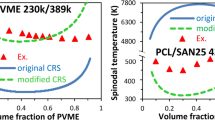
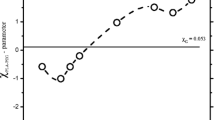
Abstract
This paper focuses on the theoretical and experimental phase behavior determination of polyvinylchloride/polycaprolactone (PVC/PCL) blend. For this aim, the capability of the compressible regular solution (CRS) model to predict the phase behavior of the PVC/PCL blend was evaluated. The extreme sensitivity of the CRS model to the solubility parameters of the blend components and the lack of a unique value for the solubility parameters of each polymer led to various phase behavior predictions from upper critical solution temperature to complete immiscibility. To verify the CRS model predictions, dilute solution viscometry (DSV), Fourier transform infrared and differential scanning calorimetry were employed. The last two showed complete miscibility, while different DSV criteria indicated partial miscibility, which were in contrast to the predictions made by the CRS model. Finally, the dynamic phase diagram of the PVC/PCL blend was determined via rheological measurements for the first time, suggesting lower critical solution temperature behavior with the critical point located at 195 °C.












Similar content being viewed by others
References
Laukaitiene A, Jankauskaite V, Zukiene K, Norvydas V, Munassipov S, Janakhmetov U (2013) Investigation of polyvinyl chloride and thermoplastic polyurethane waste blend miscibility. Mater Sci 19:397–402
Fashandi H, Karimi M (2014) Influence of solvent/polymer interaction on miscibility of PMMA/PCL blend: thermal analysis approach. Text Polym 2:71–78
White RP, Lipson JEG, Higgins JS (2012) How pure components control polymer blend miscibility. Macromolecules 45(21):8861–8871. https://doi.org/10.1021/ma3018124
Kuo SW, Lin CL, Chang FC (2002) Phase behavior and hydrogen bonding in ternary polymer blends of phenolic resin/poly(ethylene oxide)/poly(e-caprolactone). Macromolecules 35:278–285
Keshavarz L, Khansary MA, Shirazian S (2015) Phase diagram of ternary polymeric solutions containing nonsolvent/solvent/polymer: theoretical calculation and experimental validation. Polymer 73:1–8. https://doi.org/10.1016/j.polymer.2015.07.027
Ruzette AVG, Mayes AM (2001) A Simple free energy model for weakly interacting polymer blends. Macromolecules 34(6):1894–1907. https://doi.org/10.1021/ma000712+
Clark EA, Lipson JEG (2012) LCST and UCST behavior in polymer solutions and blends. Polymer 53(2):536–545. https://doi.org/10.1016/j.polymer.2011.11.045
Panuganti SR, Tavakkoli M, Vargas FM, Gonzalez DL, Chapman WG (2013) SAFT model for upstream asphaltene applications. Fluid Ph Equilib 359:2–16. https://doi.org/10.1016/j.fluid.2013.05.010
Yeganeh JK, Goharpey F, Foudazi R (2014) Anomalous phase separation behavior in dynamically asymmetric LCST polymer blends. RSC Adv 4(25):12809. https://doi.org/10.1039/c3ra47299j
Li H, Zhang Y, Jiacuo Y, Shang Y, Huo H, Jiang Sh (2012) Miscibility and rheologically determined phase diagram of poly(ethylene oxide)/poly(e-caprolactone) blends. Polym Bull 68:1405–1423
Lyu Y, Pang J, Gao Z, Zhang Q, Shi X (2019) Characterization of the compatibility of PVC/PLA blends by aid of rheological responses. Polymer 176:20–29
Niu YH, Wang ZG (2006) Rheologically determined phase diagram and dynamically investigated phase separation kinetics of polyolefin blends. Macromolecules 39:4175–4183
Zou F, Dong X, Liu W, Yang J, Lin D, Liang A, Li W, Han CH (2012) Shear induced phase boundary shift in the critical and off-critical regions for a polybutadiene/polyisoprene blend. Macromolecules 45:1692–1700
Chen H-L, Li L-J, Lin T-L (1998) Formation of segregation morphology in crystalline/amorphous polymer blends: molecular weight effect. Macromolecules 31(7):2255–2264. https://doi.org/10.1021/ma9715740
Mamun Al, Mareau VH, Chen J, Prud’homme RE (2014) Morphologies of miscible PCL/PVC blends confined in ultrathin films. Polymer 55(9):2179–2187. https://doi.org/10.1016/j.polymer.2014.03.010
Rusu M, Ursu M, Rusu D (2006) Poly(vinyl chloride) and poly(e-caprolactone) blends for medical use. Thermoplast Compos Mater 19:173–190
Yang X, Xiong Y, Guo S (2020) Effect of liquid plasticizers on crystallization of PCL in soft PVC/PCL/plasticizer blends. J Appl Polym Sci 137(24):48803. https://doi.org/10.1002/app.48803
Sun Z, Choi B, Feng A, Moad G, Thang SH (2019) Nonmigratory poly (vinyl chloride)-block-polycaprolactone plasticizers and compatibilizers prepared by sequential RAFT and ring-opening polymerization (RAFT-T̵-ROP). Macromolecules 52(4):1746–1756
Nguyen-Tri P, Carrière P, Duong A, Nanda S (2020) Graphene oxide-induced interfacial transcrystallization of single-fiber milkweed/polycaprolactone/polyvinylchloride composites. ACS Omega 5(35):22430–22439
Imran Khan M, Zagho MM, Shakoor RA (2017) A Brief overview of shape memory effect in thermoplastic polymers. In: Ponnamma D, Sadasivuni KK, Cabibihan J-J, Al-Maadeed M A-A (eds) Smart polymer nanocomposites. Springer International Publishing, Cham, pp 281–301. https://doi.org/10.1007/978-3-319-50424-7_10
Pekdemir ME, Öner E, Kök M, Qader IN (2021) Thermal behavior and shape memory properties of PCL blends film with PVC and PMMA polymers. Iran Polym J 30(6):633–641
Liu W, Zhang R, Huang M, Dong X, Xu W, Ray N, Zhu J (2016) Design and structural study of a triple-shape memory PCL/PVC blend. Polymer 104:115–122
Koleske JV, Lundberg RD (1969) Lactone polymers. I. Glass transition temperature of poly-ɛ-caprolactone by means on compatible polymer mixtures. J Polym Sci Part A-2 Polym Phys 7(5):795–807. https://doi.org/10.1002/pol.1969.160070505
Khambatta FB, Warner F, Russell T, SteinR S (1976) Small-angle X-ray and light scattering studies of the morphology of blends of poly(e-caprolactone) with poly(vinylChloride). Polym Sci 14:1391–1424
Klee D, Höcker H (2000) Advances in polymer science: biomedical applications/polymer blends. Springer, s.l.
Ong CJ, Price FP (2007) Blends of poly(ϵ-caprolactone) with poly(vinyl chloride). I. morphology. J Polym Sci Polym Symp 63(1):45–58. https://doi.org/10.1002/polc.5070630108
Ong CJ, Price FP (2007) Blends of poly(ϵ-caprolactone) with poly(vinyl chloride). II. crystallization kinetics. J Polym Sci Polym Symp 63(1):59–75. https://doi.org/10.1002/polc.5070630109
Russell TP, Stein RS (1983) An investigation of the compatibility and morphology of semicrystalline poly(ɛ-caprolactone)–poly(vinyl chloride) blends. J Polym Sci Polym Phys Ed 21(7):999–1010. https://doi.org/10.1002/pol.1983.180210701
Robeson LM (1973) Crystallization kinetics of poly-ɛ-caprolactone from poly-ɛ-caprolactone/poly(vinyl chloride) solutions. J Appl Polym Sci 17(12):3607–3617. https://doi.org/10.1002/app.1973.070171205
Haiyang Y, Pingping Z, Shiqiang W, Yiming Z, Qipeng G (1998) Viscometric study of polymer–polymer interactions in ternary systems—II. The influence of solvent. Eur Polym J 34(9):1303–1308. https://doi.org/10.1016/S0014-3057(97)00260-7
Olabisi O (1975) Polymer compatibility by gas-liquid chromatography. Macromolecules 8:316–322
Natansohn A (1985) Exothermal effect of phase separation in miscible polymer blends. Polym Sci 23:305–308
Bustamante P, Navarro-Lupion J, Escalera B (2005) A new method to determine the partial solubility parameters of polymers from intrinsic viscosity. Eur J Pharm Sci 24:229–237
Mei S, Xiao C, Hu X (2012) Interfacial microvoid formation of poly(vinyl chloride)/polyacrylonitrile blend hollow-fiber membranes. Appl Polym Sci 124:E9–E16
Sun Z, Wang W, Feng Z (1992) Criterion of polymer-polymer miscibility determined by viscometry. Eur Polym J 28(10):1259–1261. https://doi.org/10.1016/0014-3057(92)90215-N
Brunchi CE, Bercea M, Morariu S (2010) Hydrodynamic properties of polymer mixtures in solution. Chem Eng Data 55:4399–4405
Kapnistos M, Vlassopoulos D, Anastasiadis SH (1996) Determination of both the binodal and the spinodal curves in polymer blends by shear rheology. Europhys Lett 34:513–518
Ajji A, Choplin L (1991) Rheology and dynamics near phase separation in a polymer blend: model and scaling analysis. Macromolecules 24:5221–5223
Naziri AA, Khonakdar HA, Hemmati F, Jafari SH, Ehsani M (2019) Rheologically determined phase diagram of poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blends: role of ramp rate in dynamic measurements. J Appl Polym Sci 136(28):47750
Van Krevelen DW, Te Nijenhuis K (2009) Properties of polymers. Elsevier, Fourth. s.l.
Utrachi LA, Wilkie CA (2014) Polymer blends hand book. Springer, London
Pingping Z, Haiyang Y, Shiqiang W (1998) Viscosity behavior of poly(caprolactone) (PCL)/poly(vinyl chloride)(PVC) blends in various solvents. Eur Polym J 34:91–94
Pandey M, Joshi GM, Mukherjee A, Thomas P (2016) Electrical properties and thermal degradation of poly (vinyl chloride)/polyvinylidene fluoride/ZnO polymer nanocomposites. Polym Int 65(9):1098–1106
Bulla SS, Bhajantri RF, Chavan C, Sakthipandi K (2021) Synthesis and characterization of polythiophene/zinc oxide nanocomposites for chemiresistor organic vapor-sensing application. J Polym Res 28(7):1–21
Ramesh S, Leen KH, Kumutha K, Arof AK (2007) FTIR studies of PVC/PMMA blend based polymer electrolytes. Spectrochim Acta Part A Mol Biomol Spectrosc 66(4–5):1237–1242. https://doi.org/10.1016/j.saa.2006.06.012
Abdelrazek EM, Hezma AM, El-Khodary A, Elzayat AM (2016) Spectroscopic studies and thermal properties of PCL/PMMA biopolymer blend. Egypt J Basic Appl Sci 3(1):10–15
Coleman MM, Zarian J (1979) Fourier-transform infrared studies of polymer blends. II. Poly(ε-caprolactone)–poly(vinyl chloride) system. J Polym Sci Polym Phys Ed 17(5):837–850. https://doi.org/10.1002/pol.1979.180170509
Bélorgey G, Aubin M, Prud’homme RE (1982) Studies of polyester/chlorinated poly(vinyl chloride) blends. Polymer 23(7):1051–1056. https://doi.org/10.1016/0032-3861(82)90407-4
Chiu FC, Min K (2000) Miscibility, morphology and tensile properties of vinyl chloride polymer and poly(ε-caprolactone) blends. Polym Int 49:223–234
Wang J, Cheung MK, Mi Y (2002) Miscibility and morphology in crystalline/amorphous blends of poly(caprolactone)/poly(4vinylphenol) as studied by DSC, FTIR, and Csolid state NMR. Polymer 43:1357–1364
Gharachorlou A, Goharpey F (2008) Rheologically determined phase behavior of LCST blends in the presence of spherical nanoparticles. Macromolecules 41(9):3276–3283
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raftari, R., Maghsoud, Z. Evaluation of the compressible regular solution model predictions via rheologically determined phase diagram for polyvinylchloride/polycaprolactone blend. Polym. Bull. 80, 3227–3246 (2023). https://doi.org/10.1007/s00289-022-04212-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04212-3