Abstract
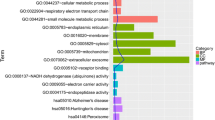
Our study aimed to identify candidate genes associated with Dupuytren’s contracture (DC) and elucidate their roles in DC development. The microarray data of GSE21221 were downloaded from Gene Expression Omnibus database, including six samples from carpal tunnel-derived fibroblasts and six samples from DC-derived fibroblasts. The differentially expressed genes (DEGs) in DC samples were screened using limma package. GO annotation and KEGG pathway analyses were performed by DAVID online tool. Protein–protein interaction network and expression correlation network were constructed to identify crucial relationships between DEGs. Finally, candidate DC-associated genes were predicted based on comparative toxicogenomics database. A total of 529 DEGs (138 up- and 391 down-regulated) in DC-derived fibroblasts were screened and compared with carpal tunnel-derived fibroblasts. Only ten DC-associated genes, such as neurotrophin 3 (NTF3) and protein kinase C, epsilon (PRKCE), were further screened. In addition, NTF3 was significantly enriched in MAPK signaling pathway, in which other DEGs, such as nuclear receptor subfamily 4, group A, member 1 (NR4A1), fibroblast growth factor 22 (FGF22) and BDNF, were enriched. Besides, NTF3 could co-express with fibrillin 2 (FBN2), and PRKCE could co-express with zinc finger protein 516 (ZNF516), solute carrier organic anion transporter family, member 2A1 (SLCO2A1), chromosome 10 open reading frame 10 (C10orf10) and Kelch domain containing 7A (KLHDC7A). Our study indicates that these DEGs, including NTF3, FBN2, NR4A1, FGF22, BDNF, PRKCE, ZNF516, SLCO2A1, C10orf10 and KLHDC7A, may play important roles in DC development and serve as candidate molecular targets for treating DC.




Similar content being viewed by others
References
Wilbrand S, Flodmark C, Ekbom A, Gerdin B (2003) Activation markers of connective tissue in Dupuytren’s contracture: relation to postoperative outcome. Scand J Plast Reconstr Surg Hand Surg 37:283–292
Beldner S (2013) Dupuytren’s contracture. Orthopedics 36:929–930. doi:10.3928/01477447-20131120-05
Ryhanen J, Forsman M (2012) Dupuytren’s contracture. Duodecim 128:421–429
Krüger-Sayn M, Porzberg G, Paschmeyer H (1998) Does the open palm technique for surgery of Dupuytren’s contracture extend treatment and disability duration? A retrospective study. [Handchirurgie, Mikrochirurgie, plastische Chirurgie: Organ der Deutschsprachigen Arbeitsgemeinschaft fur Handchirurgie: Organ der Deutschsprachigen Arbeitsgemeinschaft fur Mikrochirurgie der Peripheren Nerven und Gefasse]. Organ der V 30:269–271
Shaw RB Jr, Chong AK, Zhang A, Hentz VR, Chang J (2007) Dupuytren’s disease: history, diagnosis, and treatment. Plast Reconstr Surg 120:44e–54e
Satish L, LaFramboise WA, O’Gorman DB et al (2008) Identification of differentially expressed genes in fibroblasts derived from patients with Dupuytren’s contracture. BMC Med Genomics 1:10. doi:10.1186/1755-8794-1-10
Pan D, Watson HK, Swigart C, Thomson JG, Honig SC, Narayan D (2003) Microarray gene analysis and expression profiles of Dupuytren’s contracture. Ann Plast Surg 50:618–622
Bayat A, Walter J, Lambe H, Watson JS, Stanley JK, Marino M, Ferguson MW, Ollier WE (2005) Identification of a novel mitochondrial mutation in Dupuytren’s disease using multiplex DHPLC. Plast Reconstr Surg 115:134–141
Hu F, Nystrom A, Ahmed A et al (2005) Mapping of an autosomal dominant gene for Dupuytren’s contracture to chromosome 16q in a Swedish family. Clin Genet 68:424–429
Shih B, Wijeratne D, Armstrong DJ, Lindau T, Day P, Bayat A (2009) Identification of biomarkers in Dupuytren’s disease by comparative analysis of fibroblasts versus tissue biopsies in disease-specific phenotypes. J Hand Surg Am 34:124–136
Kraljevic Pavelic S, Sedic M, Hock K et al (2009) An integrated proteomics approach for studying the molecular pathogenesis of Dupuytren’s disease. J Pathol 217:524–533
Casalone R, Mazzola D, Meroni E et al (1997) Cytogenetic and interphase cytogenetic analyses reveal chromosome instability but no clonal trisomy 8 in Dupuytren contracture. Cancer Genet Cytogenet 99:73–76
Bayat A, Watson JS, Stanley JK, Ferguson MW, Ollier WE (2003) Genetic susceptibility to dupuytren disease: association of Zf9 transcription factor gene. Plast Reconstr Surg 111:2133–2139
Augoff K, Kula J, Gosk J, Rutowski R (2005) Epidermal growth factor in Dupuytren’s disease. Plast Reconstr Surg 115:128–133
Hinz B, Gabbiani G (2012) The role of the myofibroblast in Dupuytren’s disease: fundamental aspects of contraction and therapeutic perspectives. In: Dupuytren’s disease and related hyperproliferative disorders. Springer, Basel, Switzerland, pp 53–60
Ji X, Tian F, Tian L (2015) Identification and function analysis of contrary genes in Dupuytren’s contracture. Mol Med Rep 12:482–488
Smyth GK (2005) Limma: linear models for microarray data. In: Bioinformatics and computational biology solutions using R and bioconductor. Springer, New York, pp 397–420
Ashburner M, Ball CA, Blake JA et al (2000) Gene Ontology: tool for the unification of biology. Nat Genet 25:25–29
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30
Alvord G, Roayaei J, Stephens R, Baseler MW, Lane HC, Lempicki RA (2007) The DAVID gene functional classification tool: a novel biological module-centric algorithm to functionally analyze large gene lists. Genome Biol 8:R183
Franceschini A, Szklarczyk D, Frankild S et al (2013) STRING v9. 1: protein–protein interaction networks, with increased coverage and integration. Nucleic Acids Res 41:D808–D815
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Nepusz T, Yu H, Paccanaro A (2012) Detecting overlapping protein complexes in protein–protein interaction networks. Nat Methods 9:471–472
Janaki C, Joshi RR (2004) Motif detection in arabidopsis: correlation with gene expression data. In Silico Biol 4:149–161
Maere S, Heymans K, Kuiper M (2005) BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 21:3448–3449
Davis AP, King BL, Mockus S, Murphy CG, Saraceni-Richards C, Rosenstein M, Wiegers T, Mattingly CJ (2011) The comparative toxicogenomics database: update 2011. Nucleic Acids Res 39:D1067–D1072
Vaughan MB, Howard EW, Tomasek JJ (2000) Transforming growth factor-β1 promotes the morphological and functional differentiation of the myofibroblast. Exp Cell Res 257:180–189
Al-Qattan MM (2006) Factors in the pathogenesis of Dupuytren’s contracture. J Hand Surg Am 31:1527–1534
Petrov VV, Fagard RH, Lijnen PJ (2002) Stimulation of collagen production by transforming growth factor-β1 during differentiation of cardiac fibroblasts to myofibroblasts. Hypertension 39:258–263
Kuwahara F, Kai H, Tokuda K, Kai M, Takeshita A, Egashira K, Imaizumi T (2002) Transforming growth factor-β function blocking prevents myocardial fibrosis and diastolic dysfunction in pressure-overloaded rats. Circulation 106:130–135
Horiguchi M, Ota M, Rifkin DB (2012) Matrix control of transforming growth factor-β function. J Biochem 152:321–329
Wagenseil JE, Mecham RP (2007) New insights into elastic fiber assembly. Birth Defects Res C Embryo Today 81:229–240
Forrester HB, Li J, Leong T, McKay MJ, Sprung CN (2014) Identification of a radiation sensitivity gene expression profile in primary fibroblasts derived from patients who developed radiotherapy-induced fibrosis. Radiother Oncol 111:186–193
Nishimura A, Sakai H, Ikegawa S et al (2007) FBN2, FBN1, TGFBR1, and TGFBR2 analyses in congenital contractural arachnodactyly. Am J Med Genet A 143:694–698
Durany N, Michel T, Zöchling R, Boissl KW, Cruz-Sánchez FF, Riederer P, Thome J (2001) Brain-derived neurotrophic factor and neurotrophin 3 in schizophrenic psychoses. Schizophr Res 52:79–86
Howe EN, Cochrane DR, Cittelly DM, Richer JK (2012) miR-200c targets a NF-κB up-regulated TrkB/NTF3 autocrine signaling loop to enhance anoikis sensitivity in triple negative breast cancer. PLoS ONE 7:e49987
Dolcet X, Llobet D, Pallares J, Matias-Guiu X (2005) NF-kB in development and progression of human cancer. Virchows Arch 446:475–482
Bonni A, Brunet A, West AE, Datta SR, Takasu MA, Greenberg ME (1999) Cell survival promoted by the Ras-MAPK signaling pathway by transcription-dependent and-independent mechanisms. Science 286:1358–1362
Hirano S, Rees RS, Gilmont RR (2002) MAP kinase pathways involving hsp27 regulate fibroblast-mediated wound contraction. J Surg Res 102:77–84
Krause C, Kloen P, ten Dijke P (2011) Elevated transforming growth factor B and mitogen-activated protein kinase pathways mediate fibrotic traits of Dupuytren’s disease fibroblasts. Fibrogenesis Tissue Repair 4:14
Smith AG, Lim W, Pearen M, Muscat GE, Sturm RA (2011) Regulation of NR4A nuclear receptor expression by oncogenic BRAF in melanoma cells. Pigment Cell Melanoma Res 24:551–563
Palumbo-Zerr K, Zerr P, Distler A et al (2015) Orphan nuclear receptor NR4A1 regulates transforming growth factor-[beta] signaling and fibrosis. Nat Med 21:150–158
Mulhall JP, Thom J, Lubrano T, Shankey TV (2001) Basic fibroblast growth factor expression in Peyronie’s disease. J Urol 165:419–423
IJzer J, Roskams T, Molenbeek RF, Ultee T, Penning LC, Rothuizen J, Van den Ingh TS (2006) Morphological characterisation of portal myofibroblasts and hepatic stellate cells in the normal dog liver. Comp Hepatol 5:1–9
Nguyen N, Lee SB, Lee YS, Lee K-H, Ahn J-Y (2009) Neuroprotection by NGF and BDNF against neurotoxin-exerted apoptotic death in neural stem cells are mediated through Trk receptors, activating PI3-kinase and MAPK pathways. Neurochem Res 34:942–951
Stawowy P, Margeta C, Blaschke F, Lindschau C, Spencer-Hänsch C, Leitges M, Biagini G, Fleck E, Graf K (2005) Protein kinase C epsilon mediates angiotensin II-induced activation of β1-integrins in cardiac fibroblasts. Cardiovasc Res 67:50–59
Bogatkevich GS, Gustilo E, Oates JC, Feghali-Bostwick C, Harley RA, Silver RM, Ludwicka-Bradley A (2005) Distinct PKC isoforms mediate cell survival and DNA synthesis in thrombin-induced myofibroblasts. Am J Physiol Lung Cell Mol Physiol 288:L190–L201
Ribas J, Michailidi C, Perez J, Soudry E, Tapiaz O, Guzman P, Munoz S (2014) Genome-wide methylation profiling reveals Zinc finger protein 516 (ZNF516) and FK-506-binding protein 6 (FKBP6) promoters frequently methylated in cervical neoplasia, associated with HPV status and ethnicity in a Chilean population. Epigenetics 9:1–10
Zhang Z, Xia W, He J et al (2012) Exome sequencing identifies SLCO2A1 mutations as a cause of primary hypertrophic osteoarthropathy. Am J Hum Genet 90:125–132
Wang J, Robinson JF, O’Neil CH, Edwards JY, Williams CM, Huff MW, Pickering JG, Hegele RA (2006) Ankyrin G overexpression in Hutchinson–Gilford progeria syndrome fibroblasts identified through biological filtering of expression profiles. J Hum Genet 51:934–942
Vidhya G, Anusha B (2014) Diaretinopathy database—a gene database for diabetic retinopathy. Bioinformation 10:235
Townley W, Baker R, Sheppard N, Grobbelaar A (2006) Dupuytren’s contracture unfolded. BMJ 332:397–400
Author information
Authors and Affiliations
Corresponding author
Additional information
Haoyu Liu and Weitian Yin have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Liu, H., Yin, W., Liu, B. et al. Screening of candidate genes in fibroblasts derived from patients with Dupuytren’s contracture using bioinformatics analysis. Rheumatol Int 35, 1343–1350 (2015). https://doi.org/10.1007/s00296-015-3276-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-015-3276-3