Summary
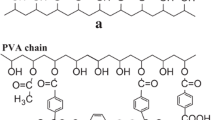
The dipolar cycloaddition of nitrile oxides with species containing unsaturation is a very attractive means of forming crosslinks in polymer systems, since the reaction is rapid and yields thermally stable links. Latent nitrile oxide precursors can be prepared by reaction of an isocyanate and an alkyl nitroacetate. These precursors release alkanol and carbon dioxide when heated to liberate the highly reactive nitrile oxide species. An improved synthetic procedure was developed to afford novel crosslinking agents based on difunctional, trifunctional and aliphatic precursors. Further, application of these agents to polymer crosslinking has been demonstrated.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 20 April 2001/Revised version: 29 August 2001/Accepted: 29 August 2001
Rights and permissions
About this article
Cite this article
Huffman, B., Schultz, R. & Schlom, P. Novel reagents for heat-activated polymer crosslinking. Polymer Bulletin 47, 159–166 (2001). https://doi.org/10.1007/s002890170007
Issue Date:
DOI: https://doi.org/10.1007/s002890170007