11033-010-0652-7
Dusunsel R, Dursun I, Gunduz Z, Poyrazoglu MH, Gurgoze MK, Dundar M (2008) Genotype-phenotype correlation in children with familial mediterranean fever in a Turkish population. Pediatr Int 50(2):208–212. https://doi.org/10.1111/j.1442-200X.2008.02554.x
Ozturk C, Halicioglu O, Coker I, Gulez N, Sutcuoglu S, Karaca N, Aksu G, Kutukculer N (2012) Association of clinical and genetical features in FMF with focus on MEFV strip assay sensitivity in 452 children from western Anatolia, Turkey. Clin Rheumatol 31(3):493–501. https://doi.org/10.1007/s10067-011-1876-1
Giaglis S, Papadopoulos V, Kambas K, Doumas M, Tsironidou V, Rafail S, Kartalis G, Speletas M, Ritis K (2007) MEFV alterations and population genetics analysis in a large cohort of Greek patients with familial mediterranean fever. Clin Genet 71(5):458–467. https://doi.org/10.1111/j.1399-0004.2007.00789.x
Mattit H, Joma M, Al-Cheikh S, El-Khateeb M, Medlej-Hashim M, Salem N, Delague V, Megarbane A (2006) Familial mediterranean fever in the Syrian population: gene mutation frequencies, carrier rates and phenotype-genotype correlation. Eur J Med Genet 49(6):481–486. https://doi.org/10.1016/j.ejmg.2006.03.002
Sönmez HE, Batu ED, Özen S (2016) Familial Mediterranean fever: current perspectives. J Inflamm Res 9:13–20. https://doi.org/10.2147/JIR.S91352
Shinar Y, Obici L, Aksentijevich I, Bennetts B, Austrup F, Ceccherini I, Costa JM, De Leener A, Gattorno M, Kania U, Kone-Paut I, Lezer S, Livneh A, Moix I, Nishikomori R, Ozen S, Phylactou L, Risom L, Rowczenio D, Sarkisian T, van Gijn ME, Witsch-Baumgartner M, Morris M, Hoffman HM, Touitou I, European Molecular Genetics Quality N (2012) Guidelines for the genetic diagnosis of hereditary recurrent fevers. Ann Rheum Dis 71 (10):1599–1605. https://doi.org/10.1136/annrheumdis-2011-201271
Yilmaz E, Ozen S, Balci B, Duzova A, Topaloglu R, Besbas N, Saatci U, Bakkaloglu A, Ozguc M (2001) Mutation frequency of familial mediterranean fever and evidence for a high carrier rate in the Turkish population. Eur J Hum Genet 9(7):553–555. https://doi.org/10.1038/sj.ejhg.5200674
Milenkovic J, Vojinovic J, Debeljak M, Toplak N, Lazarevic D, Avcin T, Jevtovic-Stoimenov T, Pavlovic D, Bojanic V, Milojkovic M, Kocic G, Veljkovic A (2016) Distribution of MEFV gene mutations and R202Q polymorphism in the Serbian population and their influence on oxidative stress and clinical manifestations of inflammation. Pediatr Rheumatol Online J 14(1):39. https://doi.org/10.1186/s12969-016-0097-1
Omma A, Sandikci SC, Kucuksahin O, Alisik M, Erel O (2017) Can the thiol/disulfide imbalance be a predictor of colchicine resistance in familial mediterranean fever? J Korean Med Sci 32(10):1588–1594. https://doi.org/10.3346/jkms.2017.32.10.1588
Yucel A, Sanhal CY, Daglar K, Kara O, Uygur D, Erel O (2016) Thiol/disulphide homeostasis in pregnant women with familial mediterranean fever. Redox Rep 21(6):287–291. https://doi.org/10.1080/13510002.2016.1168590
Erel O, Neselioglu S (2014) A novel and automated assay for thiol/disulphide homeostasis. Clin Biochem 47(18):326–332. https://doi.org/10.1016/j.clinbiochem.2014.09.026
Fridovich I (1999) Fundamental aspects of reactive oxygen species, or what’s the matter with oxygen? Ann N Y Acad Sci 893:13–18
Fang YZ, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18(10):872–879
Karaguezyan KG, Haroutjunian VM, Mamiconyan RS, Hakobian GS, Nazaretian EE, Hovsepyan LM, Hoveyan GA, Gevorkian EM, Hovakimyan SS, Zakarian AE, Quinn PJ (1996) Evidence of oxidative stress in erythrocyte phospholipid composition in the pathogenesis of familial mediterranean fever (periodical disease). J Clin Pathol 49(6):453–455
Kirkali G, Tunca M, Genc S, Jaruga P, Dizdaroglu M (2008) Oxidative DNA damage in polymorphonuclear leukocytes of patients with familial mediterranean fever. Free Radic Biol Med 44(3):386–393. https://doi.org/10.1016/j.freeradbiomed.2007.09.020
Guzel S, Andican G, Seven A, Aslan M, Bolayirli M, Guzel EC, Hamuryudan V (2012) Acute phase response and oxidative stress status in familial mediterranean fever (FMF). Mod Rheumatol 22(3):431–437. https://doi.org/10.1007/s10165-011-0517-5
Biswas S, Chida AS, Rahman I (2006) Redox modifications of protein-thiols: emerging roles in cell signaling. Biochem Pharmacol 71(5):551–564. https://doi.org/10.1016/j.bcp.2005.10.044
Lim JC, Choi HI, Park YS, Nam HW, Woo HA, Kwon KS, Kim YS, Rhee SG, Kim K, Chae HZ (2008) Irreversible oxidation of the active-site cysteine of peroxiredoxin to cysteine sulfonic acid for enhanced molecular chaperone activity. J Biol Chem 283(43):28873–28880. https://doi.org/10.1074/jbc.M804087200
Booth DR, Gillmore JD, Lachmann HJ, Booth SE, Bybee A, Soyturk M, Akar S, Pepys MB, Tunca M, Hawkins PN (2000) The genetic basis of autosomal dominant familial mediterranean fever. QJM 93(4):217–221
Aldea A, Campistol JM, Arostegui JI, Rius J, Maso M, Vives J, Yague J (2004) A severe autosomal-dominant periodic inflammatory disorder with renal AA amyloidosis and colchicine resistance associated to the MEFV H478Y variant in a Spanish kindred: an unusual familial mediterranean fever phenotype or another MEFV-associated periodic inflammatory disorder? Am J Med Genet A 124A(1):67–73. https://doi.org/10.1002/ajmg.a.20296
Tunca M, Kirkali G, Soyturk M, Akar S, Pepys MB, Hawkins PN (1999) Acute phase response and evolution of familial mediterranean fever. Lancet 353(9162):1415. https://doi.org/10.1016/S0140-6736(99)00990-3
Lachmann HJ, Sengul B, Yavuzsen TU, Booth DR, Booth SE, Bybee A, Gallimore JR, Soyturk M, Akar S, Tunca M, Hawkins PN (2006) Clinical and subclinical inflammation in patients with familial mediterranean fever and in heterozygous carriers of MEFV mutations. Rheumatology 45(6):746–750. https://doi.org/10.1093/rheumatology/kei279
Acknowledgements
The authors wish to thank the patients and healthy individuals who volunteered to participate in this study and also acknowledge Scribendi Editing Service editors for editing and proofreading of the article.
Author information
Authors and Affiliations
Contributions
TA and FB provided patient samples. MA and OE performed thiol–disulphide biochemical analysis. BB, ME and AK analyzed MEFV gene mutations and jointly wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interests.
Ethical approval
The study was approved by the Local Ethics Research Committee of Erciyes University dated 24.06.2016 with the protocol number 2016 –384 and conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.
Informed consent
All subjects provided written informed consent prior to participation in the study.
Rights and permissions
About this article
Cite this article
Balta, B., Erdogan, M., Alisik, M. et al. Does thiol–disulphide balance show oxidative stress in different MEFV mutations?. Rheumatol Int 38, 97–104 (2018). https://doi.org/10.1007/s00296-017-3914-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3914-z